Plymouth Microelectrode Techniques Workshop Single Electrode Voltage Clamping


















- Slides: 33

Plymouth Microelectrode Techniques Workshop Single Electrode Voltage Clamping Alasdair Gibb Research Department of Neuroscience, Physiology & Pharmacology, University College London E-mail: a. gibb@ucl. ac. uk ciona intestinalis (sea squirt) eggs Latin name means, literally, "pillar of intestines",

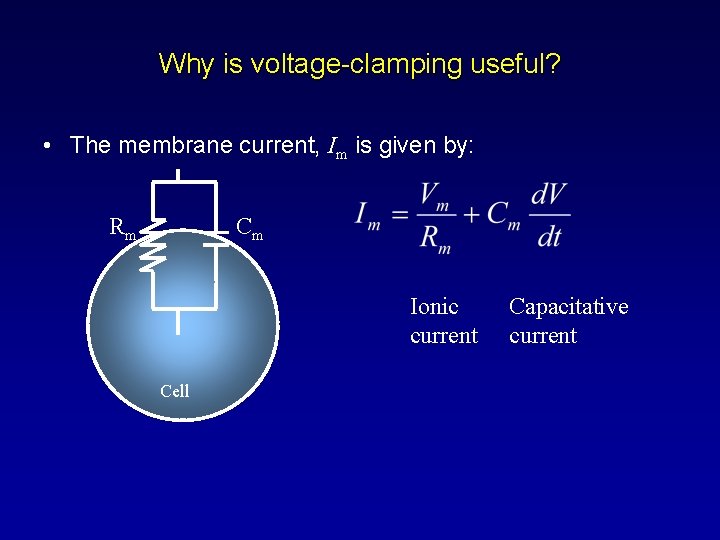
Why is voltage-clamping useful? • The membrane current, Im is given by: Rm Cm Ionic current Cell Capacitative current

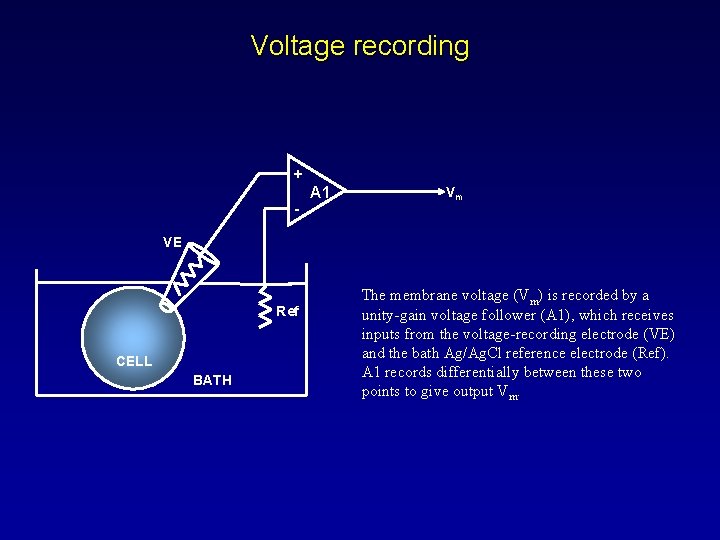
Voltage recording + - A 1 Vm VE Ref CELL BATH The membrane voltage (Vm) is recorded by a unity-gain voltage follower (A 1), which receives inputs from the voltage-recording electrode (VE) and the bath Ag/Ag. Cl reference electrode (Ref). A 1 records differentially between these two points to give output Vm.

Squid electrophysiology at the Marine Biological Association Laboratory, Plymouth, England. Common squid, Loligo

Single electrode voltage clamp Key references: Brennecke & Lindemann (1974). Theory of a membrane-voltage clamp with discontinuous feedback through a pulsed current clamp. Rev. Sci. Instrum. 45: 184 -188 Wilson & Goldner (1975). Voltage clamping with a single microelectrode. J. Neurobiology. 6: 411 -422. ** Finkel & Redman (1984). Theory and operation of a single microelectrode voltage clamp. J. Neurosci. Meth. 11: 101 -127. Axoclamp-2 B manual – available online at www. axon. com www. moleculardevices. com Alan Finkel (2007), Scholarpedia, 2(8): 3528.

