Polarity in Covalent Bonds Bonding Movie Polar Bond

Polarity in Covalent Bonds Bonding Movie

Polar Bond • Polar – has poles – North pole / South pole = magnetic poles – The ends are different – In bonds: the difference is in how the electrons are distributed – Polar bond: one end has more electrons than the other end

Nonpolar Bond • Nonpolar = No poles Non • The electron cloud on one end of the bond is the same as the other end
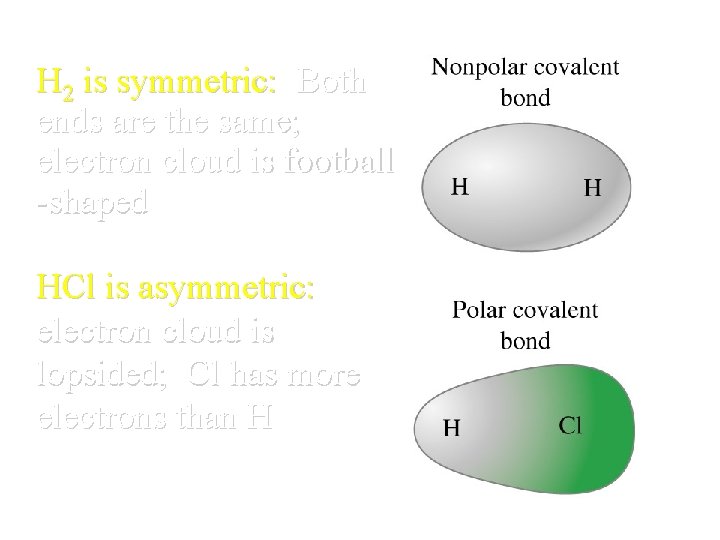
H 2 is symmetric: Both ends are the same; electron cloud is football -shaped HCl is asymmetric: electron cloud is lopsided; Cl has more electrons than H

Which bond(s) are polar? Which are nonpolar? Polar = Li. H and HF Nonpolar = H 2 Red = electron rich Blue = electron poor
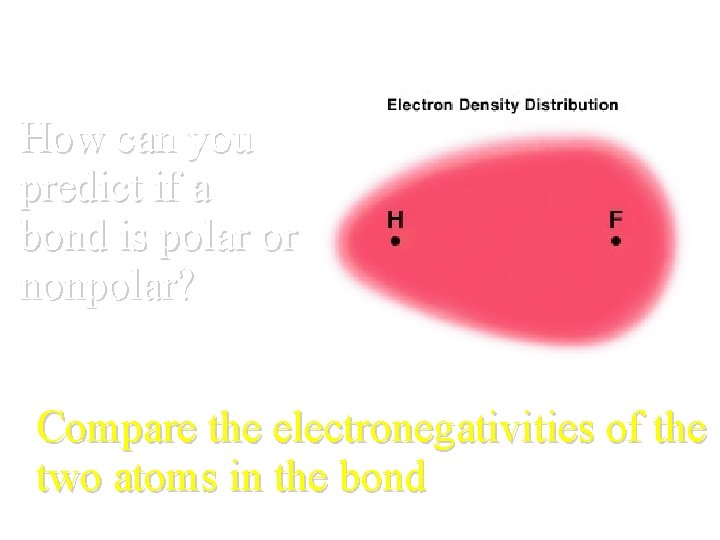
How can you predict if a bond is polar or nonpolar? Compare the electronegativities of the two atoms in the bond

