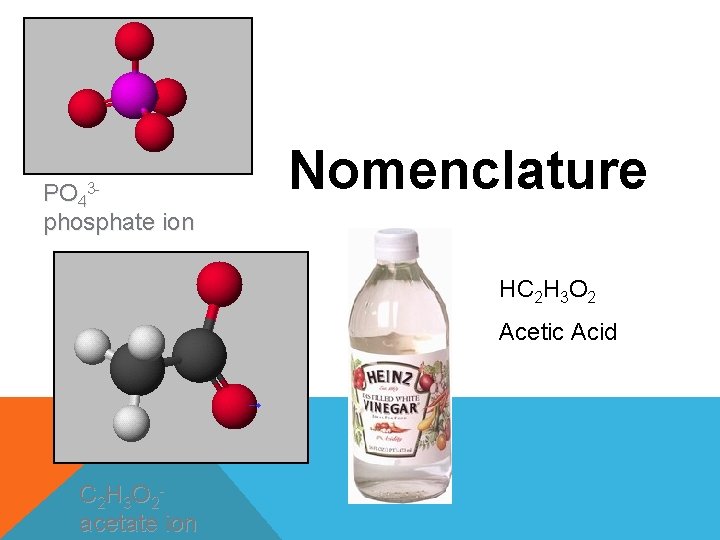
PO 43 phosphate ion Nomenclature HC 2 H





























- Slides: 45

PO 43 phosphate ion Nomenclature HC 2 H 3 O 2 Acetic Acid C 2 H 3 O 2 acetate ion

CHEMICAL BONDS There are 2 main forms of bonding atoms: Ionic—complete transfer of 1 or more electrons from one atom to another (one loses, the other gains) Covalent—some valence electrons shared between atoms

COMPOUNDS FORMED FROM IONS CATION + ANION ---> COMPOUND Na+ + Cl- --> Na. Cl A neutral compound requires equal number of + and - charges.

PROPERTIES OF IONIC COMPOUNDS FORMING NACL FROM NA AND CL 2 A metal atom can transfer an electron to a nonmetal. The resulting cation and anion are attracted to each other. (One is positive and one is negative)

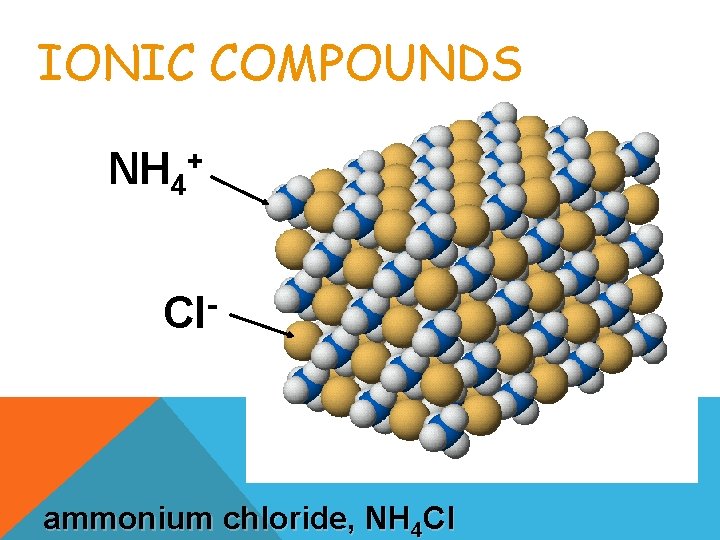
IONIC COMPOUNDS NH 4 + Cl ammonium chloride, NH 4 Cl

WRITING A FORMULA FOR AN IONIC COMPOUND Steps: 1. Write the formula for the cation and anion (Don’t forget to include the charge of each ion) 2. Decide how many cations and anions are needed so that the sum of their charges balance out to be zero 3. Write the formula of the compound by writing the number of cations followed by the number of anions which you used in step #2. (Do not write the charges for the ions since they are now balanced out to be

