Plastics and Synthetic Fibres Intermediate 2 Chemistry Unit

Plastics and Synthetic Fibres Intermediate 2 Chemistry Unit 2(d)

Examples of plastics
![Examples of plastics • • Polythene [poly(ethene)] Polystyrene Perspex PVC Nylon Bakelite Formica Silicones Examples of plastics • • Polythene [poly(ethene)] Polystyrene Perspex PVC Nylon Bakelite Formica Silicones](http://slidetodoc.com/presentation_image_h/c9ff579a0c128e4e73807d4578eb0f48/image-3.jpg)
Examples of plastics • • Polythene [poly(ethene)] Polystyrene Perspex PVC Nylon Bakelite Formica Silicones
![PVC [poly(vinyl chloride)] PVC [poly(vinyl chloride)]](http://slidetodoc.com/presentation_image_h/c9ff579a0c128e4e73807d4578eb0f48/image-4.jpg)
PVC [poly(vinyl chloride)]

Nylon

Bakelite

Formica

Silicones

Where do plastics come from? • Crude oil – Fractional distillation – Cracking • Alkenes

Uses related to properties • • Flexible Watertight Shatter-proof Light

Non-biodegradable • Can’t be broken down by living things. • “Biopol” is a recently developed biodegradable plastic.

Other problems with plastics • Low density • Durable • Burn or smoulder to give off toxic fumes – Carbon monoxide – Hydrogen chloride – Hydrogen cyanide

Types of plastic • Thermoplastic – Can be reshaped on heating • Thermosetting plastic – Cannot be reshaped on heating

Chemistry of plastics • Plastics are examples of polymers – These are long chain molecules made from smaller molecules joining together • These smaller molecules are called monomers • There are two types of polymer: – Addition polymer – Condensation polymer

Addition polymers

Addition polymers • Like all polymers, these are made from monomers. • For an addition polymer, these monomers always have a C=C • These join together in a reaction called addition polymerisation. – view animation
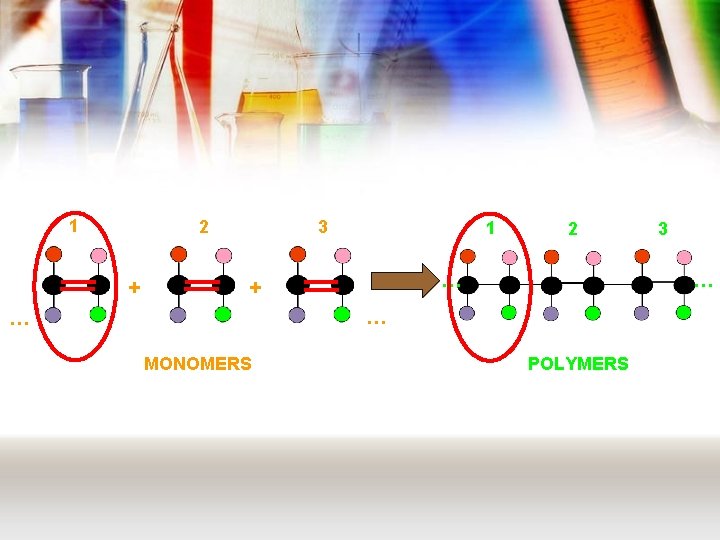
1 2 + 3 1 2 … + … … … MONOMERS 3 POLYMERS
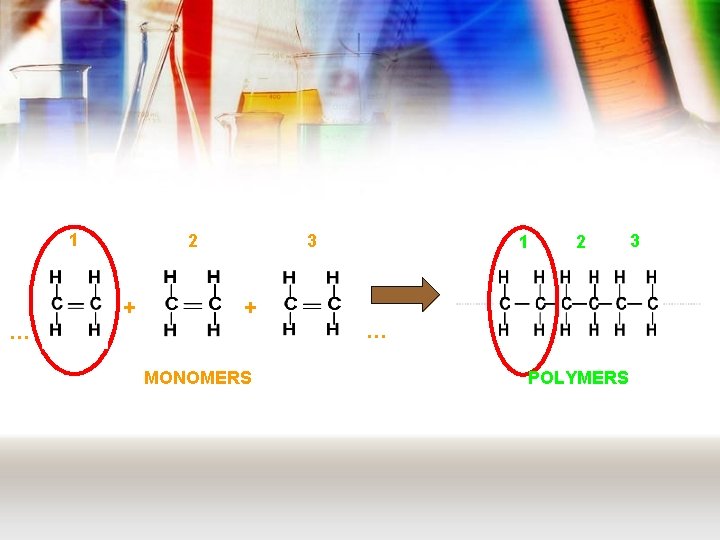
1 2 + 3 1 2 + … … MONOMERS POLYMERS 3
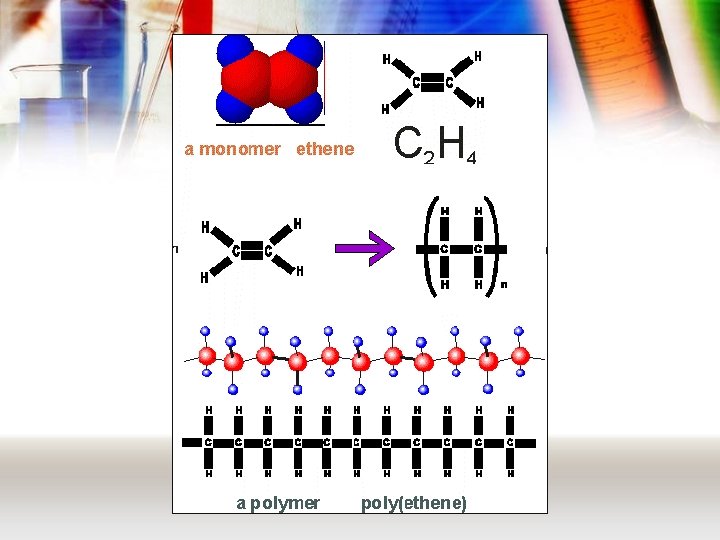

