Platinum nanoparticlescobalt oxide nanostructures as efficient binary catalyst

Platinum nanoparticles-cobalt oxide nanostructures as efficient binary catalyst for ethylene glycol electro-oxidation Ghada H. El-Nowihy Chemical Engineering Department, Faculty of Engineering, The British University in Egypt Supervisors Prof. Mohamed S. El-Deab Chemistry Department, Faculty of Science, Cairo University Prof. Ahmad M. Mohammad Chemistry Department, Faculty of Science, Cairo University Prof. Mostafa M. H. Khalil Chemistry Department, Faculty of Science, Ain Shams University Prof. Mohamed A. El-Shahir Chemical Engineering Department, Faculty of Engineering, The British University in Egypt

Outline Ø Fuel Cells: Essence and Motivation Ø Direct Ethylene Glycol Fuel Cells (DEGFCs) Ø Limitations and Means of Overcoming ØExperimental Ø Results & Discussion Ø Conclusions
Energy Crisis & Alternative Energy Sources ü Solar energy ü Wind energy ü Hydroelectric energy ü Geothermal energy ü Bioenergy ü Fuel cells energy

Fuel cells: What and Why? Fuel cell: Chemoelectric engine that convert chemical energy of the fuel direct to electricity. Clean energy: Hydrogen + Oxygen H 2 O + Heat + electricity “Fuel cell vehicle” Gasoline + Oxygen CO 2 + H 2 O + Heat + electricity “air pollutant” “Gasoline vehicle”

Fuel cells: What and Why? ü High energy density (k. Wh/kg) : Energy produced per unit weight of the fuel ü High efficiency: No moving parts Combined heat and power (CHP) generation ü Unlimited runtime: In fuel cell, no charging time like batteries
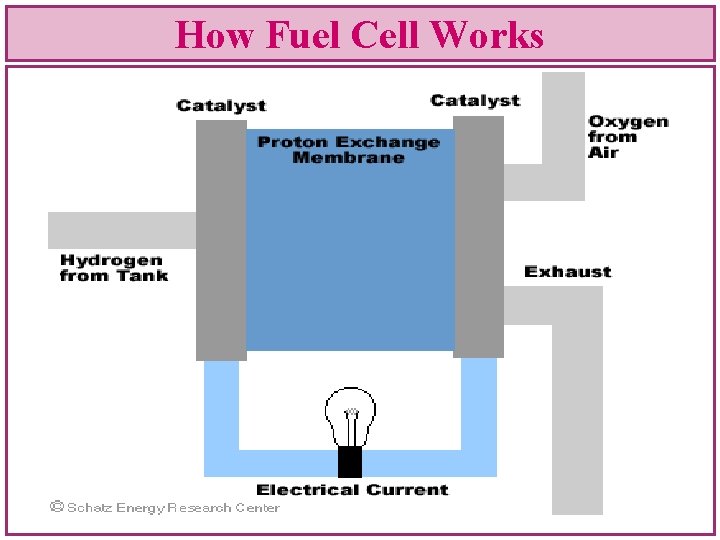
How Fuel Cell Works

Direct Ethylene Glycol Fuel Cell (DEGFC): Advantages Ø Ethylene glycol is liquid fuel much safer and easier to transport and handle than pressurized H 2 cylinders Ø Large Energy Density & less expensive hydrogen source HCOOH → CO 2 + 2 H+ + 2 e- “DFAFC provide 1. 4 k. W. h/kg” “DMFC provide 4. 2 k. W. h/kg” FA CH 3 OH + H 2 O → CO 2 + 6 H+ + 6 e- Me. OH CH 2 OH-CH 2 OH + 2 H 2 O → 2 CO 2 + 10 H+ + 10 e− EG “DEGFC provide 5. 3 k. W. h/kg”

Limitation: Poisoning of Pt catalyst Ø CO poison formation: Pt-COads main catalyst poison Ø CO 2 evolution: Ptfree Surface Modifier “MOx NPs” CH 2 OH-CH 2 OH + 2 H 2 O EG Fig. (A) 2 CO 2 + 10 H+ + 10 e− Fig. (B)

How Nanoparticles solve the problem of catalyst poisoning (1) Bifunctinal effect: Provide Oxygen containing species to adsorbed CO generating CO 2 (2) Third body effect “ensemble effect”: Change the geometry required for the adsorption of CO poison on the Pt substrate “i. e. ; prevent Pt atoms contiguity”. (3) Electronic effect: Change electronic structure of Pt to weaken the binding energy between Pt &CO.