IN CLASS PRACTICE barium chloride

Iron (II) oxide

Calcium Phosphate

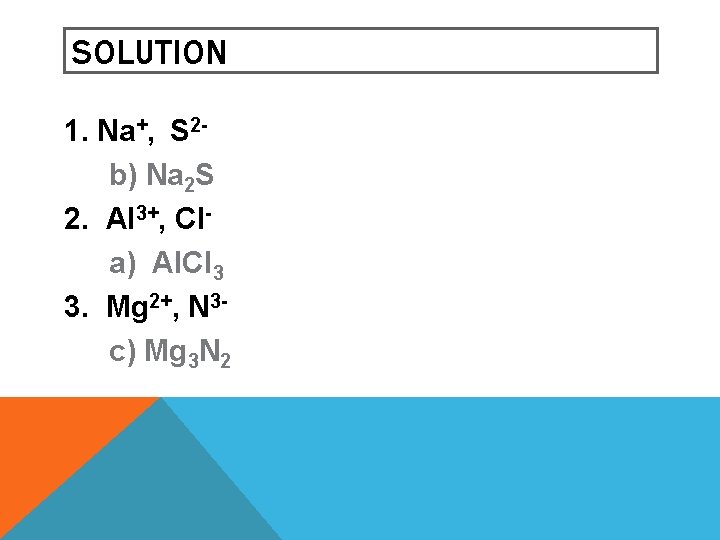
LEARNING CHECK Write the correct formula for the compounds containing the following ions: 1. sodium sulfide a) Na. S b) Na 2 S c) Na. S 2 2. aluminum chloride a) Al. Cl 3 b) Al. Cl c) Al 3 Cl 3. magnesium nitride a) Mg. N b) Mg 2 N 3 c) Mg 3 N 2

SOLUTION 1. Na+, S 2 b) Na 2 S 2. Al 3+, Cla) Al. Cl 3 3. Mg 2+, N 3 c) Mg 3 N 2

NAMING COMPOUNDS Steps: 1. Separate the compound into its positive and negative parts (positive part will be the first element, negative part will be the second element) 2. Write the name of the cation and then the name of the anion

MORE THAN ONE POSITIVE OXIDATION NUMBER Elements that can have more than one possible positive charge MUST have a Roman Numeral to indicate the charge on the individual ion. (Any element not included in group 1, group 2, silver, cadmium, zinc, and aluminum)

NAMES OF VARIABLE IONS These elements REQUIRE Roman Numerals because they can have more than one possible charge: anything except Group 1 A, 2 A, Ag, Zn, Cd, and Al (You should already know the charges on these!) Fe. Cl 3 (Fe 3+) Iron (III) chloride Cu. Cl Sn. F 4 Pb. Cl 2 Fe 2 S 3 (Cu+ ) (Sn 4+) (Pb 2+) (Fe 3+) copper (I) chloride tin (IV) fluoride lead (II) chloride iron (III) sulfide

EXAMPLES OF OLDER NAMES OF CATIONS FORMED FROM TRANSITION METALS (YOU DO NOT HAVE TO MEMORIZE THESE)

IN CLASS PRACTICE Zn. O

Sr. Cl 2

Ag 2 S

Ni. P

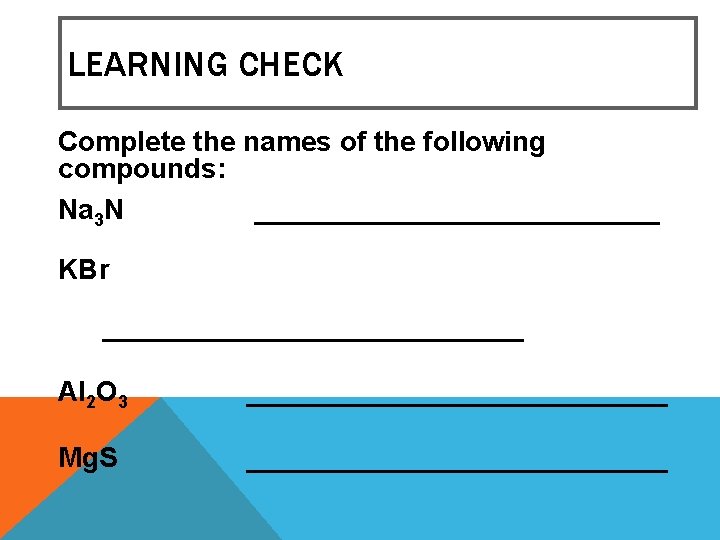
LEARNING CHECK Complete the names of the following compounds: Na 3 N _____________ KBr ______________ Al 2 O 3 ______________ Mg. S ______________