Axoclamp 2 B Amplifier Headstage www. moleculardevices. com Dr Alan Finkel, Ph. D Electrical Engineering (1981), Chancellor of Monash University (2008)

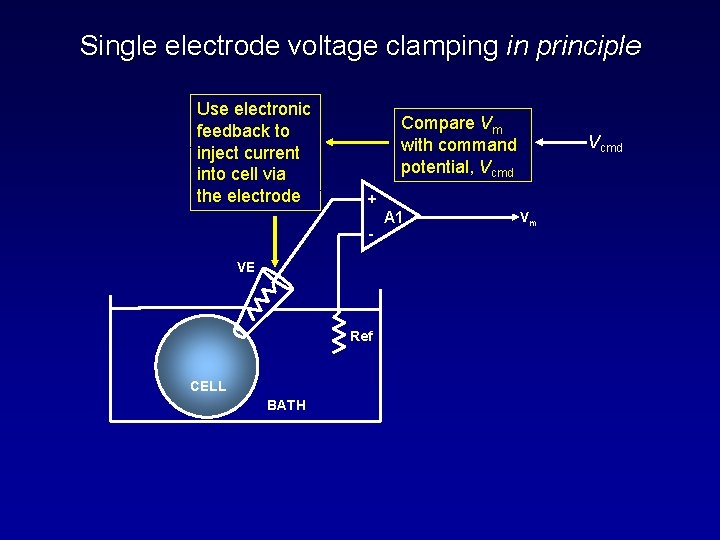
Single electrode voltage clamping in principle Use electronic feedback to inject current into cell via the electrode Compare Vm with command potential, Vcmd + - VE Ref CELL BATH Vcmd A 1 Vm

Just one big problem…… • Current passing down the microelectrode generates an error voltage due to the electrode resistance, according to Ohms Law: Verror = IRe current, I + - Re CELL A 1 Vm e. g. I = 10 n. A, Re = 10 M Verror= 10 -8 A x 107 = 10 -1 V = 100 m. V

Solution……. • Electronically switch between periods of current injection, and voltage recording…. . • A switched single electrode voltage clamp (SEVC)

SEVC – modes of operation (i) Bridge recording mode – record membrane potential (ii) Discontinuous Single Electrode Current Clamp - d. SECC (iii) Discontinuous Single Electrode Voltage Clamp - d. SEVC Bridge d. SECC d. SEVC

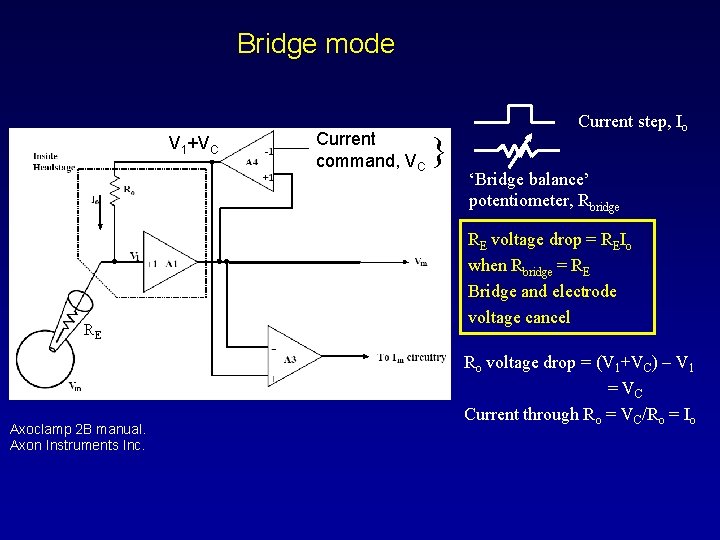
Bridge mode V 1+VC RE Axoclamp 2 B manual. Axon Instruments Inc. Current command, VC } Current step, Io ‘Bridge balance’ potentiometer, Rbridge RE voltage drop = REIo when Rbridge = RE Bridge and electrode voltage cancel Ro voltage drop = (V 1+VC) – V 1 = VC Current through Ro = VC/Ro = Io