Electronegativity • Ability of an atom to attract electrons in a bond Found in Table S!
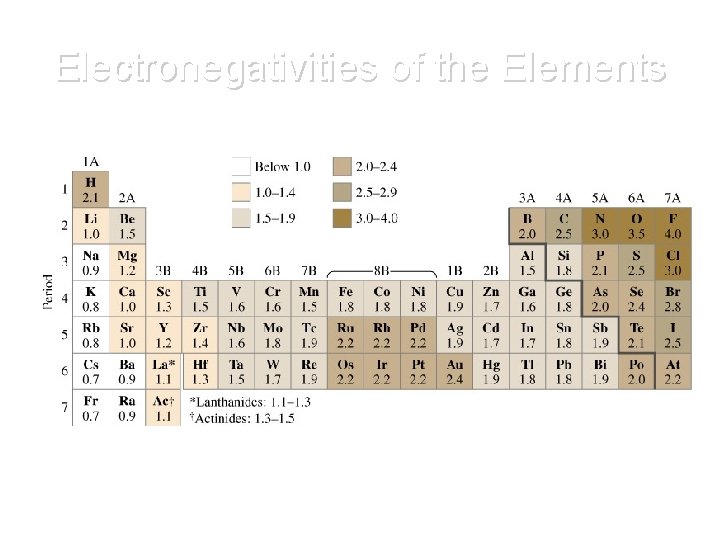
Electronegativities of the Elements

Bond Polarity • The more electronegative atom pulls the bonding electrons more strongly and gets more than its fair share of the electron cloud • Leads to separation of charge – electron-rich side is partially negative ( -) – electron-poor side is partially positive ( +)

Delta notation

Bond Polarity • Depends on the difference in electronegativity between two atoms in the bond A B EA - EB • - care only about the size of the difference, not the sign • - bigger the difference, the more polar the bond
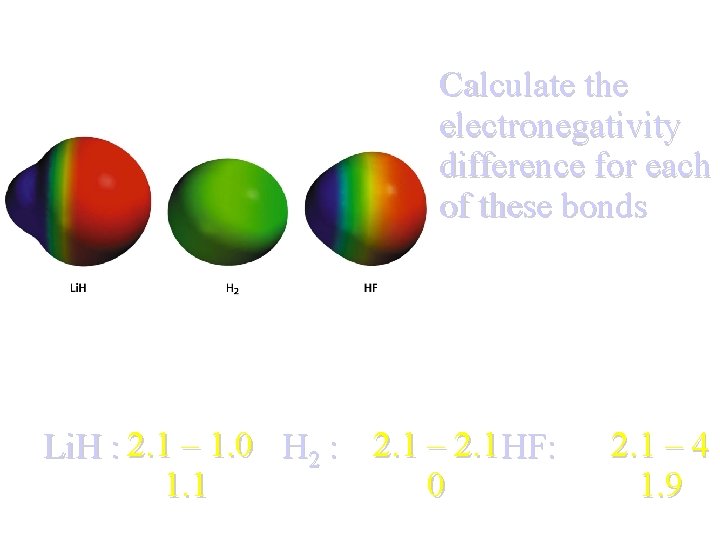
Calculate the electronegativity difference for each of these bonds Li. H : 2. 1 – 1. 0 H 2 : 2. 1 – 2. 1 HF: 0 1. 1 2. 1 – 4 1. 9

Bond Polarity • What can you say about the polarity of any bond where atom A = atom B? If both atoms in the bond are the same element: the electronegativity difference = 0

Electronegativity Difference Type of Bond 0. 0 to 0. 4 Nonpolar Covalent 0. 5 to 1. 7 Polar Covalent > 1. 7 Ionic
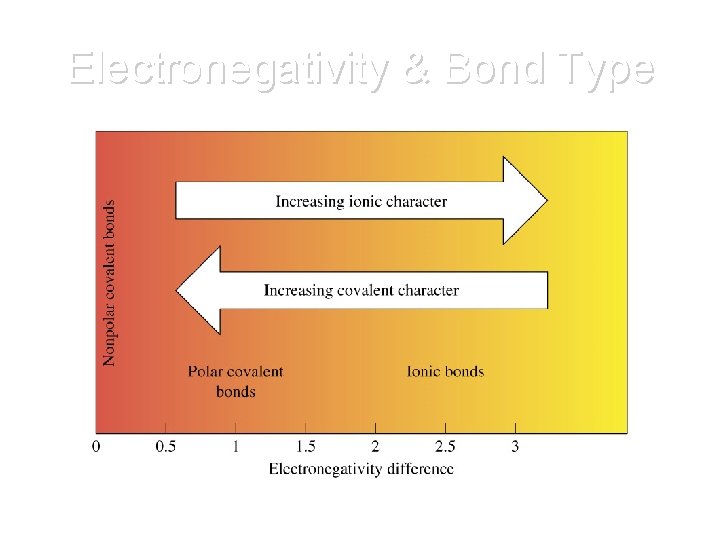
Electronegativity & Bond Type

