Polar and nonpolar covalent bonds and molecules 16

Polar and nonpolar covalent bonds and molecules 16. 3 Pages 460 -463

Vocabulary � Polar � Nonpolar � Dipole � All have the word “pole” in it, meaning “a part of a system that has opposite positions” � Ex: North Pole and South Pole refers to opposite locations and magnetic fields on earth. � In chemistry Polar refers to oppositely charged sides of a molecule

Is sharing always equal? � Main point- How bad do you want that item? � In chemistry this is electronegativity.

Electronegativity determines the bond type � We know the shortcuts for finding out the type of compound and bond formed. � Metal with non-metal = ? � Metal with metal = ? � Non-metal with non-metal = ? � This shortcut is really based on electronegativity (how much does one atom want the electrons of the other atom)
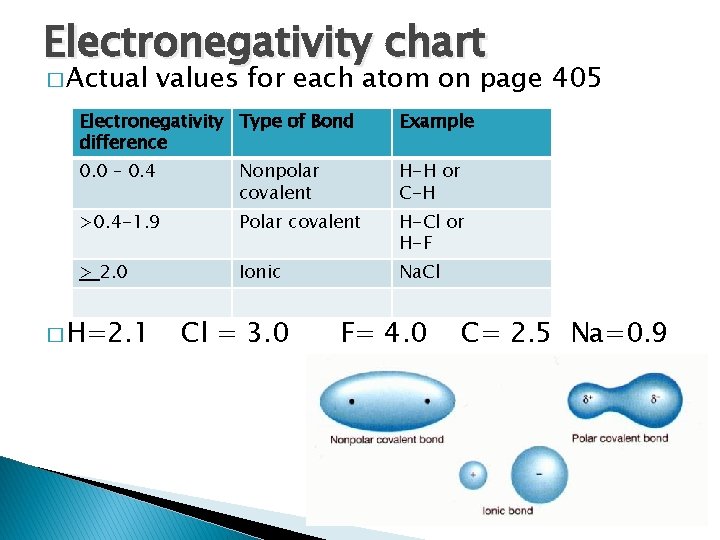
Electronegativity chart � Actual values for each atom on page 405 Electronegativity Type of Bond difference Example 0. 0 – 0. 4 Nonpolar covalent H-H or C-H >0. 4 -1. 9 Polar covalent H-Cl or H-F > 2. 0 Ionic Na. Cl � H=2. 1 Cl = 3. 0 F= 4. 0 C= 2. 5 Na=0. 9

Nonpolar covalent bonds � When both atoms pull on the electrons equally, the electrons are shared equally. � This happens when both atoms are the same (H 2, O 2, N 2, Cl 2, F 2, Br 2, I 2) � It also happens when both atoms have very similar electronegativity values � Ex: C-H, C-C

� Non-polar animation

Polar covalent bonds � When the two atoms pull on the electrons unequally. � The more electronegative atom will pull harder and acquire a slight negative charge � The less electronegative atom will pull less and acquire a slight positive charge � This is denoted by a delta (delta +, delta -)

� Polar bond animation
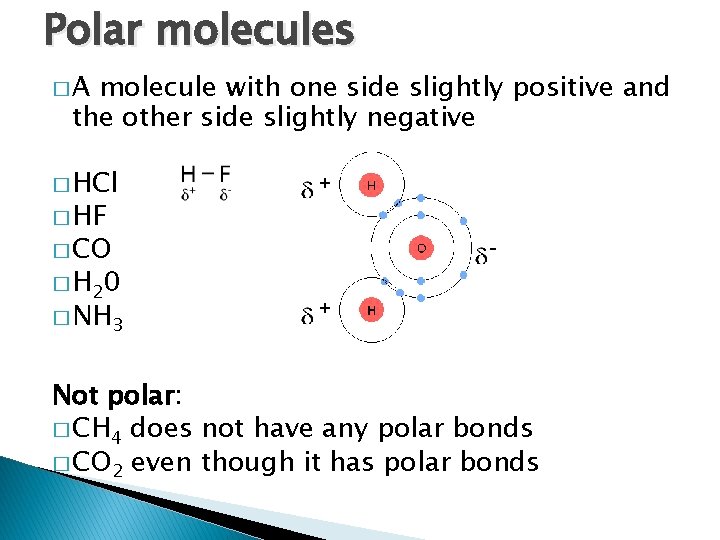
Polar molecules �A molecule with one side slightly positive and the other side slightly negative � HCl � HF � CO � H 20 � NH 3 Not polar: � CH 4 does not have any polar bonds � CO 2 even though it has polar bonds

