Plastics in the Microwave Oven CE 435 Presented

























- Slides: 33


Plastics in the Microwave Oven CE 435 Presented by: Daniel Fulcher Christopher Hunter Martin Schaefer April 17, 2001

Introduction • U. S. households owning a microwave oven increased from 15% in 1980 to 78% by 1989 • Gorman’s New Product News reported the number of new microwave product introductions increased from 278 in 1986 to almost 1000 in 1988 • Microwave frequency of ~2. 45 GHz ensures effective penetration into the food and even food heating • Annual revenues of 3 billion dollars

Scope • Max temperatures of 230 o. F for microwave ovens and 400 o. F for dual oven applications • Containers should be sturdy, rigid in shape and capable of supporting its contents • Limited migration of additives, colorants, or fillers • Federal Food and Drug Administrative approval

Home Meal Replacement Packaging

HMR Packaging • Crystallized polyethylene terephthalate (CPET) • Polyphenylene oxide, high impact polystyrene blend (PPO/HIPS)

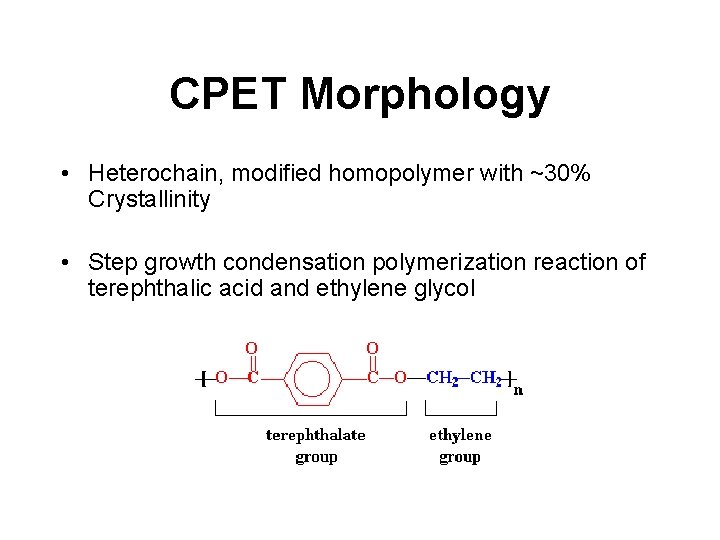
CPET Morphology • Heterochain, modified homopolymer with ~30% Crystallinity • Step growth condensation polymerization reaction of terephthalic acid and ethylene glycol

CPET Properties • Crystallization of PET increases the upper temperature resistance from 230°F to 400°F • Balance between strength properties and temperature resistance • Crystallinity of 28%-32% and an intrinsic viscosity of . 85 to. 95 • Two-layer structure of CPET and APET

CPET Properties • Good O 2 and CO 2 barrier properties • Acceptable water barrier properties • Easily colored with black carbon

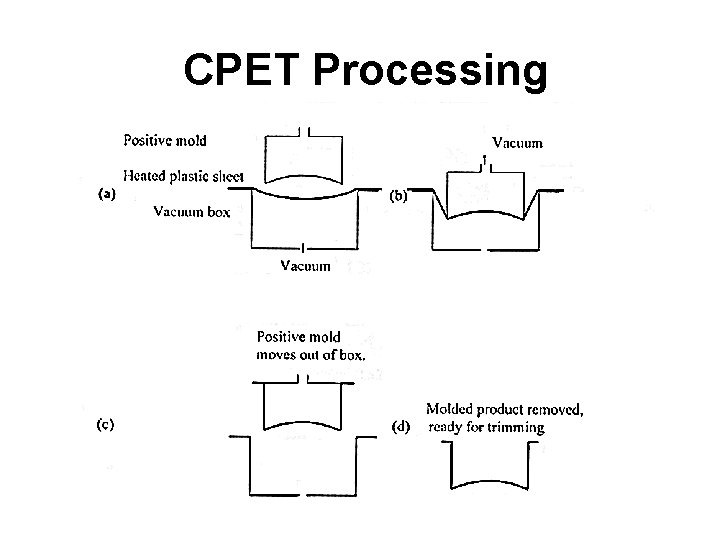
CPET Processing • In 2000, 84 million pounds produced at ~. 52 $/lb • Eastman Chemical Company produces CPET resin under the product name of Versa. Tray® • Thermoformed • Vacuum Snap-Back Thermoforming

