Plastic deformation Extension of solid under stress becomes
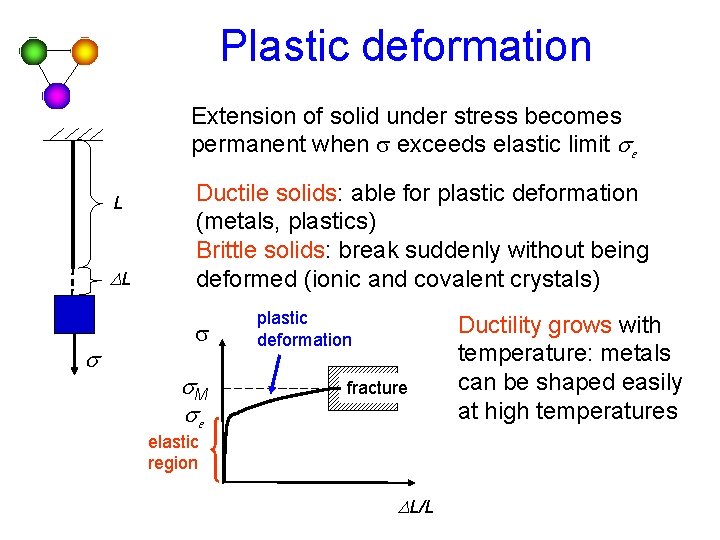
Plastic deformation Extension of solid under stress becomes permanent when s exceeds elastic limit se L DL s Ductile solids: able for plastic deformation (metals, plastics) Brittle solids: break suddenly without being deformed (ionic and covalent crystals) s s. M se plastic deformation fracture elastic region DL/L Ductility grows with temperature: metals can be shaped easily at high temperatures
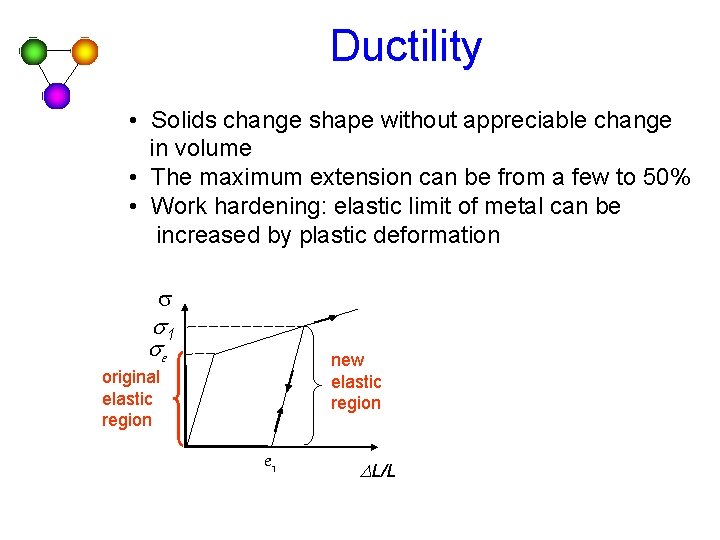
Ductility • Solids change shape without appreciable change in volume • The maximum extension can be from a few to 50% • Work hardening: elastic limit of metal can be increased by plastic deformation s s 1 se new elastic region original elastic region e 1 DL/L