Review • Which of the following bonds is the most polar? – A) O 2 3. 4 – 3. 4 = 0 – B) HCl 2. 1 – 3. 2 = -1. 1 = 1. 1 – C) NH in NH 3 3. 0 – 2. 1 = 0. 9 – D) HBr 3. 0 – 2. 1 = 0. 9 Answer = (B) HCl

Review • Which substance contains a bond with the greatest ionic character? – A) KCl 0. 8 – 3. 2 = -2. 4 = 2. 4 – B) HCl 2. 1 – 3. 2 = -1. 1 = 1. 1 – C) Cl 2 3. 2 – 3. 2 = 0 – D) CCl 4 2. 6 – 3. 2 = -0. 6 = 0. 6 Answer = (A) KCl

Summary • Nonpolar covalent bonds form: – between atoms having equal or close electronegativity values: 0. 0 - 0. 5 • Polar covalent bonds form: – between atoms with an electronegativity difference between 0. 5 and 1. 7

Predict the Polarity • • N 2 Nonpolar HF Polar HCl Polar O 2 Nonpolar Cl 2 Nonpolar HI Polar HBr Polar

Molecular Polarity • Diatomic molecules: – molecular polarity is same as bond polarity • Molecules with 3 or more atoms: – polarity depends on: • types of bonds and • shape of the molecule

Molecular Polarity • Depends on 2 factors 1. Type of bonds in the molecule 2. Arrangement of bonds or shape of molecule Think SYMMETRY!!
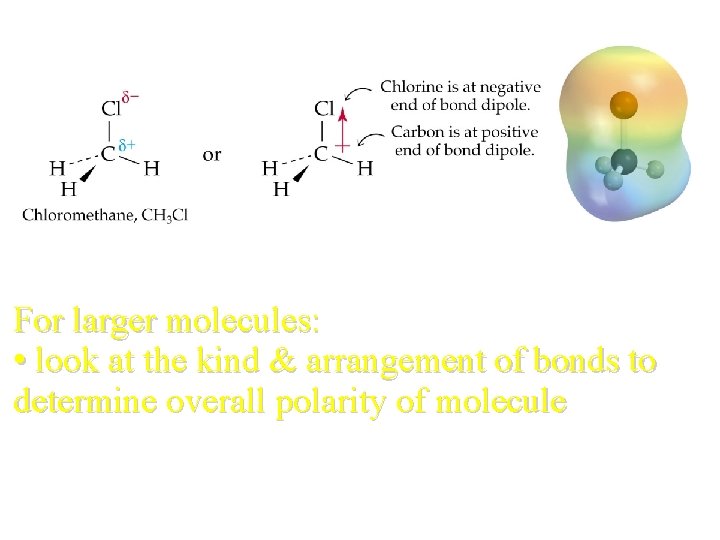
For larger molecules: • look at the kind & arrangement of bonds to determine overall polarity of molecule