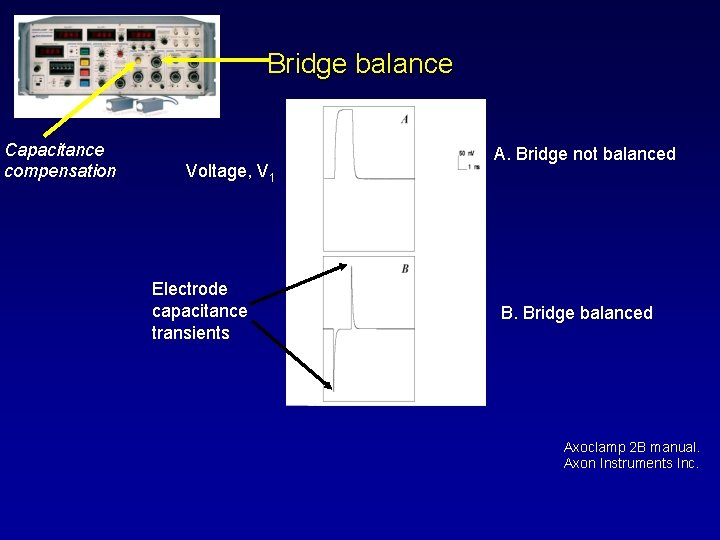
Bridge balance Capacitance compensation Voltage, V 1 Electrode capacitance transients A. Bridge not balanced B. Bridge balanced Axoclamp 2 B manual. Axon Instruments Inc.

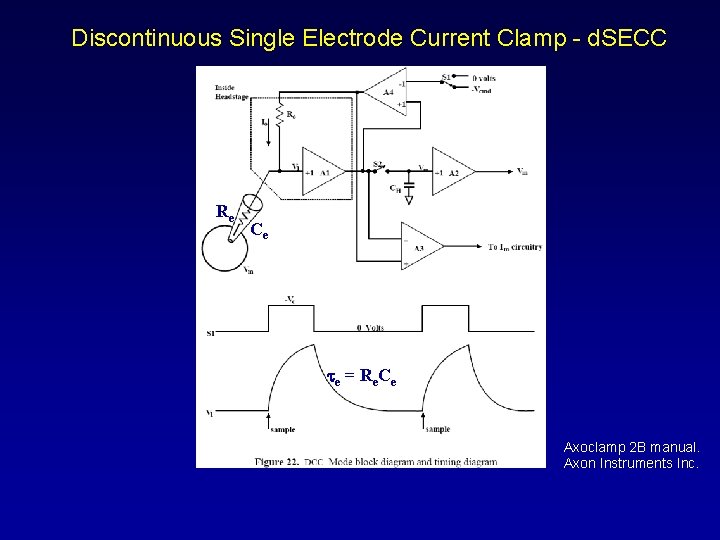
Discontinuous Single Electrode Current Clamp - d. SECC Re Ce te = Re. Ce Axoclamp 2 B manual. Axon Instruments Inc.

Discontinuous Single Electrode Voltage Clamp - d. SEVC Switching frequency fs = 1/T Axoclamp 2 B manual. Axon Instruments Inc.

Axoclamp 900 A Software Control www. moleculardevices. com

NPI Electronic www. npielectronic. com

SEVC - Advantages • Requires only a single electrode – some cells are easily damaged with two electrodes • Penetration can be done ‘blind’: useful in brain slices or in vivo CNS • Series resistance errors are avoided • Can clamp large cells (100 p. F) and large currents (~10 n. A)

SEVC - Disadvantages • Clamp has more noise, slower response and lower fidelity than TEVC or patch • Clamp has low gain/instability with small cells -: better to use patch clamp • Practical aspects of circuit design and construction, electrodes and recording setup are all more difficult than for TEVC or for patch clamp