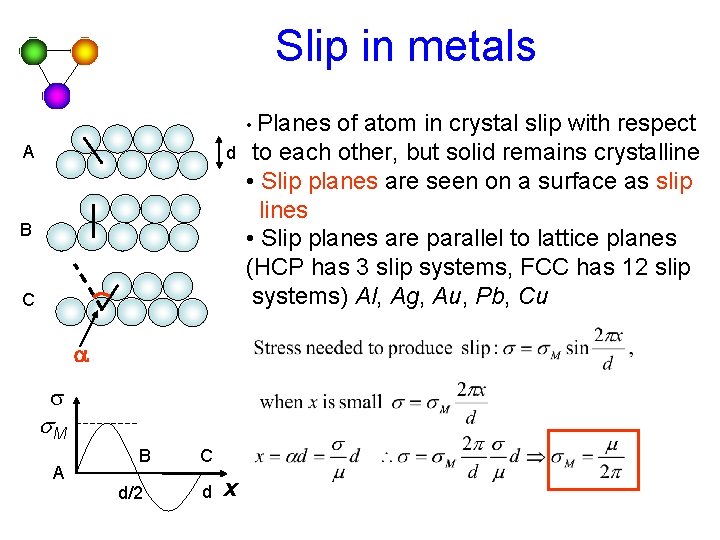
Slip in metals • Planes A d B C a s s. M A B d/2 C d x of atom in crystal slip with respect to each other, but solid remains crystalline • Slip planes are seen on a surface as slip lines • Slip planes are parallel to lattice planes (HCP has 3 slip systems, FCC has 12 slip systems) Al, Ag, Au, Pb, Cu
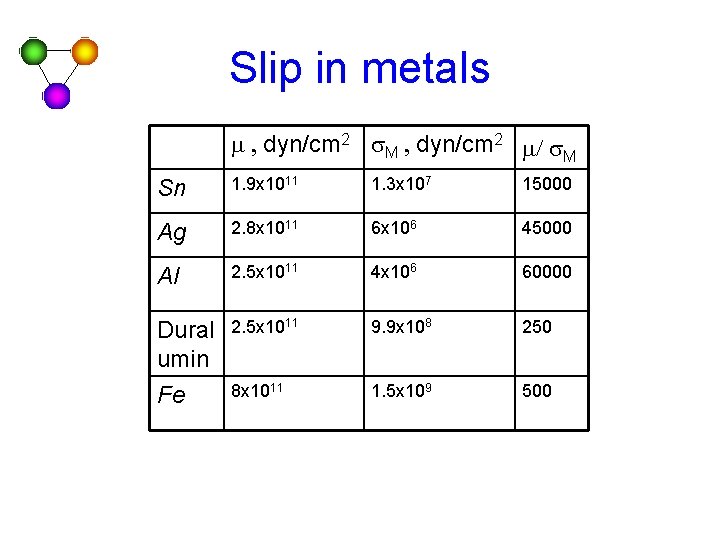
Slip in metals m , dyn/cm 2 s. M , dyn/cm 2 m/ s. M Sn 1. 9 x 1011 1. 3 x 107 15000 Ag 2. 8 x 1011 6 x 106 45000 Al 2. 5 x 1011 4 x 106 60000 Dural umin Fe 2. 5 x 1011 9. 9 x 108 250 8 x 1011 1. 5 x 109 500
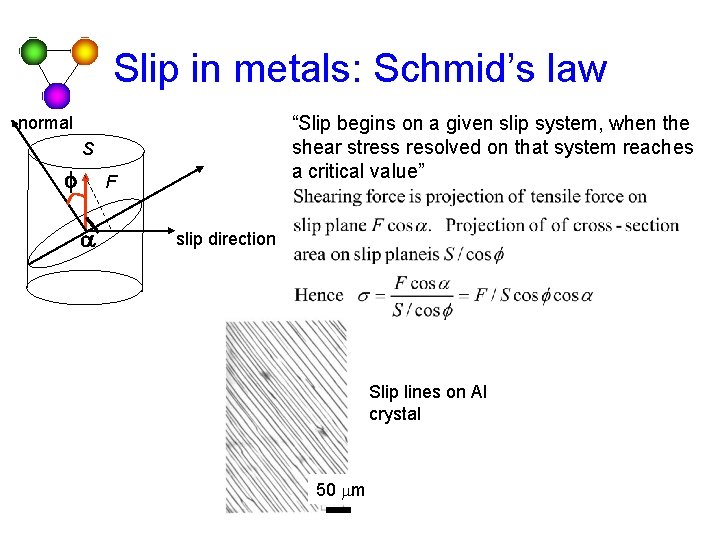
Slip in metals: Schmid’s law “Slip begins on a given slip system, when the shear stress resolved on that system reaches a critical value” normal S f F a slip direction Slip lines on Al crystal 50 mm

