Polar Bonds and Molecules Chapter 8 Section 4

Polar Bonds and Molecules Chapter 8 Section 4

Bond Polarity • Covalent bonds involve electron sharing between atoms • Each covalent bond differs in how the bonded atoms share the electrons • Bonding pairs of electrons in covalent bonds are pulled, as in tug of war, between the nuclei of the atoms sharing the electrons
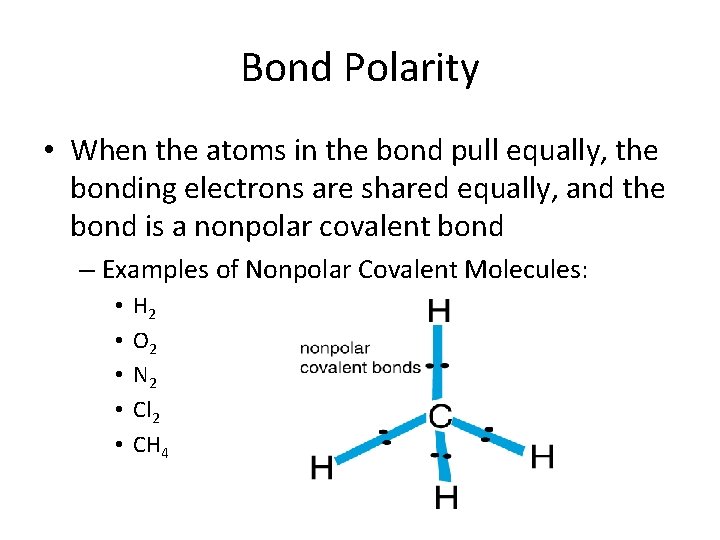
Bond Polarity • When the atoms in the bond pull equally, the bonding electrons are shared equally, and the bond is a nonpolar covalent bond – Examples of Nonpolar Covalent Molecules: • • • H 2 O 2 N 2 Cl 2 CH 4

Bond Polarity • A polar covalent bond, known also as a polar bond, is a covalent bond between atoms in which the electrons are shared unequally • The more electronegative atom attracts electrons more strongly and gains a slightly negative charge • Less electronegative atom has a slightly positive charge

Bond Polarity • Example: HCl – Hydrogen has an electronegativity of 2. 1 – Chlorine has an electronegativity of 3. 0 – These values are not equal, creating a polar bond • 3. 0 -2. 1=0. 9
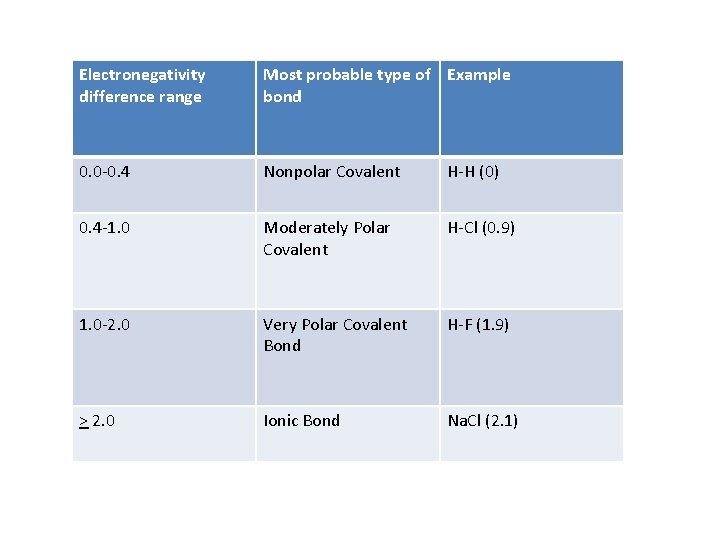
Electronegativity difference range Most probable type of Example bond 0. 0 -0. 4 Nonpolar Covalent H-H (0) 0. 4 -1. 0 Moderately Polar Covalent H-Cl (0. 9) 1. 0 -2. 0 Very Polar Covalent Bond H-F (1. 9) > 2. 0 Ionic Bond Na. Cl (2. 1)

Polar Molecules • The presence of a polar bond in a molecule often makes the entire molecule polar • In a polar molecule, one end of the molecule is slightly negative and the other end is slightly positive • A molecule that has two poles is called a dipolar molecule, or dipole

Polar Molecules • When polar molecules are placed between oppositely charged plates, they tend to become oriented with respect to the positive and negative plates.

