PLASMA SURFACE INTERACTIONS FOR ATMOSPHERIC PRESSURE FUNCTIONALIZATION OF












![BREAKDOWN IN HID LAMP: THE IONIZATION FRONT Te [Sources] 10 e. V [e] MCS BREAKDOWN IN HID LAMP: THE IONIZATION FRONT Te [Sources] 10 e. V [e] MCS](https://slidetodoc.com/presentation_image_h/2f0ddee152c78505f7f4ae69f3e949f9/image-19.jpg)
![BREAKDOWN IN HID LAMP: THE IONIZATION FRONT Te 10 e. V [Sources] [e] 1021 BREAKDOWN IN HID LAMP: THE IONIZATION FRONT Te 10 e. V [Sources] [e] 1021](https://slidetodoc.com/presentation_image_h/2f0ddee152c78505f7f4ae69f3e949f9/image-20.jpg)


![REPETITIVELY PULSED DISCHARGES – [e] · Electron avalanche from the powered electrode. · The REPETITIVELY PULSED DISCHARGES – [e] · Electron avalanche from the powered electrode. · The](https://slidetodoc.com/presentation_image_h/2f0ddee152c78505f7f4ae69f3e949f9/image-26.jpg)
![[O] – WITHOUT AND WITH FORCED GAS FLOW Without forced flow [O] 1015 cm-3 [O] – WITHOUT AND WITH FORCED GAS FLOW Without forced flow [O] 1015 cm-3](https://slidetodoc.com/presentation_image_h/2f0ddee152c78505f7f4ae69f3e949f9/image-31.jpg)
![[O] – WITHOUT AND WITH FORCED GAS FLOW Without forced flow [O] 1015 cm-3 [O] – WITHOUT AND WITH FORCED GAS FLOW Without forced flow [O] 1015 cm-3](https://slidetodoc.com/presentation_image_h/2f0ddee152c78505f7f4ae69f3e949f9/image-32.jpg)
![[O 3] – WITHOUT AND WITH FORCED GAS FLOW Without forced flow [O 3] [O 3] – WITHOUT AND WITH FORCED GAS FLOW Without forced flow [O 3]](https://slidetodoc.com/presentation_image_h/2f0ddee152c78505f7f4ae69f3e949f9/image-33.jpg)
![[O 3] – WITHOUT AND WITH FORCED GAS FLOW Without forced flow [O 3] [O 3] – WITHOUT AND WITH FORCED GAS FLOW Without forced flow [O 3]](https://slidetodoc.com/presentation_image_h/2f0ddee152c78505f7f4ae69f3e949f9/image-34.jpg)
![[OH] – WITHOUT AND WITH FORCED GAS FLOW Without forced flow [OH] 1014 cm-3 [OH] – WITHOUT AND WITH FORCED GAS FLOW Without forced flow [OH] 1014 cm-3](https://slidetodoc.com/presentation_image_h/2f0ddee152c78505f7f4ae69f3e949f9/image-35.jpg)
![[OH] – WITHOUT AND WITH FORCED GAS FLOW Without forced flow [OH] 1014 cm-3 [OH] – WITHOUT AND WITH FORCED GAS FLOW Without forced flow [OH] 1014 cm-3](https://slidetodoc.com/presentation_image_h/2f0ddee152c78505f7f4ae69f3e949f9/image-36.jpg)







- Slides: 54

PLASMA SURFACE INTERACTIONS FOR ATMOSPHERIC PRESSURE FUNCTIONALIZATION OF POLYMERS Mark J. Kushner Iowa State University Ames, IA 50011 USA mjk@iastate. edu http: //uigelz. ece. iastate. edu March 2007 EUJapan_0307_01

ACKNOWLEDGEMENTS · Group Members · Ananth N. Bhoj · Ramesh Arakoni · Natalie Babeava · Funding Agencies · · EUJapan_0307_02 3 M Corporation Semiconductor Research Corporation National Science Foundation Air Force Office of Scientific Research Iowa State University Optical and Discharge Physics