LEARNING CHECK Complete the names of the following compounds : Fe. Br 2 ____________ Cu. Cl ____________ KBr ____________ Fe 2 O 3 ____________ Fe(OH)3 ____________

MIXED PRACTICE! Name/Write the formula for the following: 1. 2. 3. 4. 5. 6. Na 2 O Barium Fluoride Pb. S 2 Calcium nitride Cu 3 PO 4 Hg. F 2 7. Copper (II) chlorate 8. Sn 3 N 2 9. Aluminum carbonate 10. Potassium bromide 11. Ca. CO 3 12. Cesium hydroxide

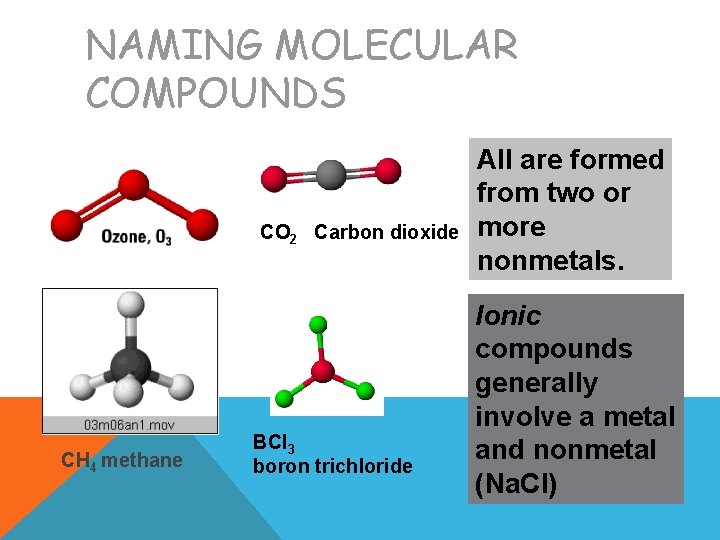
NAMING MOLECULAR COMPOUNDS CO 2 Carbon dioxide CH 4 methane BCl 3 boron trichloride All are formed from two or more nonmetals. Ionic compounds generally involve a metal and nonmetal (Na. Cl)

MOLECULAR (COVALENT) NOMENCLATURE FOR TWO NONMETALS Prefix System (binary compounds) 1. Less electronegative atom comes first. 2. Add prefixes to indicate # of atoms. Omit mono- prefix on the FIRST element. Mono- is OPTIONAL on the SECOND element (in this class, it’s NOT optional!). 3. Change the ending of the second element to -ide.

MOLECULAR NOMENCLATURE PREFIXES PREFIX monoditritetrapentahexaheptaoctanonadeca- NUMBER 1 2 3 4 5 6 7 8 9 10

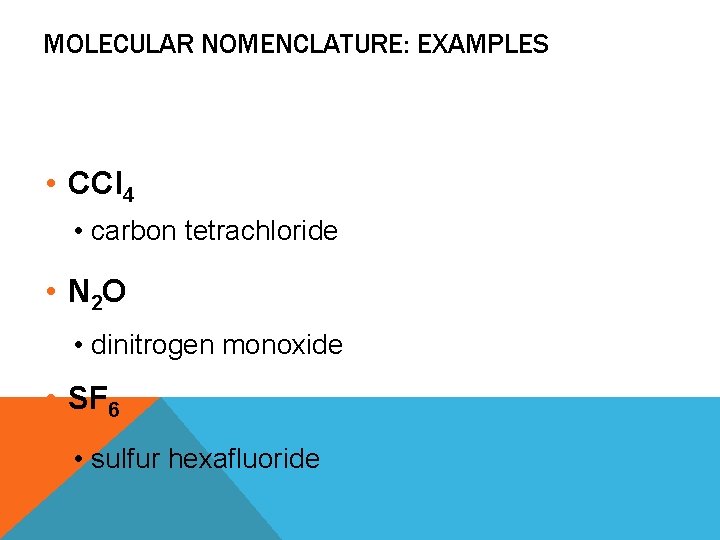
MOLECULAR NOMENCLATURE: EXAMPLES • CCl 4 • carbon tetrachloride • N 2 O • dinitrogen monoxide • SF 6 • sulfur hexafluoride