CPET Processing

PPO/HIPS Morphology • Fully miscible blended amorphous polymer • Noryl® manufactured by General Electric Plastics • Noryl® PPO/HIPS blend is approximately 25% PPO and 75% HIPS by weight

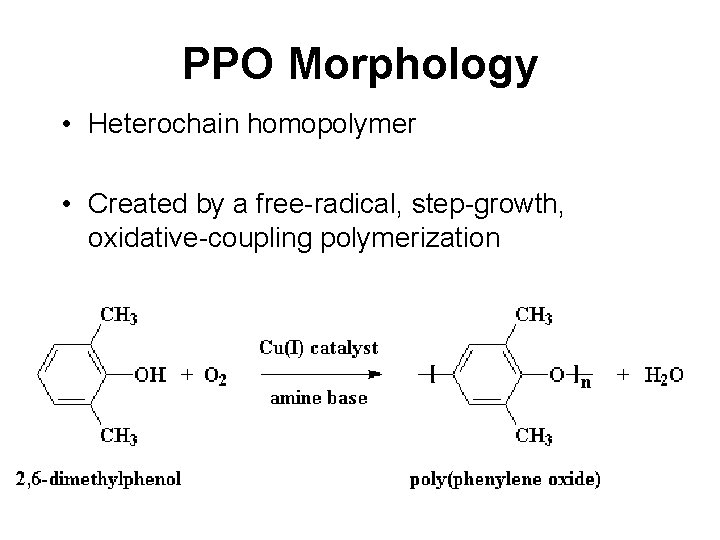
PPO Morphology • Heterochain homopolymer • Created by a free-radical, step-growth, oxidative-coupling polymerization

HIPS Morphology • Grafted copolymer of cis-1, 4 -poly(1, 3 -butadiene) and styrene monomer • Polystyrene chains are atactic • 2% to 15% by weight of polybutadiene

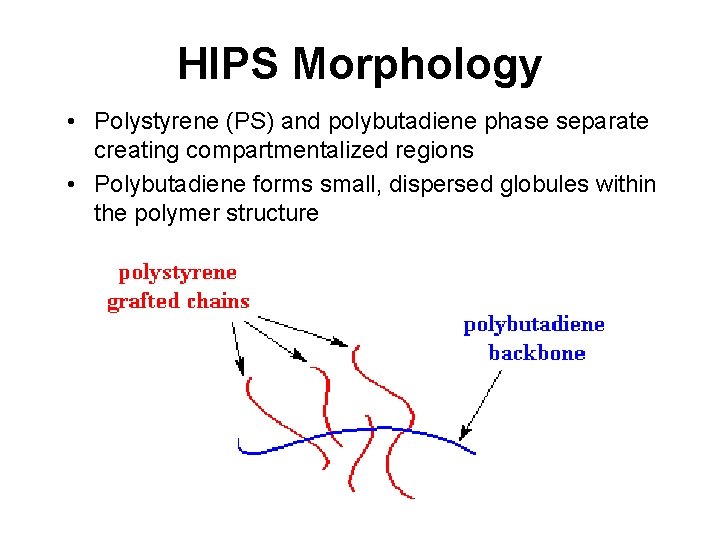
HIPS Morphology • Polystyrene (PS) and polybutadiene phase separate creating compartmentalized regions • Polybutadiene forms small, dispersed globules within the polymer structure

PPO/HIPS Properties • Improvements in final blend properties are in direct proportion to the amount of PPO added • PPO is added to HIPS to increase temperature resistance from approximately 180°F to 230°F • Blending with HIPS improves PPO’s low resin flow characteristics

PPO/HIPS Properties • Small, dispersed globules of polybutadiene elastomer within the PS improves the blend impact strength • PPO improves the poor gaseous and water vapor barrier properties of HIPS • Easily colored with black carbon

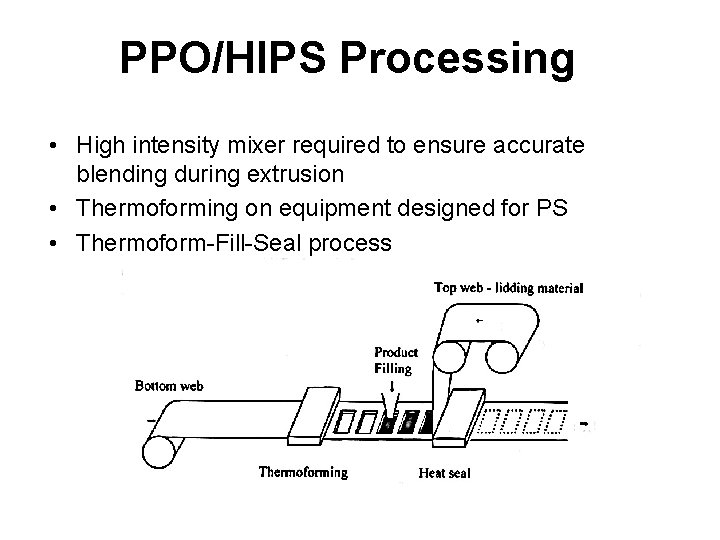
PPO/HIPS Processing • High intensity mixer required to ensure accurate blending during extrusion • Thermoforming on equipment designed for PS • Thermoform-Fill-Seal process