AGENDA · Introduction to plasma functionalization of polymers · Description of non. PDPSIM · Corona treatment of Polymers: Pulsing, Flowing, Moving · Optimizing Uniformity · Concluding Remarks EUJapan_0307_03 Iowa State University Optical and Discharge Physics

FUNCTIONALIZATION OF POLYMER SURFACES Functionalization occurs by chemical interaction of plasma produced species - ions, radicals and photons with the surface. (a) (b) (c) · Example: H abstraction by O atom enables affixing O atoms as a peroxy site. · Increase surface energy increase wettability. Process treats the top few layers. Wettability on PE film with 3 zones of treatment. Courtesy: http: //www. polymer-surface. com EUJapan_0307_04 Iowa State University Optical and Discharge Physics

SURFACE MODIFICATION OF POLYMERS · Pulsed atmospheric filamentary discharges (coronas) routinely treat commodity polymers like poly-propylene (PP) and polyethylene (PE). · Filamentary Plasma 10 s – 200 m EUJapan_0307_05 Iowa State University Optical and Discharge Physics

COMMERCIAL CORONA PLASMA EQUIPMENT · Sherman Treaters · Tantec, Inc. EUJapan_0307_06 Iowa State University Optical and Discharge Physics

MODELING OF FUNCTIONALIZATION OF POLYMERS · Goals: · Investigate fundamental physics of plasma-surface interactions for functionalization for industrially relevant conditions. · Develop scaling laws to be able to customize surface functionalization. · Methodology: · Integrated multi-scale modeling of corona discharge. · Test system: Functionalization of polypropylene in oxygen containing plasmas. EUJapan_0307_07 Iowa State University Optical and Discharge Physics

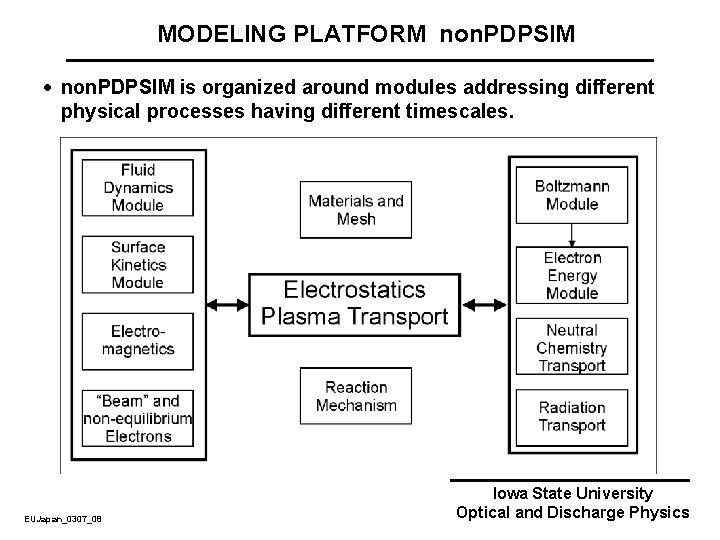
MODELING PLATFORM non. PDPSIM · non. PDPSIM is organized around modules addressing different physical processes having different timescales. EUJapan_0307_08 Iowa State University Optical and Discharge Physics

CHARGED PARTICLE TRANSPORT · Poisson’s equation for the electric potential · F = electric potential · qj = charge Nj = density s = surface charge · Transport equations for conservation of the charged species j · Gj = Flux Sj = sources due to collisions, photons · Surface charge balance EUJapan_0307_09 Iowa State University Optical and Discharge Physics

CHARGED PARTICLE TRANSPORT · Electron fluxes given by Sharfetter-Gummel form · = mobility, D = diffusion coefficient, E = Electric field · Automatically selects upwind-or-downwind · Ion fluxes given by SG or by accounting for full momentum · Pj = partial pressure · v = velocity ij = collision frequency · Finite volume discretization to insure 100% conservative as solution of Poisson’s equation requires 10 -6 to 10 -7 resolution. EUJapan_0307_10 Iowa State University Optical and Discharge Physics