MORE MOLECULAR EXAMPLES • arsenic trichloride • As. Cl 3 • dinitrogen pentoxide • N 2 O 5 • tetraphosphorus decoxide • P 4 O 10

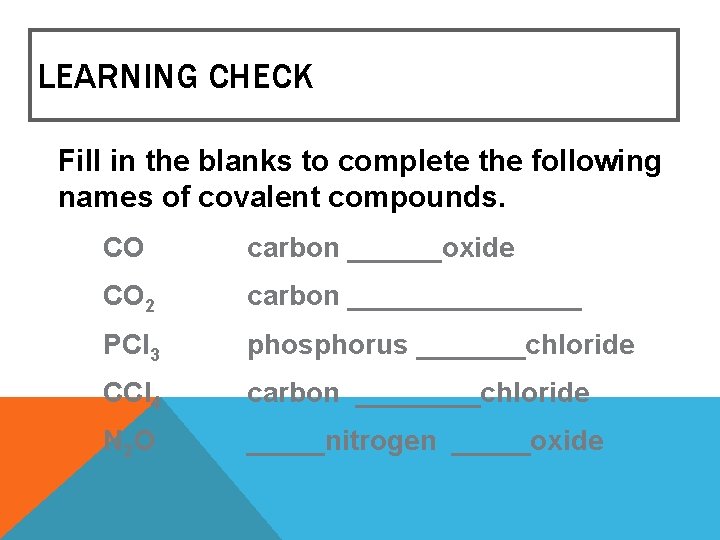
LEARNING CHECK Fill in the blanks to complete the following names of covalent compounds. CO carbon ______oxide CO 2 carbon ________ PCl 3 phosphorus _______chloride CCl 4 carbon ____chloride N 2 O _____nitrogen _____oxide

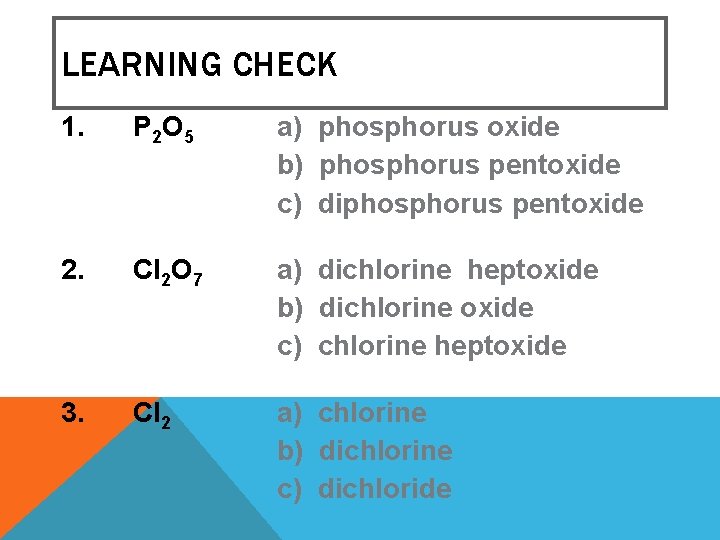
LEARNING CHECK 1. P 2 O 5 a) phosphorus oxide b) phosphorus pentoxide c) diphosphorus pentoxide 2. Cl 2 O 7 a) dichlorine heptoxide b) dichlorine oxide c) chlorine heptoxide 3. Cl 2 a) chlorine b) dichlorine c) dichloride

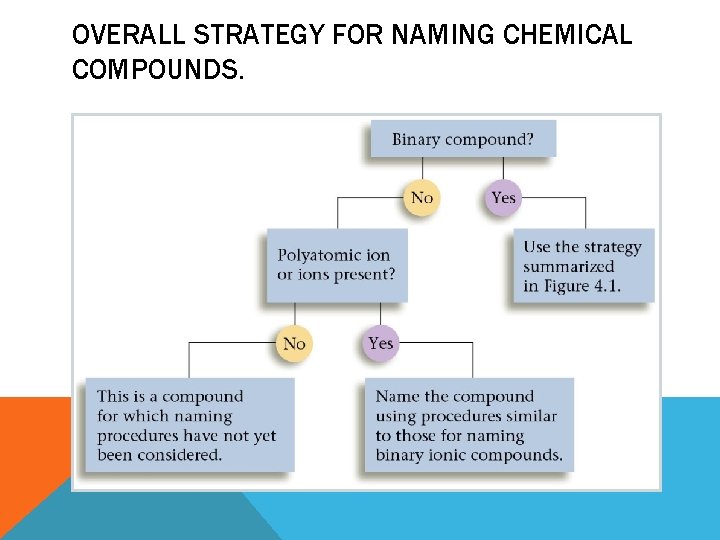
OVERALL STRATEGY FOR NAMING CHEMICAL COMPOUNDS.