Naming polymers • The name of the polymer is made by taking the name of the monomer, putting brackets around it, and adding “poly” in front – Poly(ethene) is made from ethene; – Poly(vinyl chloride) is made from vinyl chloride.

Naming practice • Name the polymer made from: – Propene – Chloroethene • Name the monomer used to make: – Poly(heptene) – Poly(phenylethene)

Drawing polymers, monomers and the repeating unit

C=C H H C 6 H 5 H Poly(phenylethene)


Condensation polymers

• What type of molecule is produced from an alkanol and alkanoic acid? – Esters • Polyesters are made from similar molecules.

Polyesters
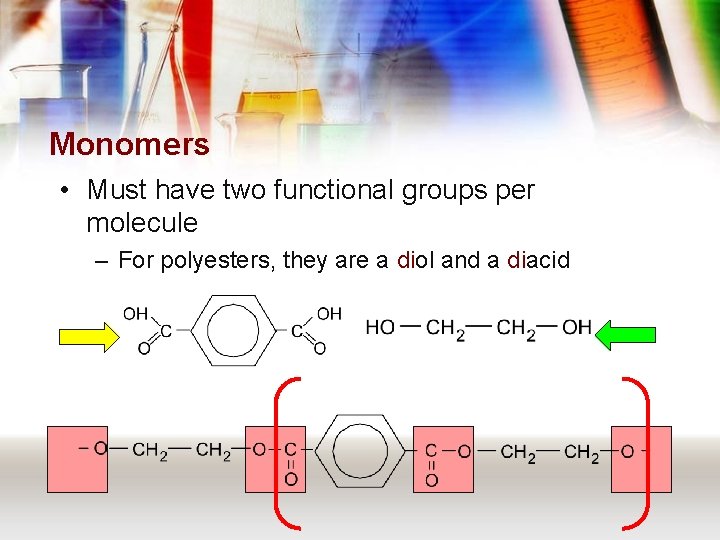
Monomers • Must have two functional groups per molecule – For polyesters, they are a diol and a diacid

Condensation polymerisation • The joining up of two molecules, producing water, is condensation. • Because giant molecules are made, this is condensation polymerisation. • Specifically with a diol and a diacid, a polyester is produced.

Polyamides • Are produced when an amine group reacts with a carboxyl group.

