Platform Technologies for New Drug DiscoveryDevelopment Small Molecules
















- Slides: 39


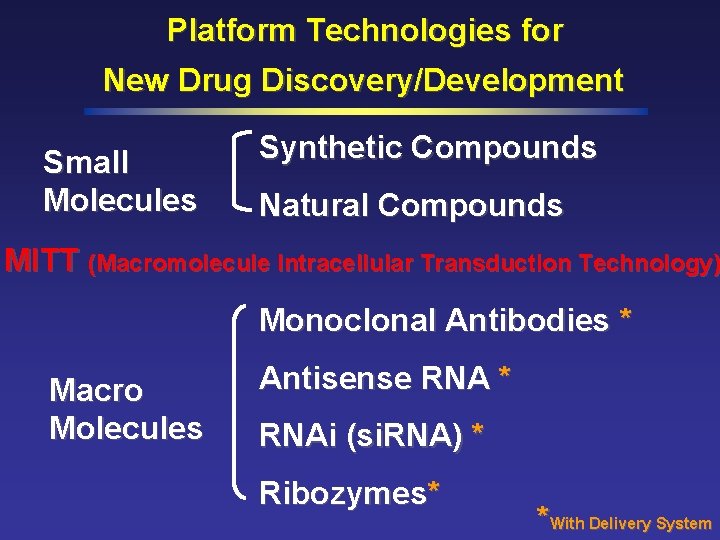
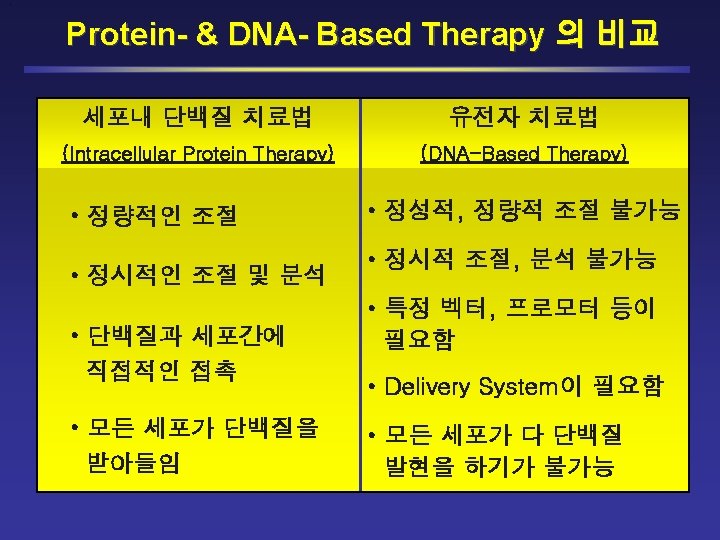
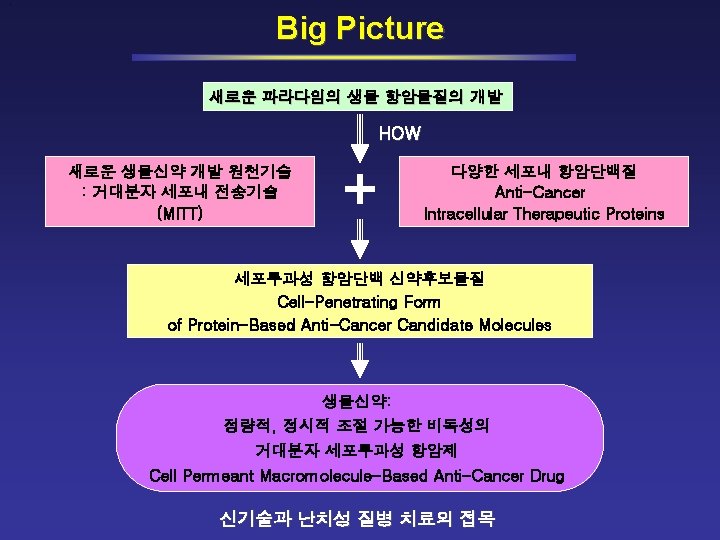
Platform Technologies for New Drug Discovery/Development Small Molecules Synthetic Compounds Natural Compounds MITT (Macromolecule Intracellular Transduction Technology) Monoclonal Antibodies * Macro Molecules Antisense RNA * RNAi (si. RNA) * Ribozymes* *With Delivery System