HMR Covers • Heat-seal the food containers • Most widely used film is multi-layer polypropylene • Adhesives cause complications in migration testing • PP is a low cost packaging film ~. 53$/lb

Food Storage Containers

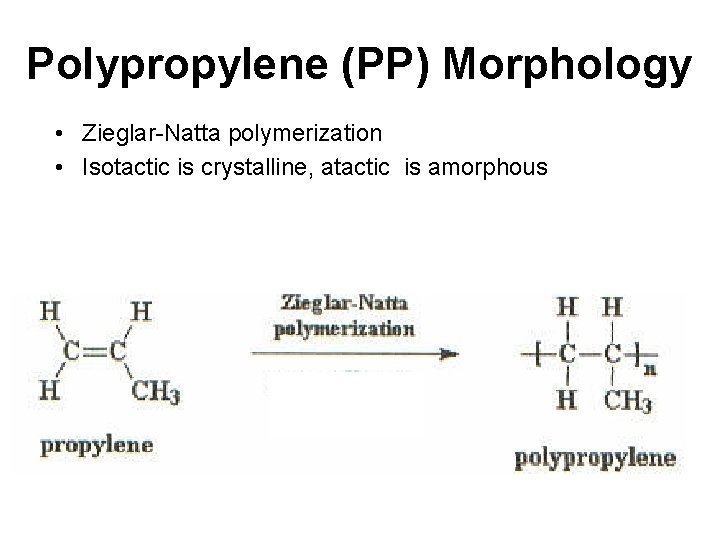
Polypropylene (PP) Morphology • Zieglar-Natta polymerization • Isotactic is crystalline, atactic is amorphous

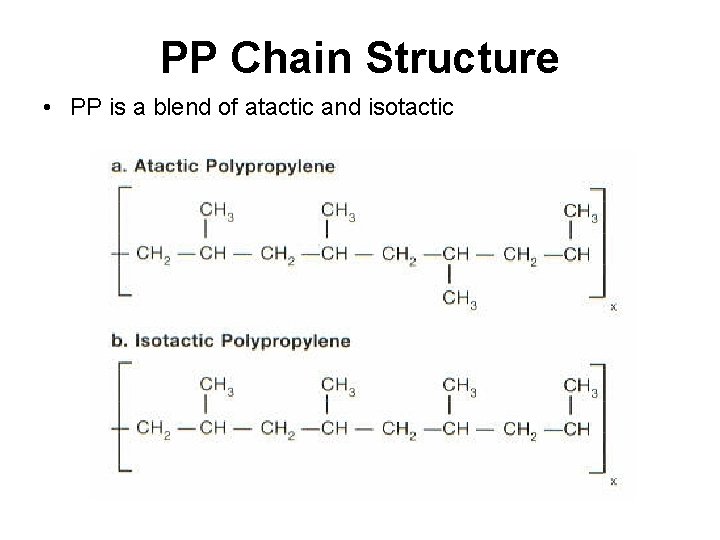
PP Chain Structure • PP is a blend of atactic and isotactic

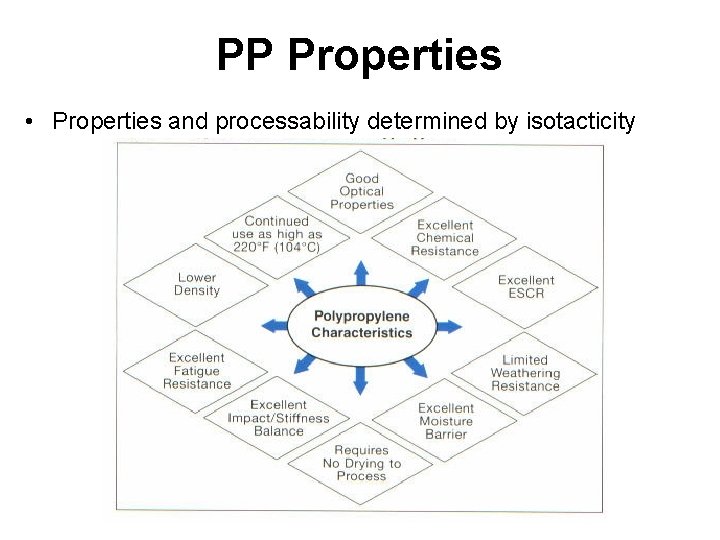
PP Properties • Properties and processability determined by isotacticity