Polar Molecules • Effect of polar bonds on the polarity of an entire molecule depends on the shape of the molecule and the orientation of the polar bonds • How is CO 2 polar while H 20 is not?

Attractions Between Molecules • Molecules can attract each other by a variety of forces • Intermolecular attractions are weaker than either ionic or covalent bonds • Attractions are responsible for determining whether a molecular compound is a gas, liquid, or a solid at a given temperature

Van der Waals Forces • Two weakest attractions between molecules are collectively called van der Waals forces – Named after Johannes van der Waals – Consist of dipole interactions and dispersion forces

Dipole Interactions • Occur when polar molecules are attracted to one another • Electrical attraction occurs between the oppositely charged regions of polar molecules Molecule of Hydrochloric Acid – Partial negative of one molecule is attracted to the partially positive region of another polar molecule • Dipole interactions are similar to but much weaker than ionic bonds Molecule of Water
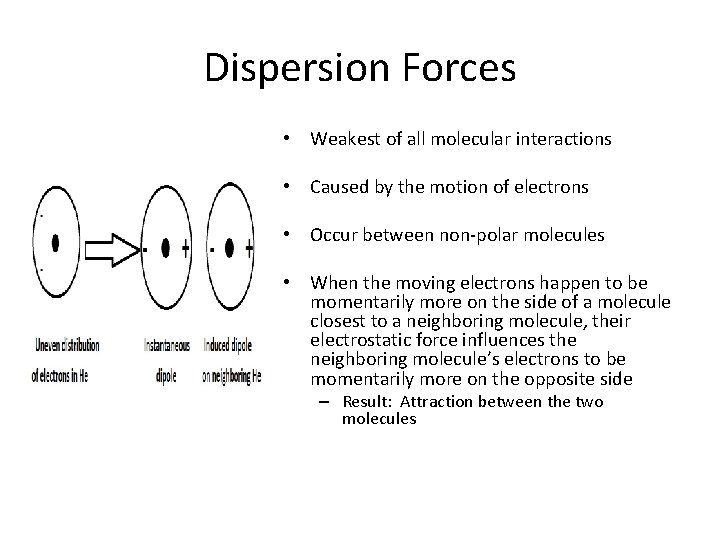
Dispersion Forces • Weakest of all molecular interactions • Caused by the motion of electrons • Occur between non-polar molecules • When the moving electrons happen to be momentarily more on the side of a molecule closest to a neighboring molecule, their electrostatic force influences the neighboring molecule’s electrons to be momentarily more on the opposite side – Result: Attraction between the two molecules

Dispersion Forces • The strength of dispersion forces generally increases as the number of electrons in a molecule increases • Halogen diatomic molecules (F, Cl, Br, I) attract each other mainly by means of dispersion forces • List them in increasing strength of dispersion forces.

Hydrogen Bonds • Attractive forces in which a hydrogen covalently bonded to a very electronegative atom is also weakly bonded to an unshared electron pair of another electronegative atom • Other atom may be in the same molecule or in a nearby molecule • For a hydrogen bond to form, a covalent bond must already exist between a hydrogen atom and a high electronegative atom

Hydrogen Bonds • The combination of this strongly polar bond and the lack of shielding effect in a hydrogen atom is responsible for the relative strength of hydrogen bonds • They are extremely important in determining the properties of water and biological molecules

Intermolecular Attractions and Molecular Properties • A great range of physical properties exist among covalent compounds due to varying intermolecular attractions – At room temperature, some compounds are gases, some liquids, some solids – Melting and boiling points are low compared to ionic compounds • Only the weak attractions between molecules need to be broken • Few solids that consist of molecules do not melt until the temperature reaches 1000°C • Most of these very stable substances are network solids, solids in which all of the atoms are covalently bonded to each other • Ex) Diamond
- Slides: 17