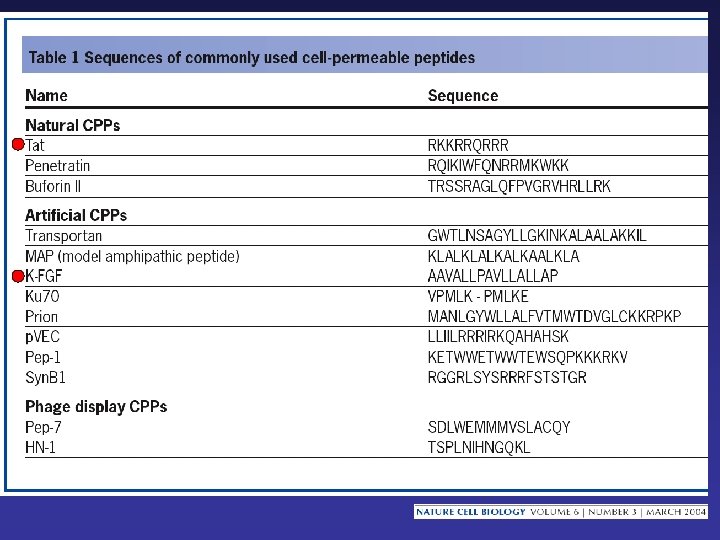
Macromolecules and Particles Transferred into Cells by MTD Peptides Proteins DNA RNA Drugs Imaging and Therapeutic Isotopes Phage Liposomes Beads


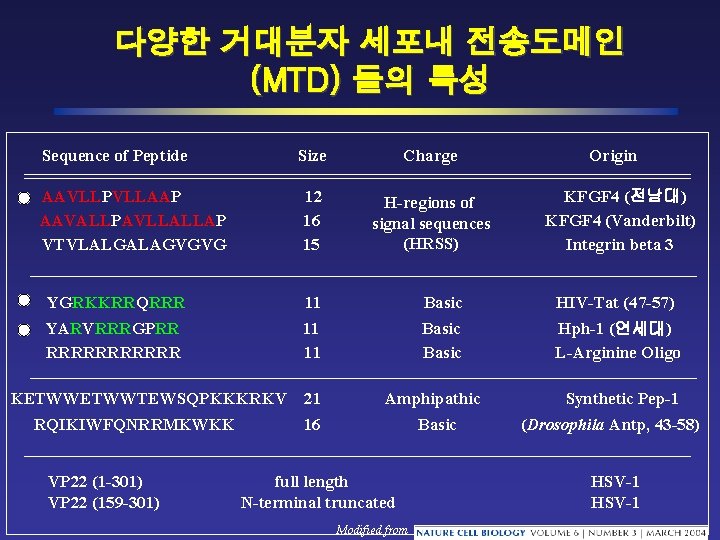
다양한 거대분자 세포내 전송도메인 (MTD) 들의 특성 Sequence of Peptide Size Charge AAVLLPVLLAAP AAVALLPAVLLALLAP VTVLALGALAGVGVG 12 16 15 H-regions of signal sequences (HRSS) Origin KFGF 4 (전남대) KFGF 4 (Vanderbilt) Integrin beta 3 YGRKKRRQRRR 11 Basic HIV-Tat (47 -57) YARVRRRGPRR RRRRRR 11 11 Basic Hph-1 (연세대) L-Arginine Oligo KETWWTEWSQPKKKRKV RQIKIWFQNRRMKWKK VP 22 (1 -301) VP 22 (159 -301) 21 Amphipathic 16 Basic full length N-terminal truncated Modified from Synthetic Pep-1 (Drosophila Antp, 43 -58) HSV-1

The First Report …. . This results open new possibilities for direct delivery of proteins into patients in the context of protein therapy…. 그러나, 이후 안된다는 보고가 잇따랐다.

The Second, But Real First Report …. . This is the first use of Protein Transduction to induce the enzymatic conversion of a substrate in living cells and animals….

Setback of Basic PTD HIV TAT PTD ? ? HIV Tat Basic PTD가 in vivo 에서 안된다는 많은 보고 중 하나

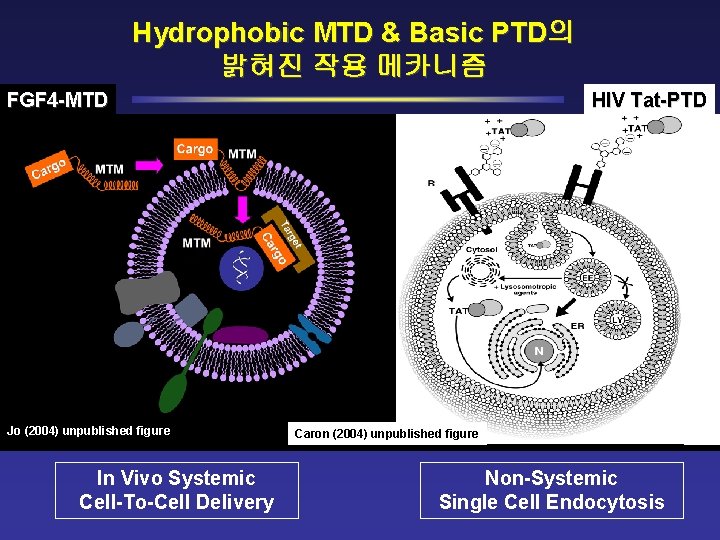
Hydrophobic MTD & Basic PTD의 밝혀진 작용 메카니즘 FGF 4 -MTD Jo (2004) unpublished figure In Vivo Systemic Cell-To-Cell Delivery HIV Tat-PTD Caron (2004) unpublished figure Non-Systemic Single Cell Endocytosis