General Rule for identifying bonds as polar or nonpolar � Same atoms on bond = non-polar bond � C and H = non-polar bond � Every other combination usually polar � Sure solution: Look up electronegativities on page 405 and compare to table on page 463
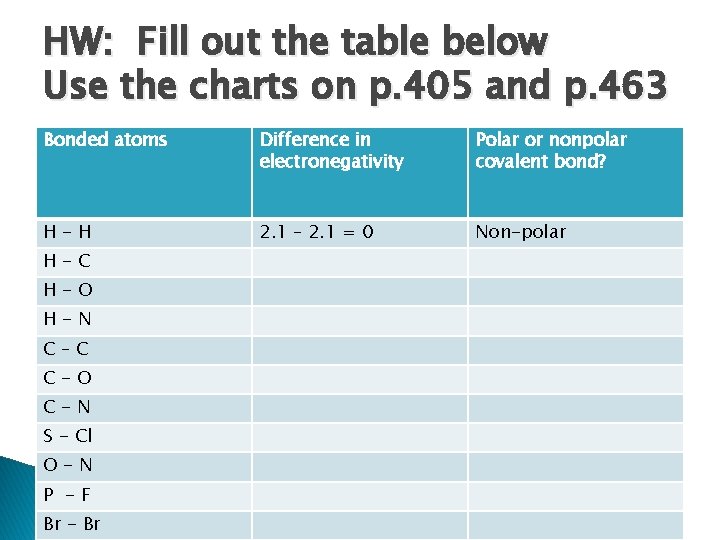
HW: Fill out the table below Use the charts on p. 405 and p. 463 Bonded atoms Difference in electronegativity Polar or nonpolar covalent bond? H-H 2. 1 – 2. 1 = 0 Non-polar H-C H-O H-N C–C C-O C-N S - Cl O-N P -F Br - Br

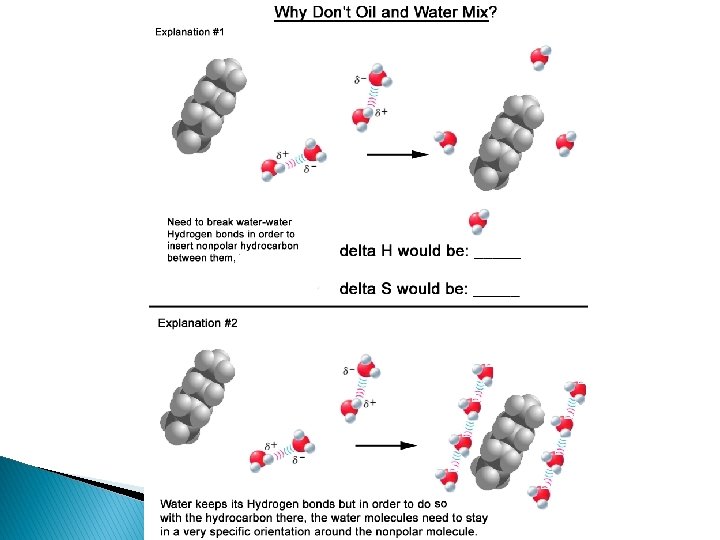
What does the term “likes dissolve likes” mean in regards to polarity? � Likes dissolve likes is a phrase that tells you which molecules will mix and which will not. � Polar dissolves polar � Nonpolar dissolves nonpolar � Polar will not mix with nonpolar � Demonstrate with a bottle that has oil and water in it � Demonstrate with two beakers, one water, one oil, and 2 cubes of sugar in each one.