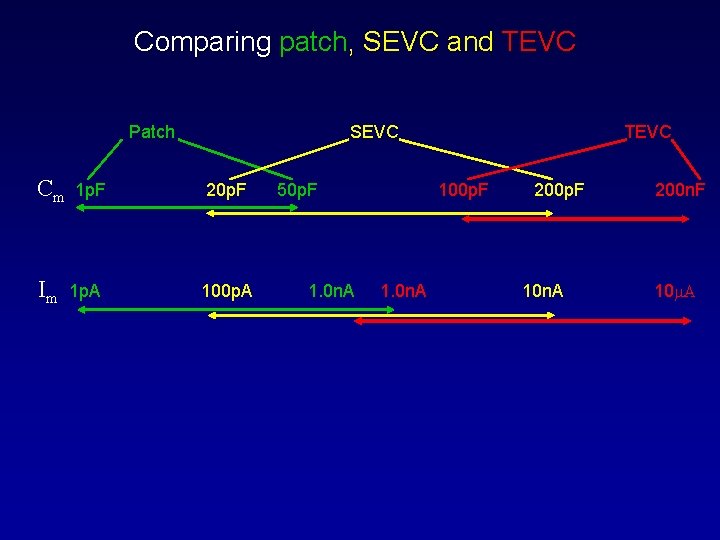
Comparing patch, SEVC and TEVC Patch Cm Im 1 p. F 1 p. A SEVC 20 p. F 100 p. A 50 p. F 1. 0 n. A TEVC 100 p. F 1. 0 n. A 200 p. F 10 n. A 200 n. F 10 m. A

Practical considerations 1. Electrodes: aim for lowest resistance and capacitance compatible with stable recordings Re Electrode time constant limits switching frequency, which limits maximum gain with clamp Ce t e = R e Ce

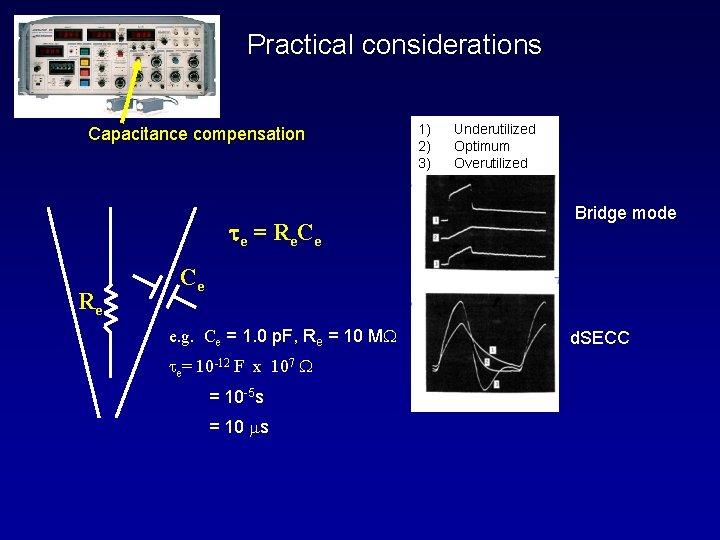
Practical considerations Capacitance compensation t e = R e Ce Re 1) 2) 3) Underutilized Optimum Overutilized Bridge mode Ce e. g. Ce = 1. 0 p. F, Re = 10 M te= 10 -12 F x 107 = 10 -5 s = 10 ms d. SECC

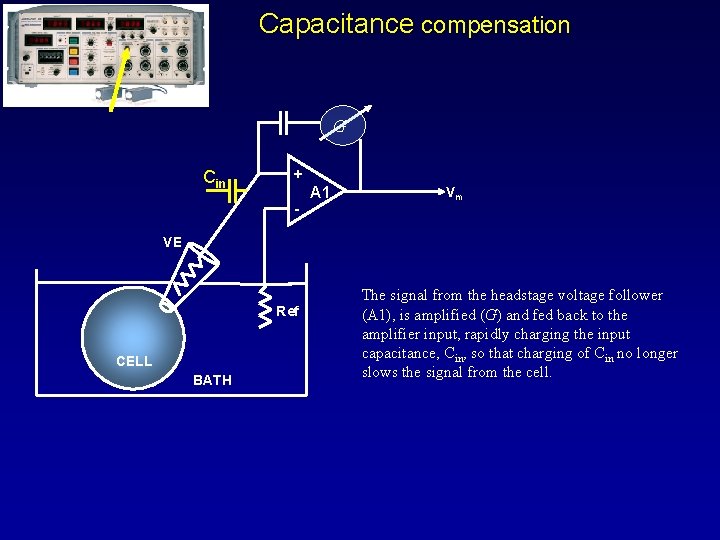
Capacitance compensation G Cin + - A 1 Vm VE Ref CELL BATH The signal from the headstage voltage follower (A 1), is amplified (G) and fed back to the amplifier input, rapidly charging the input capacitance, Cin, so that charging of Cin no longer slows the signal from the cell.