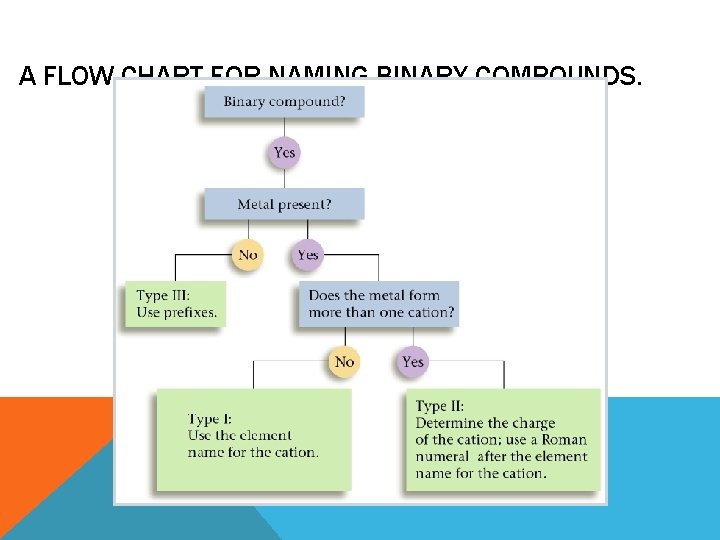
A FLOW CHART FOR NAMING BINARY COMPOUNDS.

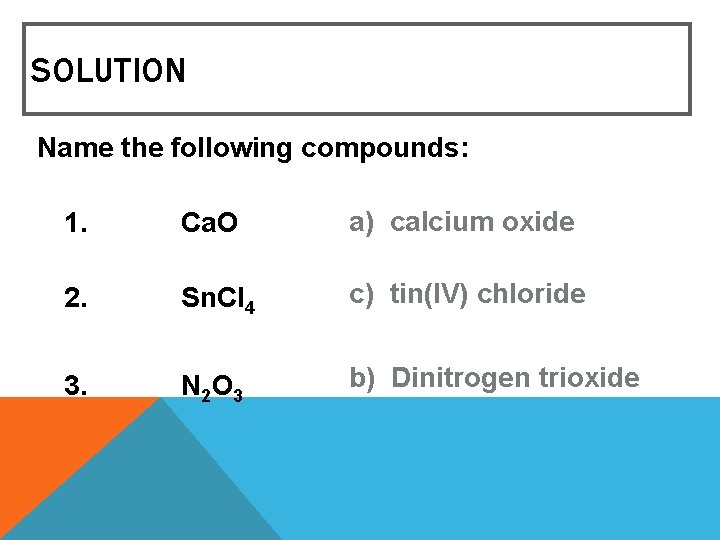
MIXED REVIEW Name the following compounds: 1. Ca. O a) calcium oxide c) calcium (II) oxide 2. 3. Sn. Cl 4 a) tin tetrachloride c) tin(IV) chloride N 2 O 3 a) nitrogen oxide c) nitrogen trioxide b) calcium(I) oxide b) tin(II) chloride b) dinitrogen trioxide

SOLUTION Name the following compounds: 1. Ca. O a) calcium oxide 2. Sn. Cl 4 c) tin(IV) chloride 3. N 2 O 3 b) Dinitrogen trioxide

MIXED PRACTICE 1. Dinitrogen monoxide 2. Potassium sulfide 3. Copper (II) nitrate 4. Dichlorine heptoxide 5. Chromium (III) sulfate 6. Iron (III) sulfite 7. Calcium oxide 8. Barium carbonate

MIXED PRACTICE 1. 2. 3. 4. 5. 6. 7. 8. 9. Ba. I 2 P 4 S 3 Ca(OH)2 Fe. CO 3 Na 2 Cr 2 O 7 I 2 O 5 Cu(Cl. O 4)2 CS 2 B 2 Cl 4