Experimental Ø A. Electrodes and pretreatments § Chemicals & solutions § Electrochemical measurements Potentiostat Electrochemical cell § Electrodes Working electrode: GC Reference electrode: Ag/Ag. Cl/KCl(sat. ) Counter electrode: spiral Pt wire Ø B. Electrode modification nano-Pt & nano-MOx Ø C. Materials Characterization Electrode morphology & surface composition FE-SEM & EDS

Characterization of electrodes: Morphological FE-SEM Pt/GC electrode. FE-SEM of Ni. Ox/Pt/GC electrode. FE-SEM of Mn. Ox/Pt/GC electrode. FE-SEM of Co. Ox/Pt/GC electrode.

Characterization of electrodes: Compositional EDS of Pt/GC electrode. EDS of Ni. Ox/Pt/GC electrode. EDS of Mn. Ox/Pt/GC electrode. EDS of Co. Ox/Pt/GC electrode.

Characterization of electrodes: Electrochemical CV of Pt/GC electrode. CV of Ni. Ox/Pt/GC electrode. Ni(OH)2 ↔ Ni. OOH + H+ + e-

Co. Ox/Pt electrode: Characterization CV of Mn. Ox/Pt/GC electrode. 2 Mn. OOH + 2 OH− ↔ 2 Mn. O 2 + 2 H 2 O + 2 e− CV of Co. Ox/Pt/GC electrode. 3 Co (OH)2 + 2 OH- → Co 3 O 4+ 4 H 2 O+ 2 e− Co(II)&Co(IV) Co 3 O 4 + 2 OH- + H 2 O → 3 Co. OOH + e− Co(II)&Co(IV) Co(III) Co. OOH + OH- → Co. O 2 + H 2 O + e− Co(III) Co(IV)

Electrocatalytic activity of ethylene glycol (EG) oxidation at various electrodes Co. Ox/Pt/GC Highest enhancement at Co. Ox/Pt/GC Ip is 2 times of that obtained at Pt/GC Mn. Ox/Pt/GC Ni. Ox/Pt/GC LSVs for EGO at a) Pt/GC, b) Ni. Ox/Pt/GC, c) Mn. Ox/Pt/GC and d) Co. Ox/Pt/GC electrodes in 0. 5 M Na. OH solutions containing 0. 5 M EG. Potential scan rate is 50 m. V s− 1.

Stability of Co. Ox/Pt/GC electrode Ni. Ox/Pt Co. Ox/Pt/GC Pt Pt/GC I-t curve for 3 h of continuous electrolysis. Highest activity Highest stability

Origin of catalysis Ø Ni. Ox Ni(OH)2 ↔ Ni. OOH + H+ + e. Ni(II) Ni(III) Ø Mn. Ox 2 Mn. OOH + 2 OH− ↔ 2 Mn. O 2 + 2 H 2 O + 2 e− Mn(III) Mn(IV) Ø Co. Ox 2 pathways Co(OH)2 + OH- ↔ Co. OOH + H 2 O + e. Co(II) Co(III) Co. OOH + OH- ↔ Co. O 2 + H 2 O + e. Co(III) Co(IV) Co. OOH + EG Co. O 2 + EG → intermediates + Co(OH)2 Co. OOH + intermediates → products + Co(OH)2 → intermediates + Co. OOH Co. O 2 + intermediates → products + Co. OOH

Conclusions 1) nano-Ni. Ox, nano-Mn. Ox and nano-Co. Ox act as catalytic mediators facilitate charge transfer of the EGO. better konetis for EGO. higher energy obtained from the DEGFC. 2) Co. Ox/Pt/GC electrode highest catalytic activity towards EGO when compared to Pt/GC, Ni. Ox/Pt/GC and Mn. Ox/Pt/GC electrodes. 3) Co. Ox/Pt/GC electrode high stability; stable oxidation current over prolonged time of oxidation. Role of “nano-Co. Ox”


Origin of catalysis 2 Mn. OOH + 2 OH− ↔ 2 Mn. O 2 + 2 H 2 O + 2 e− Mn(III) Mn(IV) 2 Mn. O 2 + Pt−COads + H 2 O → 2 Mn. OOH + Ptfree + CO 2 Mn(IV) (main poison) Mn(III) Pt−COads + 2 OH − → Ptfree + CO 2 + H 2 O + 2 e−

Origin of catalysis Ni(OH)2 ↔ Ni. OOH + H+ + e. Ni(II) Ni(III) + EG → intermediates + Ni(II) Ni(III) + intermediates → products + Ni(II) Co(OH)2 + OH- ↔ Co. OOH + H 2 O + e. Co(II) Co(III) Co. OOH + EG → intermediates + Co(OH)2 Co. OOH + intermediates → products + Co(OH)2 Co. OOH + OH- ↔ Co. O 2 + H 2 O + e. Co(III) Co(IV) Co. O 2 + EG → intermediates + Co. OOH Co. O 2 + intermediates → products + Co. OOH
- Slides: 21