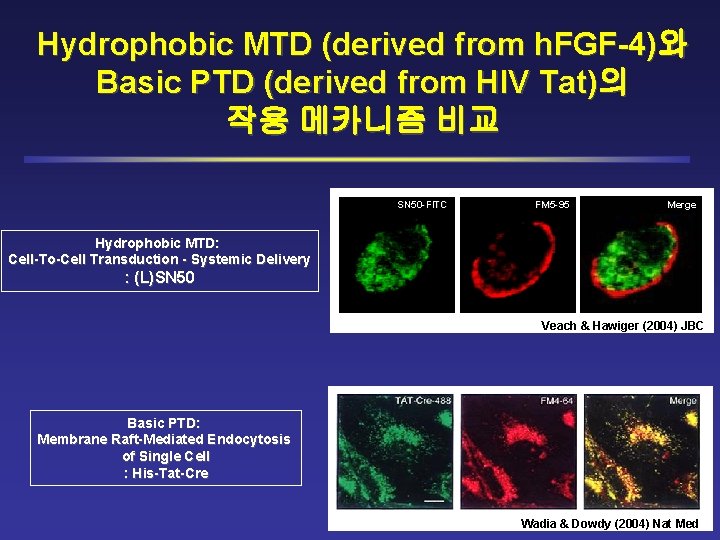
Hydrophobic MTD (derived from h. FGF-4)와 Basic PTD (derived from HIV Tat)의 작용 메카니즘 비교 SN 50 -FITC FM 5 -95 Merge Hydrophobic MTD: Cell-To-Cell Transduction - Systemic Delivery : (L)SN 50 Veach & Hawiger (2004) JBC Basic PTD: Membrane Raft-Mediated Endocytosis of Single Cell : His-Tat-Cre Wadia & Dowdy (2004) Nat Med

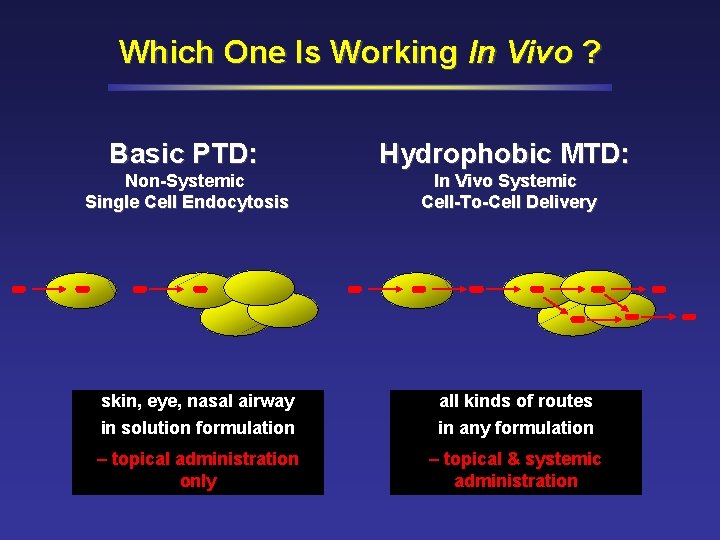
Which One Is Working In Vivo ? Basic PTD: Hydrophobic MTD: Non-Systemic Single Cell Endocytosis In Vivo Systemic Cell-To-Cell Delivery skin, eye, nasal airway in solution formulation all kinds of routes in any formulation – topical administration only – topical & systemic administration

MITT에 대한 국제 과학계의 시각 Technology Representing Figure: Jo et al (2001) Nat Biotech 19: 929



거대분자 세포내 전송기술 (MITT)의 응용 Block intracellular protein trafficking Interfere with protein-protein interactions Signal transduction, Transcription, Cell cycle control Map biologically important protein surfaces Analyze post-translational modifications Deliver functional enzymes and substrates into cells

Proof-Of-Concept I: Intracellular Protein Therapy In Inflammation

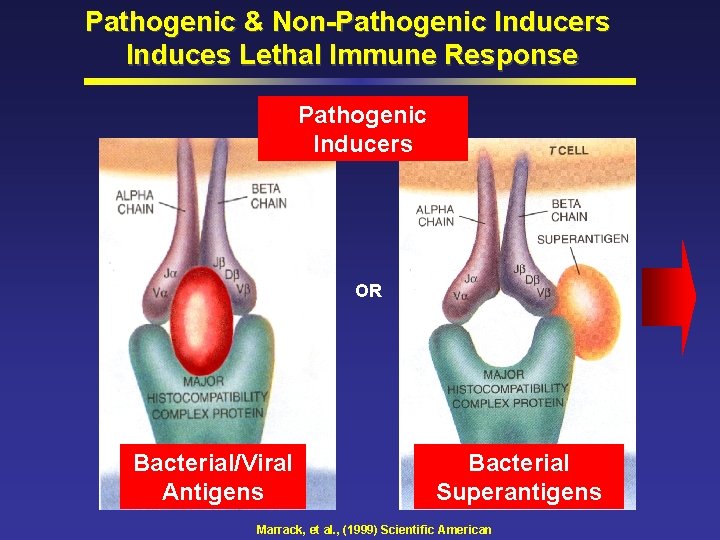
Pathogenic & Non-Pathogenic Inducers Induces Lethal Immune Response Pathogenic Inducers OR Bacterial/Viral Antigens Bacterial Superantigens Marrack, et al. , (1999) Scientific American