Polarity of Molecules • A molecule may contain polar bonds, but not be polar! – Depends on the geometry of the molecule • If molecule is symmetric: – the “pull” of one polar bond is offset by the “pull” of another polar bond – It’s a tug-of-war that no one can win!
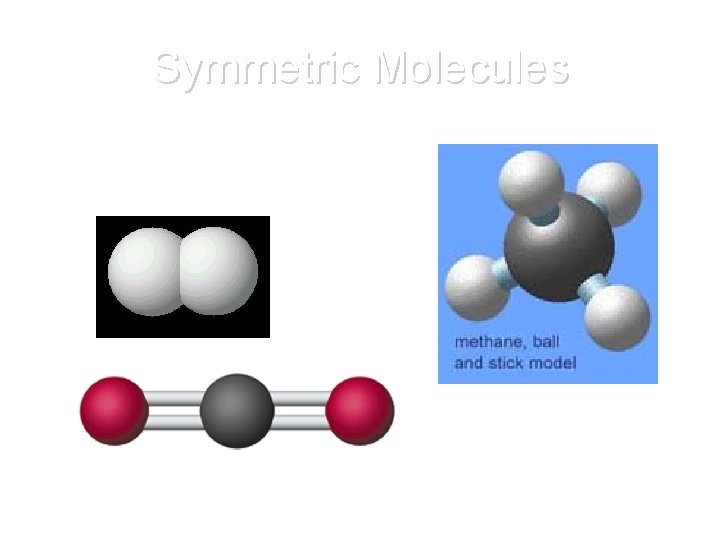
Symmetric Molecules • Contain at least one mirror plane
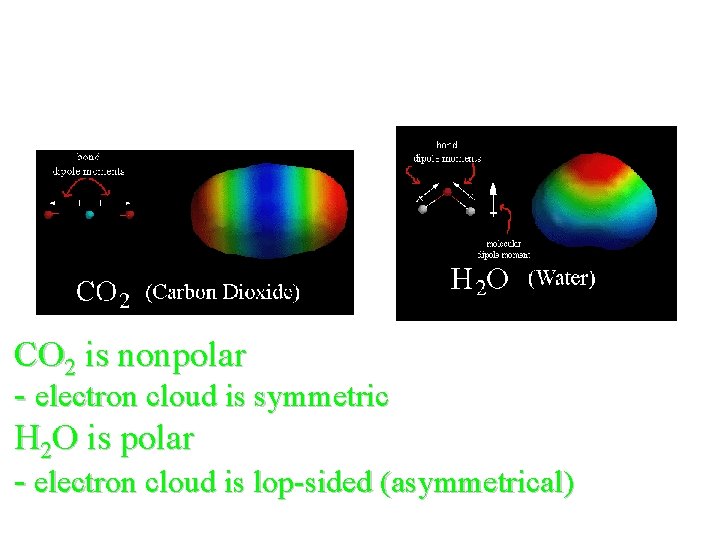
Symmetric vs. Asymmetric CO 2 is nonpolar - electron cloud is symmetric H 2 O is polar - electron cloud is lop-sided (asymmetrical)
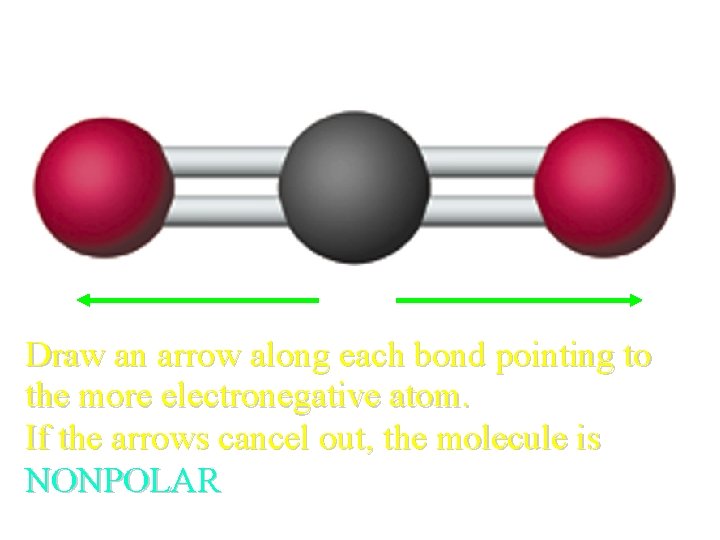
Polarity of CO 2? Draw an arrow along each bond pointing to the more electronegative atom. If the arrows cancel out, the molecule is NONPOLAR
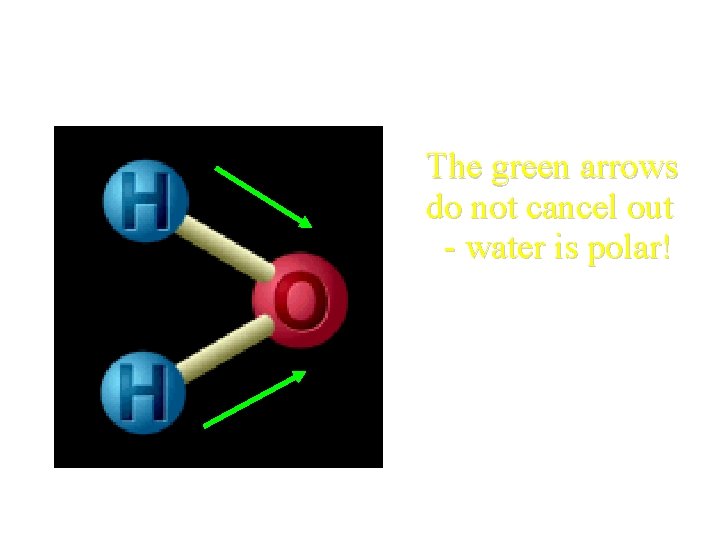
Polarity of H 2 O? The green arrows do not cancel out - water is polar!
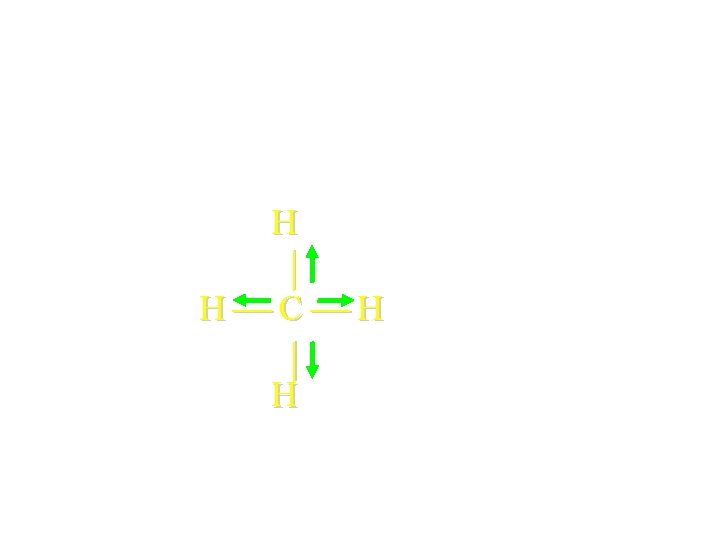
Use the structural formula to predict Molecular Polarity! H H C H H