Practical considerations Clamp phase shift In a parallel RC circuit, at high frequencies Cm will introduce a 90 o phase shift between membrane current and membrane voltage. Real cell membranes often show less than 90 o phase shift so phase lead (achieved by boosting the high frequency gain of the clamp amplifier) could be used to compensate – however tends to cause instability. Phase lag is generally used – effect is to decrease the high frequency gain of the clamp, so improves stability. In some situations, phase lag may reduce clamp noise, but may also tend to cause ringing.

Correctly set up SEVC Im Vm Rm = 100 M , Cm = 33 p. F True Vm Re = 100 M fs = 7 k. Hz, GT = 1 n. A/m. V Headstage voltage

Incorrectly set up SEVC (‘false clamp’) Im Vm True Vm Headstage voltage Capacitance neutralisation under-utilised Phase shift: maximum lag, t = 2 ms

SEVC using a suction (patch) electrode Rm = 300 M , Cm = 33 p. F Re = 3 M fs = 50 k. Hz, GT = 0. 7 n. A/m. V Phase lag, t = 0. 2 ms

SEVC of action potential currents KCl pipette Cs. Cl pipette Rm = 500 M , Cm = 13 p. F Re = 3 M fs = 40 k. Hz, GT = 5 n. A/m. V Sah, Gibb & Gage (1987) J. Gen. Physiol.

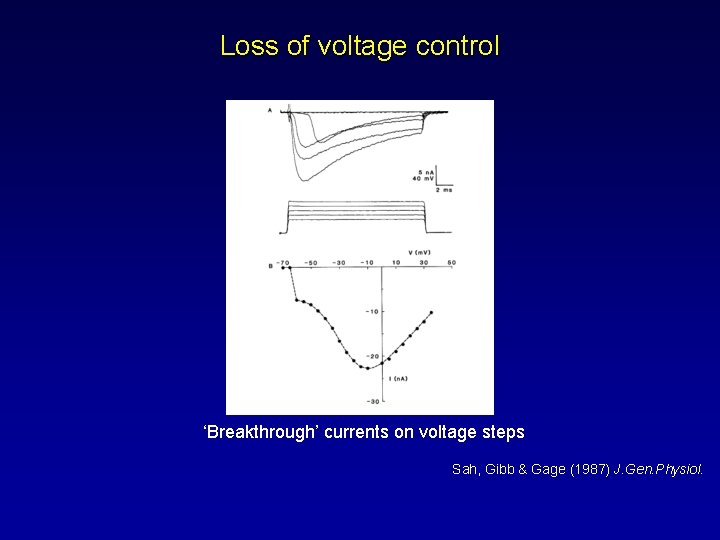
Loss of voltage control ‘Breakthrough’ currents on voltage steps Sah, Gibb & Gage (1987) J. Gen. Physiol.

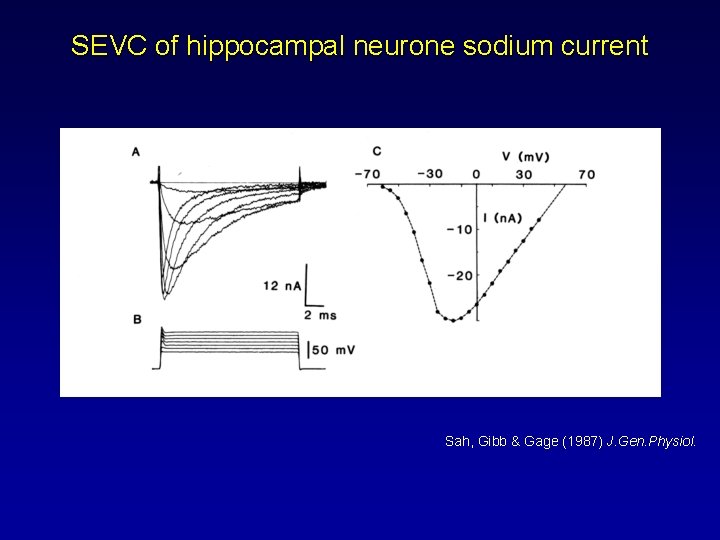
SEVC of hippocampal neurone sodium current Sah, Gibb & Gage (1987) J. Gen. Physiol.