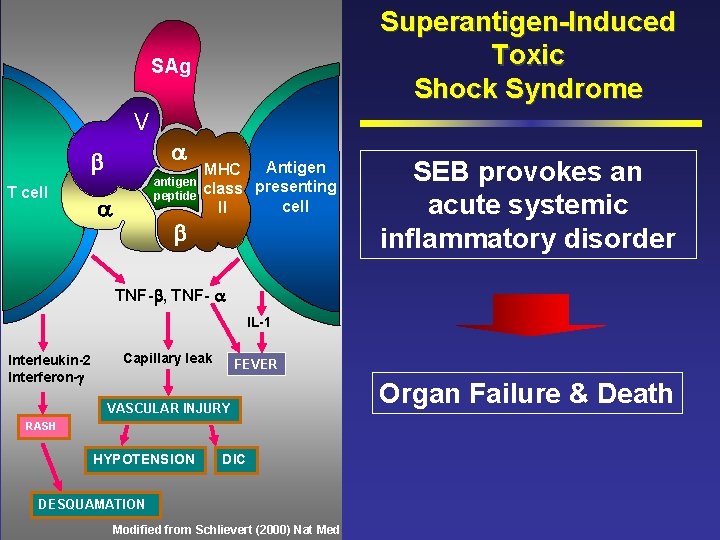
Superantigen-Induced Toxic Shock Syndrome SAg V T cell antigen peptide Antigen MHC class presenting cell II SEB provokes an acute systemic inflammatory disorder TNF- , TNF- IL-1 Interleukin-2 Interferon- Capillary leak FEVER VASCULAR INJURY RASH HYPOTENSION DIC DESQUAMATION Modified from Schlievert (2000) Nat Med Organ Failure & Death

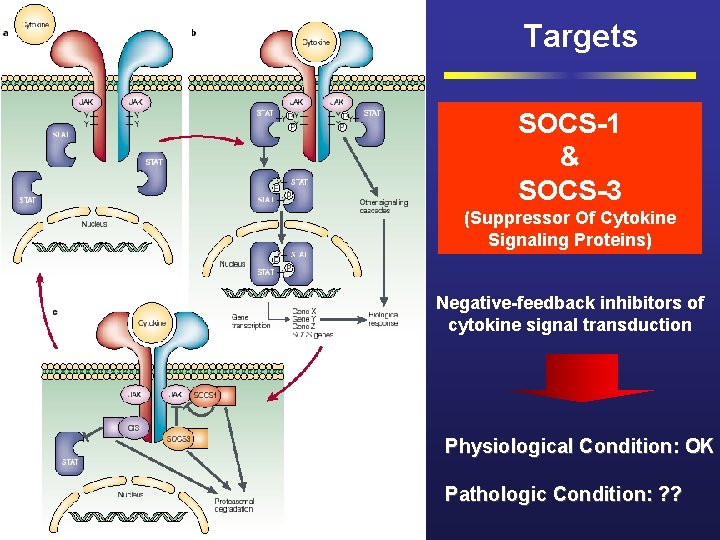
Targets SOCS-1 & SOCS-3 (Suppressor Of Cytokine Signaling Proteins) Negative-feedback inhibitors of cytokine signal transduction Physiological Condition: OK Pathologic Condition: ? ?