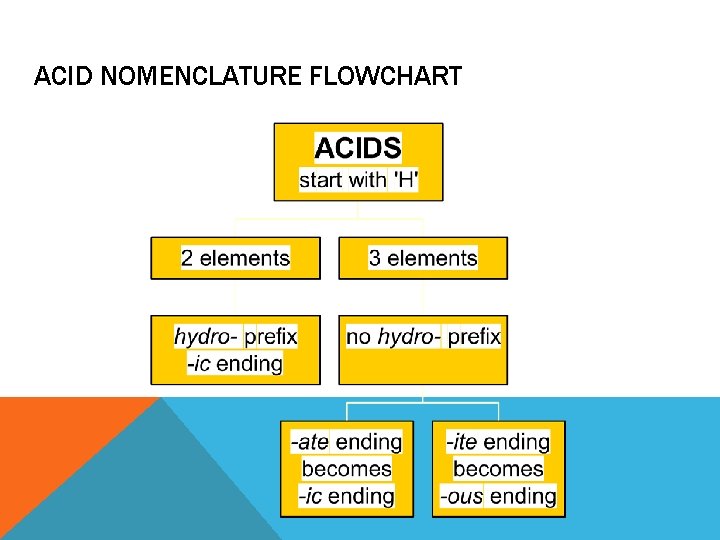
ACID NOMENCLATURE Acids § Compounds that form H+ in water. § Formulas usually begin with ‘H’. § In order to be an acid instead of a gas, binary acids must be aqueous (dissolved in water) § Ternary acids are ALL aqueous Examples: § HCl (aq) – hydrochloric acid § HNO 3 – nitric acid § H 2 SO 4 – sulfuric acid

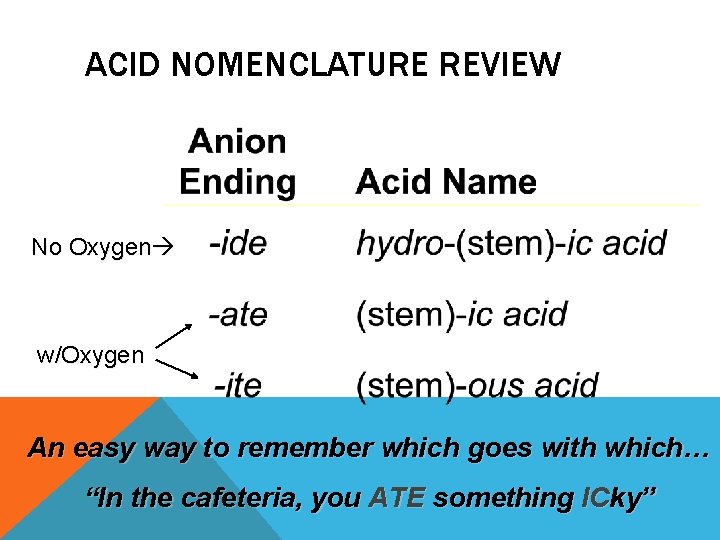
ACID NOMENCLATURE REVIEW No Oxygen w/Oxygen An easy way to remember which goes with which… “In the cafeteria, you ATE something ICky”

ACID NOMENCLATURE FLOWCHART

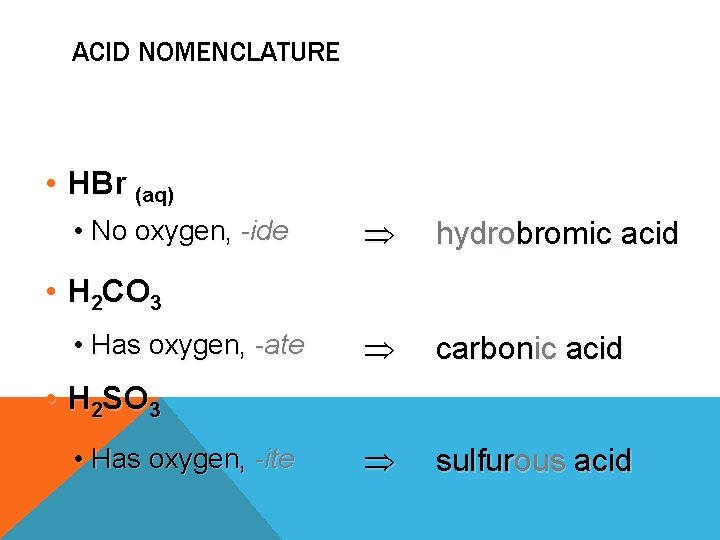
ACID NOMENCLATURE • HBr (aq) • No oxygen, -ide hydrobromic acid carbonic acid sulfurous acid • H 2 CO 3 • Has oxygen, -ate • H 2 SO 3 • Has oxygen, -ite