Dislocations: edge Edge dislocation in simple cubic crystal Burger vector ^ dislocation line Burger circuit- path around dislocation line: 3 steps to->, 3 steps down, 3 steps to <-, 3 steps up. “Failure” to close this circuit is Burger vector b Dislocation may “glide”- little energy should be supply to slip crystal. Elastic limit in real crystal << ideal crystal.

Dislocations: screw dislocation line Atoms/ molecules bond to screw dislocation during crystal growth. Burger vector II dislocation line • Density of dislocations: from 102 dislocations/cm 2 in the best Ge and Si to 10 12 dislocations/cm 2 in heavily deformed metals • Motion of dislocation can be blocked by: another dislocation, grain boundary, point defect • Work-hardening: annealing decrease dislocation density to 106 dislocations/cm 2. After plastic deformation density increases to 1012 dislocations/cm 2, but motion of the dislocation can be blocked by pinning at other dislocations

Dislocations: grain boundaries grain boundary dislocations • Dislocations are blocked by grain boundaries -> slip is blocked • Smaller grain size-> larger surface of grain boundaries -> larger elastic limit • Empirical equation for maximum elastic stress (Petch) s. M =A+Bd-1/2, d -grain diameter • Elastic limit in cooper doubles when grain size falls from 100 mm to 25 mm

Alloys contain several mixed constituent metals • Substitutional solution: dissolve atoms replace those of basic metal (Cu in Ni ) • Interstitual solution: added elements are lodged in in interstitial sites (C in Fe) • Motion of dislocations is impeded by irregularities -> elastic limit increases • Elastic limit C, N Si Mn Mo Ni 1 2 Concentration of alloyed element %

Alloys with precipitates Alloy contains two phases: predominant phase of matrix and precipitated phase is dispersed in form of fine grains. This take place at high concentration of added element Motion of dislocations are blocked by this grains -> larger elastic limit. Dislocation can pass the precipitated grains

Alloys with Guinier-Preston zones Alloy with GP zones is intermediate between homogeneous and precipitated phases: added element is concentrated in ~10 nm GP zones. These zones block motion of dislocation Structure hardening Al+ 5% Cu alloy is homogeneous at 550 OC Cooled by quenching to room temperature Alloy with GP zones: high elastic limit Alloy in Concord: Al +2% Cu +1. 5% Mg+1% Ni has s. M~450 Mpa with operation temperature up to 120 O C
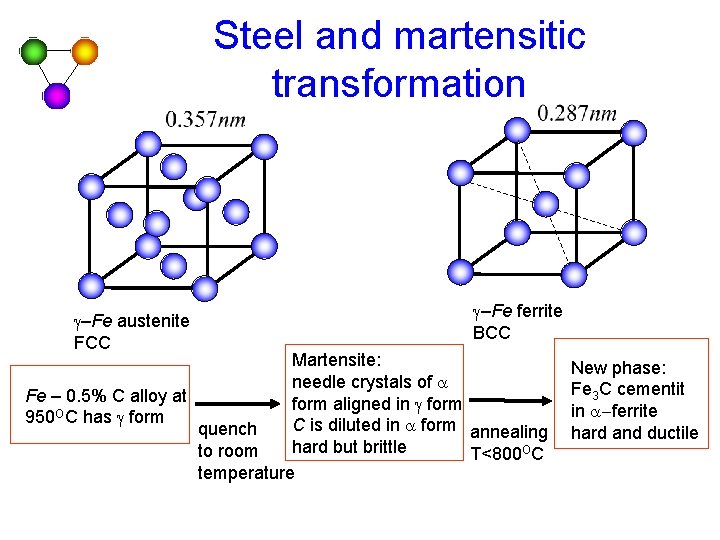
Steel and martensitic transformation g–Fe austenite FCC g–Fe ferrite BCC Martensite: needle crystals of a Fe – 0. 5% C alloy at form aligned in g form 950 OC has g form C is diluted in a form annealing quench hard but brittle to room T<800 OC temperature New phase: Fe 3 C cementit in a-ferrite hard and ductile

Interaction of dislocation with impurities s s. U s. L U L elastic region work hardening plastic deformation DL/L Impurities diffuse to dislocations and form “clouds”-> dislocations are pinned -> higher elastic limit When s exceeds s. U dislocations escapes impurities -> stress needed for plastic deformation decreases
- Slides: 13