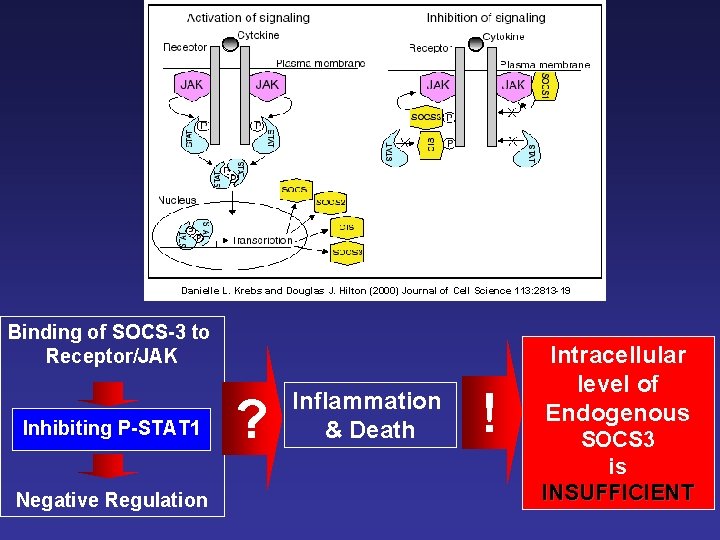
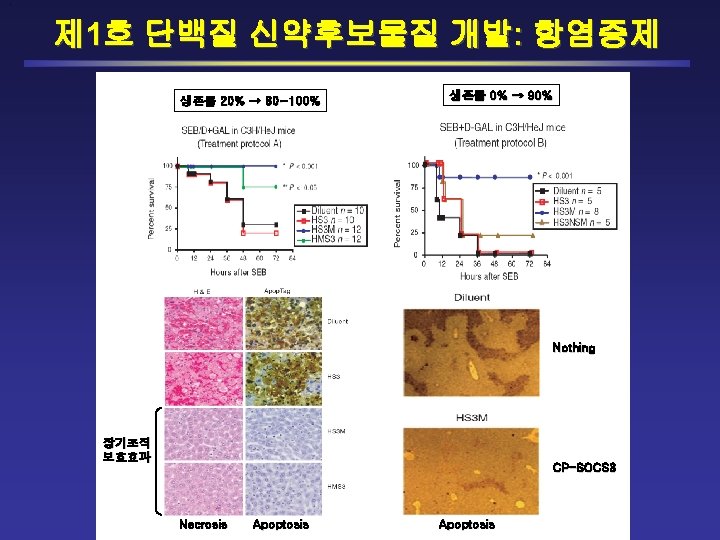
Danielle L. Krebs and Douglas J. Hilton (2000) Journal of Cell Science 113: 2813 -19 Binding of SOCS-3 to Receptor/JAK Inhibiting P-STAT 1 Negative Regulation ? Inflammation & Death ! Intracellular level of Endogenous SOCS 3 is INSUFFICIENT

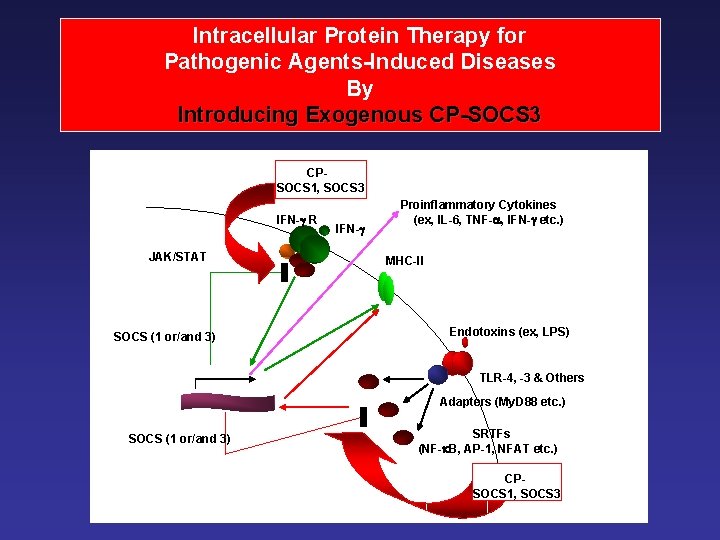
Intracellular Protein Therapy for Pathogenic Agents-Induced Diseases By Introducing Exogenous CP-SOCS 3 CPSOCS 1, SOCS 3 IFN- R JAK/STAT SOCS (1 or/and 3) IFN- Proinflammatory Cytokines (ex, IL-6, TNF- , IFN- etc. ) MHC-II Endotoxins (ex, LPS) TLR-4, -3 & Others Adapters (My. D 88 etc. ) SOCS (1 or/and 3) SRTFs (NF- B, AP-1, NFAT etc. ) CPSOCS 1, SOCS 3

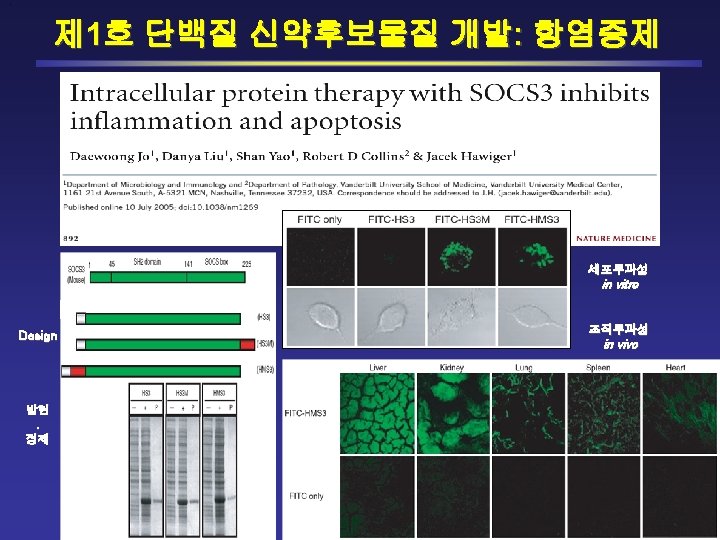
Structure & Design & Purification of CP-SOCS 3 1 45 SH 2 domain 141 SOCS-box 225 SOCS 3 Full-Length CP-SOCS 3 His-SOCS 3 (HS 3) His-SOCS 3 -MTM (HS 3 M) His-MTM-SOCS 3 (HMS 3) Truncated CP-SOCS 3 HS 3 NM NT (KIR+ESS, 1 -44) HMS 3 N HS 3 SM SH 2 domain (45 -141) HMS 3 S HS 3 BM SOCS box (142 -225) HMS 3 B HS 3 NSM NT + SH 2 (1 -141) HMNSS 3 HS 3 SBM HMS 3 SB SH 2 + SOCS box (141 -225)