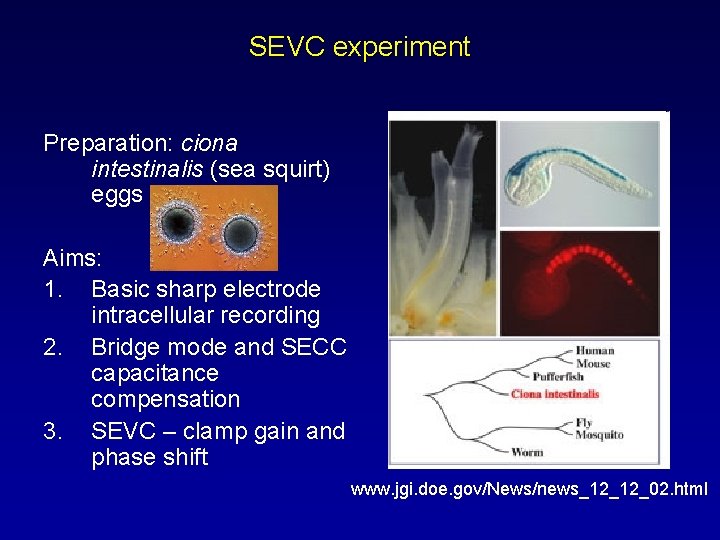
SEVC experiment Preparation: ciona intestinalis (sea squirt) eggs Aims: 1. Basic sharp electrode intracellular recording 2. Bridge mode and SECC capacitance compensation 3. SEVC – clamp gain and phase shift www. jgi. doe. gov/News/news_12_12_02. html

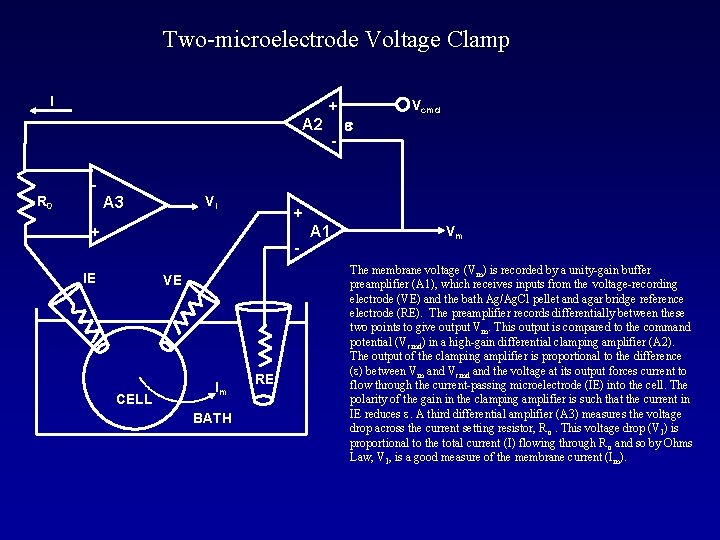
Two-microelectrode Voltage Clamp I + A 2 - e Vcmd R 0 VI A 3 + + - IE VE CELL Im BATH RE A 1 Vm The membrane voltage (Vm) is recorded by a unity-gain buffer preamplifier (A 1), which receives inputs from the voltage-recording electrode (VE) and the bath Ag/Ag. Cl pellet and agar bridge reference electrode (RE). The preamplifier records differentially between these two points to give output Vm. This output is compared to the command potential (Vcmd) in a high-gain differential clamping amplifier (A 2). The output of the clamping amplifier is proportional to the difference (e) between Vm and Vcmd and the voltage at its output forces current to flow through the current-passing microelectrode (IE) into the cell. The polarity of the gain in the clamping amplifier is such that the current in IE reduces e. A third differential amplifier (A 3) measures the voltage drop across the current setting resistor, R o. This voltage drop (VI) is proportional to the total current (I) flowing through R o and so by Ohms Law, VI, is a good measure of the membrane current (I m).