The 3 intermolecular forces 16. 3 Pages 463 -466

What does the prefix “inter” mean? � “Intermolecular forces” means � “Attractions between molecules”

Intermolecular attractions � Atoms bond to have complete outer orbitals � When they get there, they stop bonding and become the compound they become � If only covalent bonds existed, each little molecule would be completely independent and every molecular compound would be a ? � Gas! � We would have no liquid!!! � So there must be OTHER interactions taking place between molecules…

The 3 weak intermolecular forces/attractions � Van Der Waals Forces ◦ 1) London Dispersion forces ◦ 2) Dipole-dipole interactions � 3) Hydrogen bonds- ◦ The strongest of the intermolecular forces

London Dispersion forces � The electrons of a molecule will shift to one side, creating a temporary dipole. This dipole will cause the other molecule to create a dipole as well and attraction will occur for a very short time. � This occurs between nonpolar molecules

Dipole-dipole forces � The slightly positive part of one molecule will attract the slightly negative part of another molecule. � This occurs between two polar molecules.
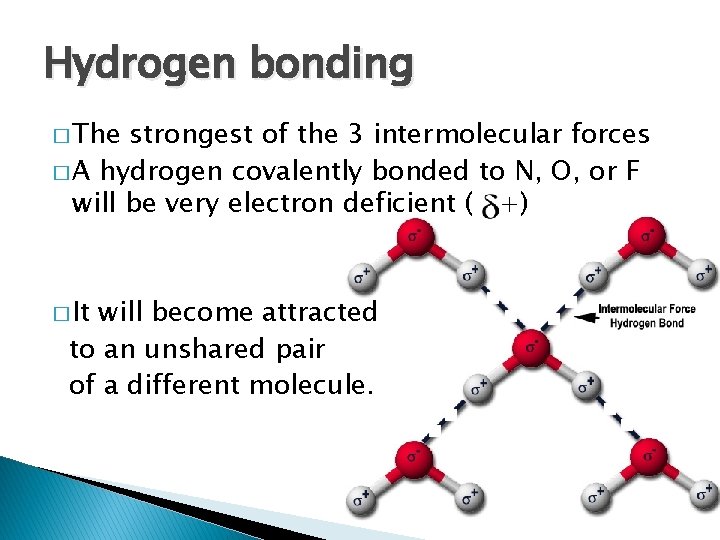
Hydrogen bonding � The strongest of the 3 intermolecular forces � A hydrogen covalently bonded to N, O, or F will be very electron deficient ( +) � It will become attracted to an unshared pair of a different molecule.

Why does this matter? � 1) Non-polar vs polar ◦ Polarity explains why water and oil do not mix! �Oil is non-polar, Water is polar ◦ Polarity explains why water dissolves ionic compounds!

Why does this matter? � 2) Intermolecular forces ◦ Explain why liquids and solids exist! ◦ Explains the melting and boiling points of molecular compounds. ◦ Hydrogen bonding explains why water has such high surface tension! ◦ Helps DNA and proteins take the shapes that they take!

HW: � 21) You find the difference in electronegativity between the atoms. � <0. 4 is nonpolar covalent � 0. 5 -1. 9 is polar covalent � >2 is ionic � 22) dispersion, dipole-dipole, hydrogen bond � 23)CCl 4 has polar bonds, but is not a polar molecule because of symmetry (it doesn’t have a (+) and (-) side. � 24)HOOH, � 26)shared Br. Cl, HBr, H 2 O bonding electrons, solubility ranges from high to low, exist as molecules

� � � Polar bond – a covalent bond where electrons are shared unequally ◦ The more electronegative atom is slightly negative ◦ The less electronegative atom is slightly positive Non-Polar bond –a covalent bond where electrons are shared equally ◦ Both atoms have similar or identical electronegativity Dispersion – weak forces caused by the random motion of electrons ◦ The only attractions non-polar molecules can have Dipole-dipole – attractions between polar molecules ◦ The slightly positive end of one molecule attracts the slightly negative end of another Hydrogen bond – an attraction where a hydrogen (that is bonded to an N, O, or F) is attracted to an unshared pair of electrons on another molecule. ◦ The strongest intermolecular attraction of the three
- Slides: 25