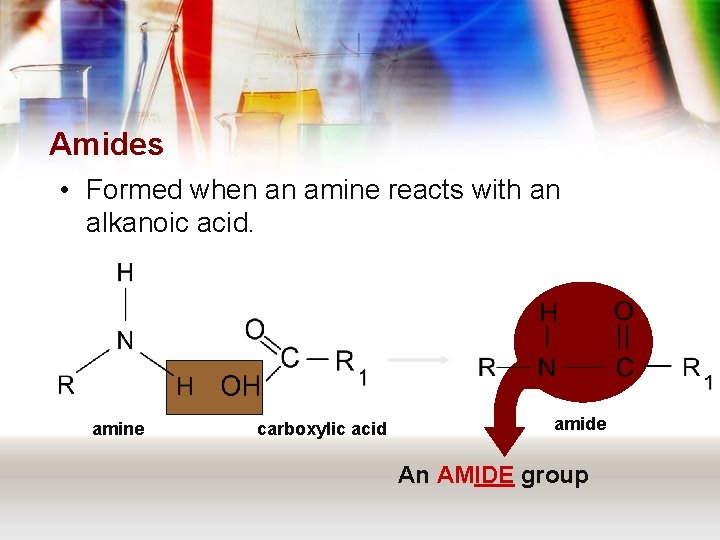
Amides • Formed when an amine reacts with an alkanoic acid. amine carboxylic acid amide An AMIDE group

Polyamides • Are condensation polymers. • Formed when a diamine reacts with a diacid.

Recently developed plastics • Kevlar – Very strong • Poly(ethenol) – Water soluble • Biopol – Biodegradable

Kevlar
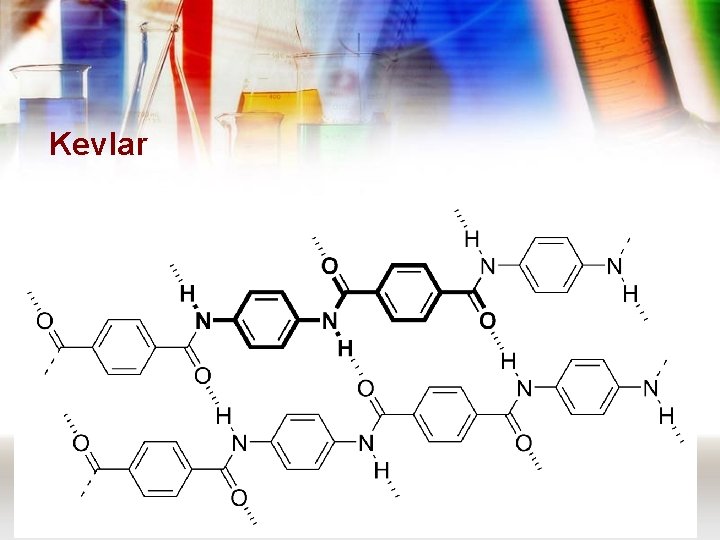
Kevlar
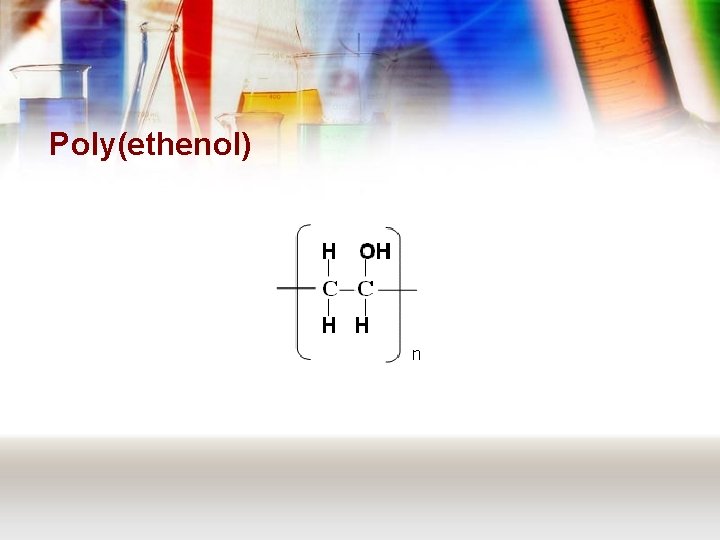
Poly(ethenol)

Poly(ethenol)

- Slides: 39