Development of CP-SOCS 3 As An Anti-Inflammatory Agent In Clinic

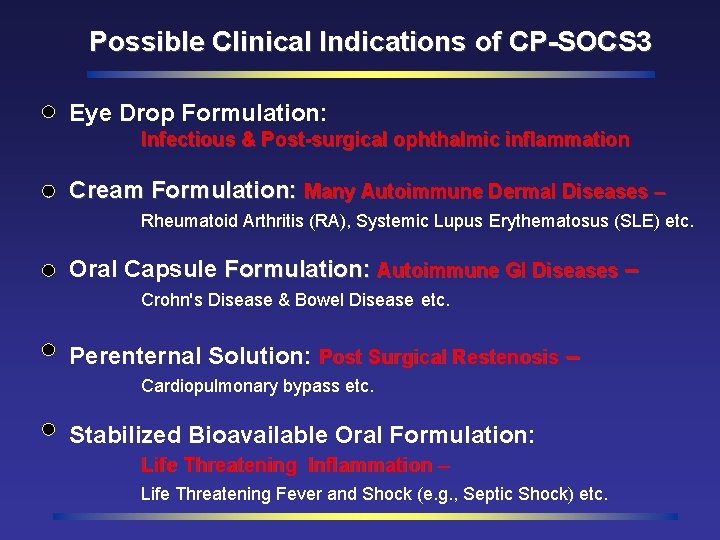
Possible Clinical Indications of CP-SOCS 3 Eye Drop Formulation: Infectious & Post-surgical ophthalmic inflammation Cream Formulation: Many Autoimmune Dermal Diseases – Rheumatoid Arthritis (RA), Systemic Lupus Erythematosus (SLE) etc. Oral Capsule Formulation: Autoimmune GI Diseases – Crohn's Disease & Bowel Disease etc. Perenternal Solution: Post Surgical Restenosis – Cardiopulmonary bypass etc. Stabilized Bioavailable Oral Formulation: Life Threatening Inflammation – Life Threatening Fever and Shock (e. g. , Septic Shock) etc.

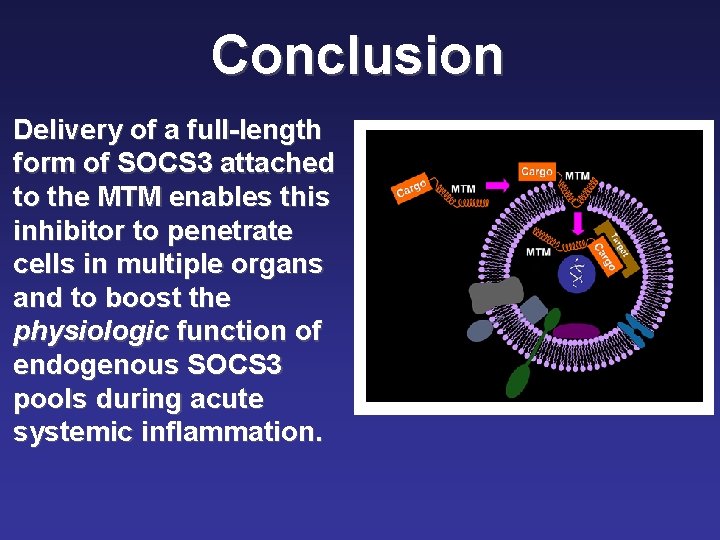
Conclusion Delivery of a full-length form of SOCS 3 attached to the MTM enables this inhibitor to penetrate cells in multiple organs and to boost the physiologic function of endogenous SOCS 3 pools during acute systemic inflammation.