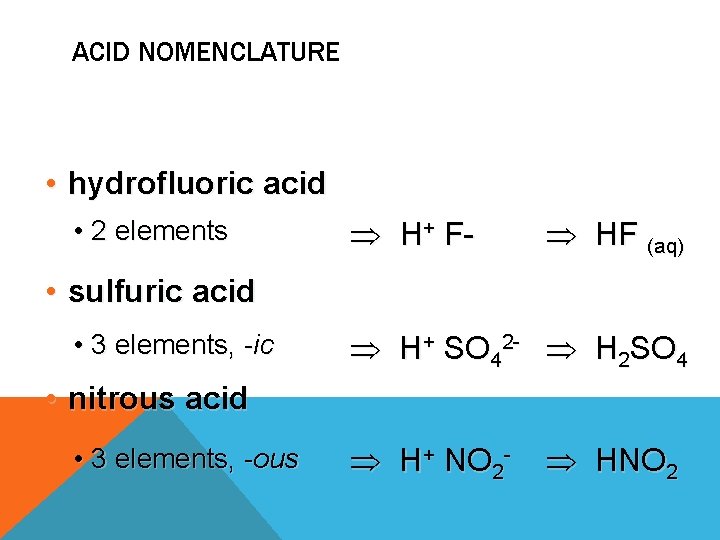
ACID NOMENCLATURE • hydrofluoric acid • 2 elements H+ F- HF (aq) • sulfuric acid • 3 elements, -ic H+ SO 42 - H 2 SO 4 • nitrous acid • 3 elements, -ous H+ NO 2 - HNO 2

NAME ‘EM! HI (aq) HCl H 2 SO 3 HNO 3 HIO 4

WRITE THE FORMULA! Hydrobromic acid Nitrous acid Carbonic acid Phosphoric acid Hydrotelluric acid

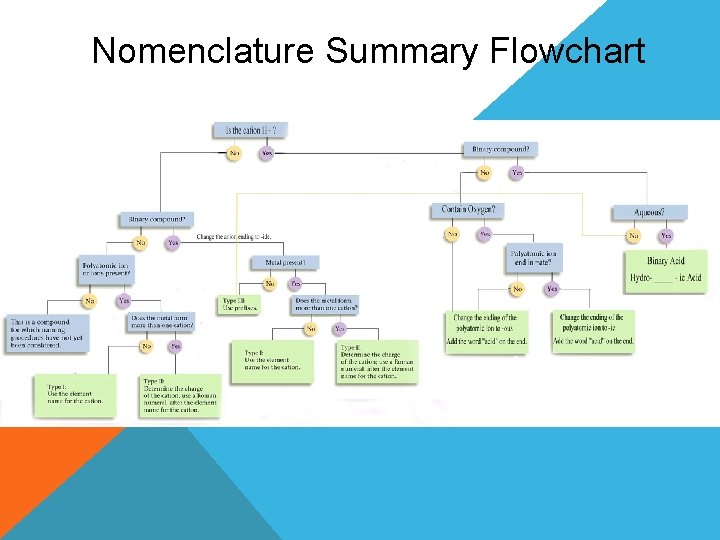
Nomenclature Summary Flowchart

DONE

RAINBOW MATRIX GAME Link on Chemistry Geek. com on Chemistry I page http: //chemistrygeek. com/rainbow Use [ ] to represent subscripts since you can’t enter subscripts into the computer So H 2 O would be H[2]O And Al 2(SO 4)3 would be Al[2](SO[4])[3] Additional Polyatomic Ions (you do not have to memorize these, but they are in the game!) Borate = BO 3 -3 ; Silicate = Si. O 4 -4 ; Manganate = Mn. O 4 -2 (permanganate is -1)