Symmetry of Larger Molecules CF 4 is fairly symmetric so it is nonpolar
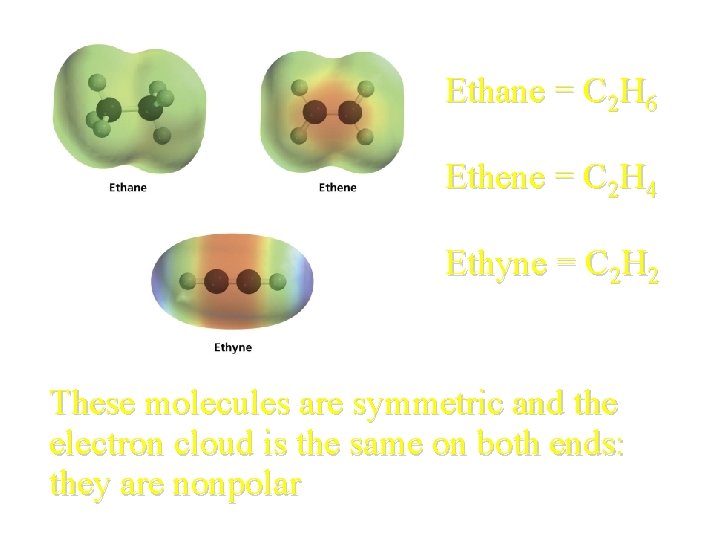
Ethane = C 2 H 6 Ethene = C 2 H 4 Ethyne = C 2 H 2 These molecules are symmetric and the electron cloud is the same on both ends: they are nonpolar
- Slides: 30