Proof-Of-Concept II: Intracellular Protein Therapy in Cancer


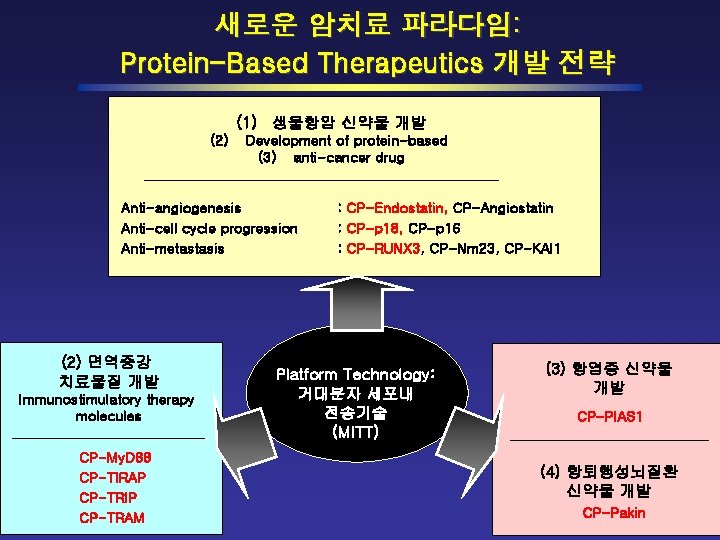
새로운 암치료 파라다임: Protein-Based Therapeutics 개발 전략 (1) 생물항암 신약물 개발 (2) Development of protein-based (3) anti-cancer drug Anti-angiogenesis Anti-cell cycle progression Anti-metastasis (2) 면역증강 치료물질 개발 Immunostimulatory therapy molecules CP-My. D 88 CP-TIRAP CP-TRIP CP-TRAM : CP-Endostatin, CP-Angiostatin : CP-p 18, CP-p 16 : CP-RUNX 3, CP-Nm 23, CP-KAI 1 Platform Technology: 거대분자 세포내 전송기술 (MITT) (3) 항염증 신약물 개발 CP-PIAS 1 (4) 항퇴행성뇌질환 신약물 개발 CP-Pakin

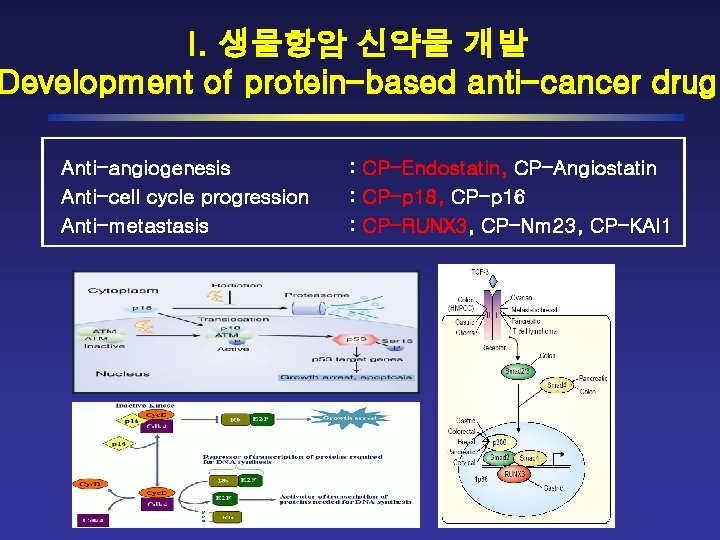
I. 생물항암 신약물 개발 Development of protein-based anti-cancer drug Anti-angiogenesis Anti-cell cycle progression Anti-metastasis : CP-Endostatin, CP-Angiostatin : CP-p 18, CP-p 16 : CP-RUNX 3, CP-Nm 23, CP-KAI 1

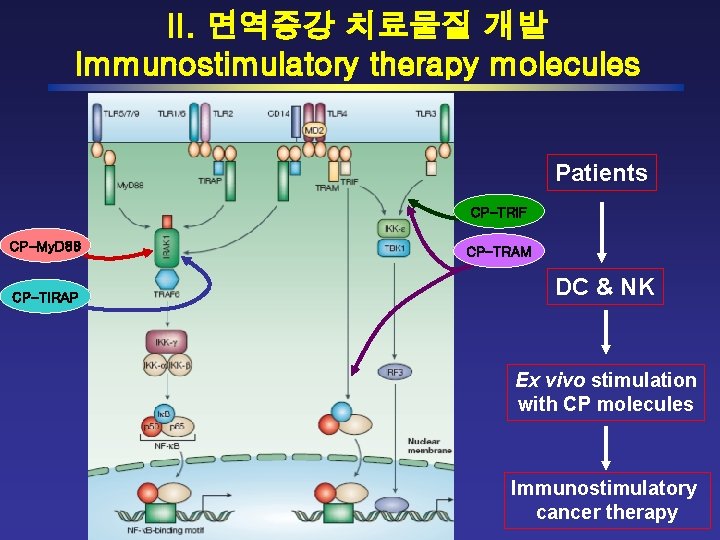
II. 면역증강 치료물질 개발 Immunostimulatory therapy molecules Patients CP-TRIF CP-My. D 88 CP-TIRAP CP-TRAM DC & NK Ex vivo stimulation with CP molecules Immunostimulatory cancer therapy