PP Processing • 1550 million pounds produced in 2000 at ~. 53 $/lb. • Injected or blow molded • INSPIRE® PP produced by Dow Plastics

Regulations • The code of federal regulations provides guidance on polymers allowed in the direct contact of food. • Article 177 presents data specific to each polymer • Composition specifications, allowable coatings, modifiers, emulsifiers, and testing criteria

CPET Regulations • Article 177. 1630 • Emulsifier < 2. 0 percent of the dry weight • Additional substances allowed must be generally recognized as safe by the FDA • < 0. 02 mg/in 2 of chloroform-soluble extractives for heptane and distilled water migration tests

PPO/HIPS Regulations • Articles 177. 1810 and 177. 2460 • PS: molecular weight > 29, 000 and soluble in toluene • Maximum extractable fractions of chloroform in distilled water and 50% ethanol are 0. 0039 mg/cm 2 • PPO: intrinsic viscosity > 0. 30 deciliter/gram • < 0. 02 weight percent extractable with n-heptane

PP Regulations • Article 177. 1520 • Density of 0. 880 -0. 913 and m. p. 160 C - 180 C • Maximum extractable fraction of n-hexane is 6. 4% • Maximum soluble fraction of xylene is 9. 8%

Testing and Health Issues • No specific requirements for microwave food containers • FDA provides guidance for proper microwave migration testing protocols • Migration tests using food simulants (e. g. cooking oil) • Migration testing at the highest cooking temperature

Testing and Health Issues • Size and type of food, cooking time, and food geometry play part in the maximum temperature • Consult with FDA before deciding on a migration testing protocol for microwave only containers • Plasticizers have been linked to endocrine disorders • Further study needed to determine possible health risks due to migration of polymer additives

Conclusion • Primary polymer materials: CPET, PPO/HIPS, PP • Applications ranging from single use HMR to repeated use food storage containers • Essentially no health risks • Cost effective containers

Questions?

References annotated in the Polymers in the Microwave written report. 1. Rubbright, H. A. , Davis N. O. , The Microwave Decade, Packaging Strategies, West Chester, PA, 1989. 2. Becker, R. “As a Matter of Fact. ” Chem Matters April 2000: Volume 18 Number 2. Online. Available HTTP: www. acs. org/education/curriculum/chemmatt. html 3. Dow Plastics, Polypropylene Resins Molding Guide. The Dow Chemical Company, 1998. 4. Brady, A. L. , Marsh, K. S. , The Wiley Encyclopedia of Packaging Technology. New York: John Wiley and Sons, Inc. , 1997. 5. Hanlon, J. F. , Kelsey, R. J. , Forcinio, H. E. , Handbook of Package Engineering. Technomic Publishing Company, Inc. , 1998. 6. Selke, S. E. , Understanding Plastics Packaging Technology. Cincinnati: Hanser/Gardner Publications, Inc. , 1997. 1999 CFR Title 21 Food and Drugs, Volume 3. Part 177 Indirect Food Additives: Polymers. Online. Available HTTP: www. access. gpo. gov/nara/cfr/waisidx_99/21 cfr 177_99. html 8. Eastman Chemical Company. Online. Available HTTP: www. eastman. com/productfiles/prod 0018. html 9. General Electric Company. Plastics Division. Online. Available HTTP: www. geplastics. com/resins/techsolution/technifacts. html 10. Department of Polymer Science, University of Southern Mississippi, Macrogalleria, 1996. Online. Available HTTP: www. psrc. usm. edu/macrog/floor 2. html 11. Modern Plastics Encyclopedia. New York: Mc. Graw-Hill Companies, Inc. , 2001. 12. U. S. Food and Drug Administration. Online. Available HTTP: vm. cfsan. fda. gov/~dms/opa-pmnc. html 13. U. S. Food and Drug Administration. FDA Consumer Magazine, Volume 97, Number 11. Maryland; FDA, 1998.