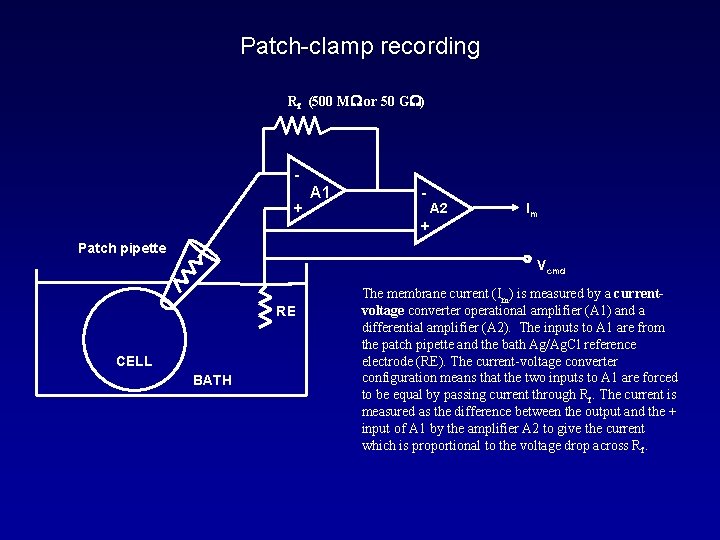
Patch-clamp recording Rf (500 MW or 50 GW) + A 1 - A 2 + Im Patch pipette Vcmd RE CELL BATH The membrane current (Im) is measured by a currentvoltage converter operational amplifier (A 1) and a differential amplifier (A 2). The inputs to A 1 are from the patch pipette and the bath Ag/Ag. Cl reference electrode (RE). The current-voltage converter configuration means that the two inputs to A 1 are forced to be equal by passing current through Rf. The current is measured as the difference between the output and the + input of A 1 by the amplifier A 2 to give the current which is proportional to the voltage drop across Rf.

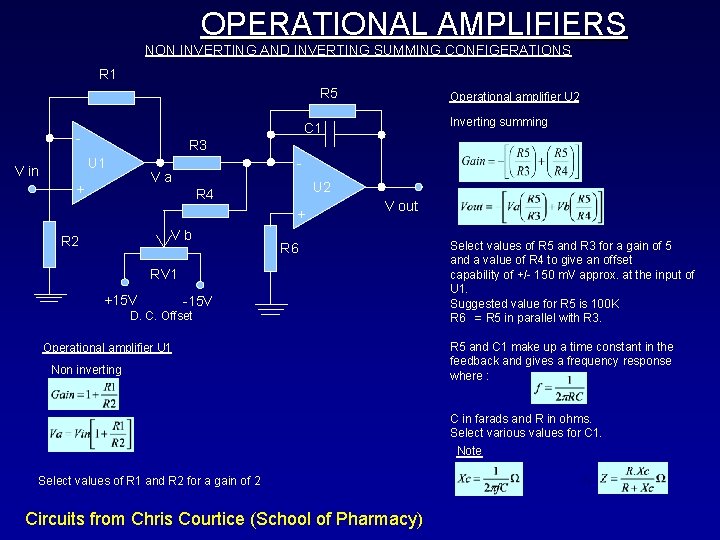
OPERATIONAL AMPLIFIERS NON INVERTING AND INVERTING SUMMING CONFIGERATIONS R 1 R 5 Inverting summing C 1 - R 3 U 1 V in Operational amplifier U 2 - Va + U 2 R 4 + Vb R 2 V out R 6 RV 1 +15 V -15 V D. C. Offset Operational amplifier U 1 Non inverting Select values of R 5 and R 3 for a gain of 5 and a value of R 4 to give an offset capability of +/- 150 m. V approx. at the input of U 1. Suggested value for R 5 is 100 K R 6 = R 5 in parallel with R 3. R 5 and C 1 make up a time constant in the feedback and gives a frequency response where : C in farads and R in ohms. Select various values for C 1. Note Select values of R 1 and R 2 for a gain of 2 Circuits from Chris Courtice (School of Pharmacy)