Polar Covalent Bonds Polar bond A type of

Polar Covalent Bonds

Polar bond A type of covalent bond between two atoms in which electrons are shared unequally, resulting in a bond in which one atom has a slightly negative charge and the other a slightly positive charge. In a polar molecule, the ends of the molecule (called dipoles) carry an unequal charge.

Water is polar A polar molecule is water soluble. In order for a solution to form with molecules, they must be either both polar or both non-polar.

solutions water is a polar molecule - so is alcohol. When you mix water and alcohol you see a solution with both molecules in the solution. The molecules mix together. When you try to mix a polar and a non polar molecule what happens? They don't mix. water (a polar molecule) does not mix with oil (a non polar molecule).

To determine a molecule's polarity: Determine each atom's electronegativity.

Electronegativity • is defined as the ability of an atom in a particular molecule to attract electrons to itself • (the greater the value, the greater the attractiveness for electrons)

Electronegativity is a function of: • the atom's ionization energy (how strongly the atom holds on to its own electrons) • the atom's electron affinity (how strongly the atom attracts other electrons)

An element with high electronegativity: • Attract electrons from other atoms • Resist having its own electrons attracted away • Fluorine is the most electronegative element (electronegativity = 4. 0) • the least electronegative is Cesium (notice that are at diagonal corners of the periodic chart)

General trends: • Electronegativity increases from left to right along a period • For the representative elements (s and p block) the electronegativity decreases as you go down a group • The transition metal group is not as predictable as far as electronegativity

Predicting the type of bond based upon electronegativity differences: • if the electronegativity difference is 0, the bond is non-polar covalent • If the difference in electronegativities is between 0 - 2, the bond is polar covalent • If the difference in electronegativities is 2. 0 or more, the bond is ionic

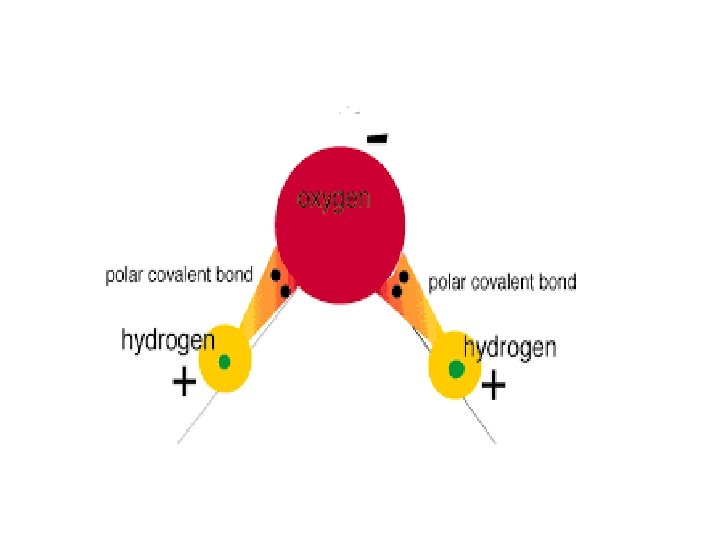
Water is polar because: • of the differences in the electronegativity between oxygen and hydrogen. • Oxygen is to the right on the periodic table, so Oxygen is highly negative compared to hydrogen.

Water Molecule Structure • The oxygen attracts negative charges making the area around the oxygen much more negative than the area around the hydrogen. • The hydrogen area (positive charges) makes the molecule bend so that the 2 hydrogen atoms in the water are on the same side, pointing away from the negative area around the oxygen.

Polar or nonpolar? F 2 the electrons are shared equally between the atoms, the bond is nonpolar covalent HF • fluorine has greater electronegativity than the hydrogen atom. • fluorine attracts electrons away from the hydrogen • The bond is a polar covalent bond
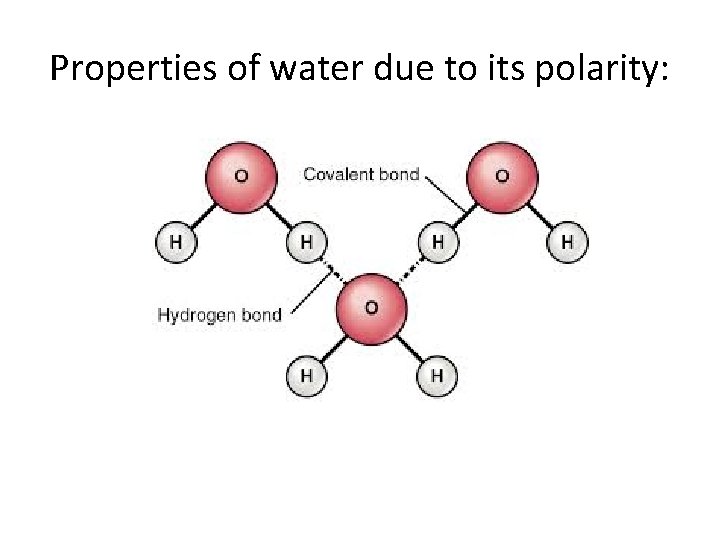
Properties of water due to its polarity:

Properties of water due to its polarity: • • • Hydrogen bonds Cohesion Adhesion Capillarity Universal solvent
- Slides: 16