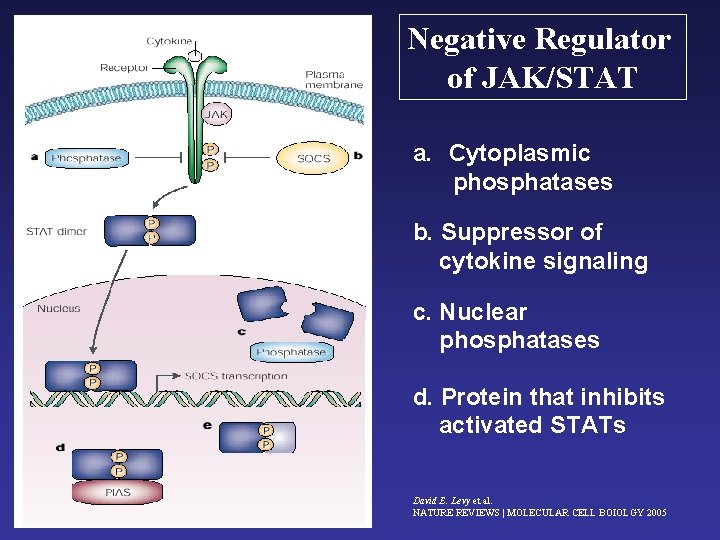
Negative Regulator of JAK/STAT a. Cytoplasmic phosphatases b. Suppressor of cytokine signaling c. Nuclear phosphatases d. Protein that inhibits activated STATs David E. Levy et al. NATURE REVIEWS | MOLECULAR CELL BOIOLGY 2005

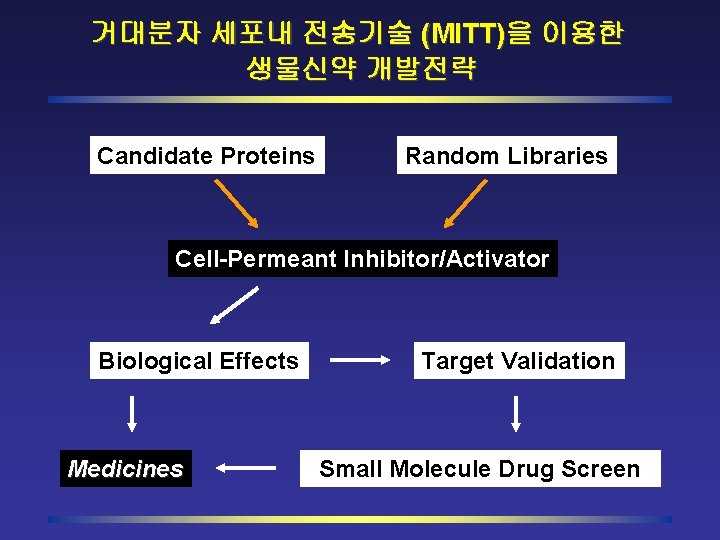
거대분자 세포내 전송기술 (MITT)을 이용한 생물신약 개발전략 Candidate Proteins Random Libraries Cell-Permeant Inhibitor/Activator Biological Effects Medicines Target Validation Small Molecule Drug Screen




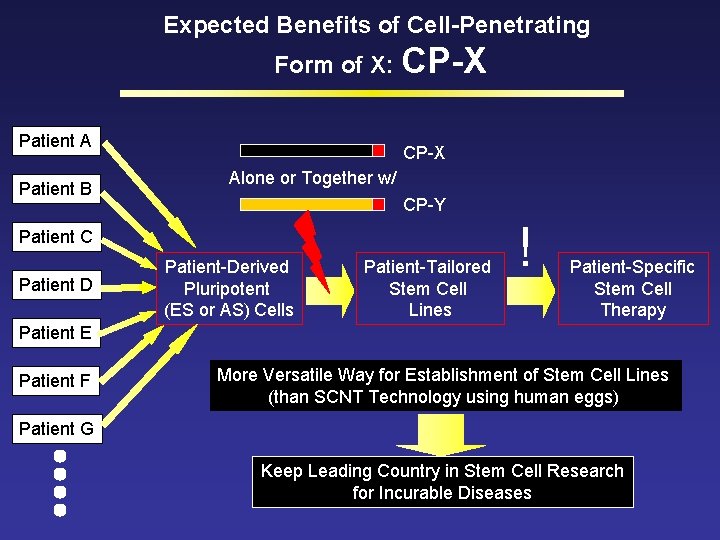
Expected Benefits of Cell-Penetrating Form of X: CP-X Patient A Patient B CP-X Alone or Together w/ CP-Y Patient C Patient D Patient-Derived Pluripotent (ES or AS) Cells Patient-Tailored Stem Cell Lines ! Patient-Specific Stem Cell Therapy Patient E Patient F More Versatile Way for Establishment of Stem Cell Lines (than SCNT Technology using human eggs) Patient G Keep Leading Country in Stem Cell Research for Incurable Diseases

New Drug & Contribution to Patient Treatment Research & Discovery Investment & Development