BULK-BEAM ELECTRON ENERGY TRANSPORT · Bulk Electrons: Continuum electron energy equation. · Integrate implicitly using method of Successive-Over-Relaxation. · Transport coefficients obtained by solving (and periodically updating) spatially homogeneous Boltzmann’s equation for the electron energy distribution. · Secondary electrons emitted in high E-fields: Monte Carlo simulation. EUJapan_0307_11 Iowa State University Optical and Discharge Physics

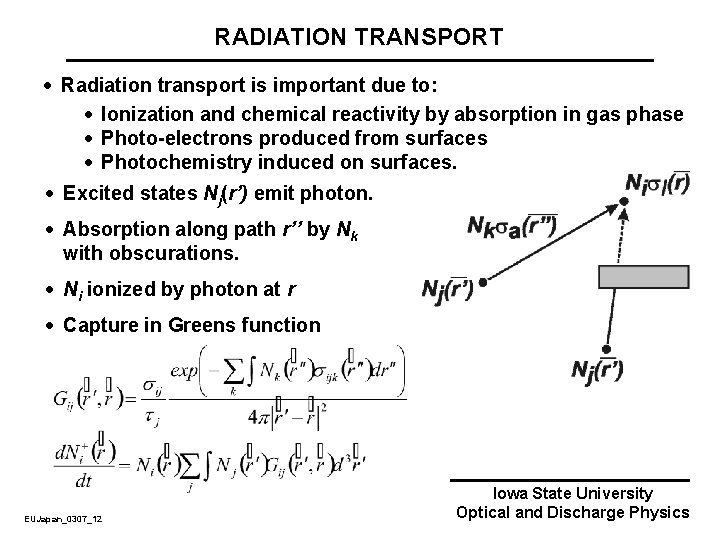
RADIATION TRANSPORT · Radiation transport is important due to: · Ionization and chemical reactivity by absorption in gas phase · Photo-electrons produced from surfaces · Photochemistry induced on surfaces. · Excited states Nj(r’) emit photon. · Absorption along path r’’ by Nk with obscurations. · Ni ionized by photon at r · Capture in Greens function EUJapan_0307_12 Iowa State University Optical and Discharge Physics

FLUID MODULE : NEUTRAL PARTICLE TRANSPORT · Fluid averaged values of mass density, mass momentum and thermal energy density obtained in using unsteady algorithms. Continuity: Momentum: Energy: Individual neutral species diffuse within the bulk flow. EUJapan_0307_13 Iowa State University Optical and Discharge Physics

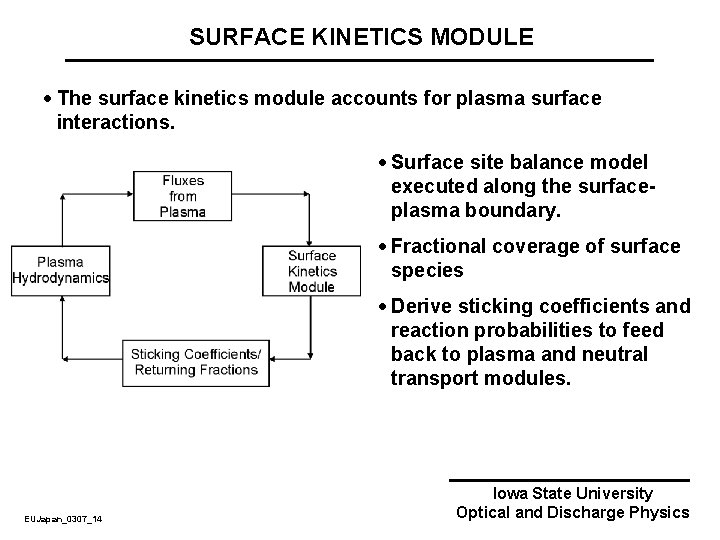
SURFACE KINETICS MODULE · The surface kinetics module accounts for plasma surface interactions. · Surface site balance model executed along the surfaceplasma boundary. · Fractional coverage of surface species · Derive sticking coefficients and reaction probabilities to feed back to plasma and neutral transport modules. EUJapan_0307_14 Iowa State University Optical and Discharge Physics

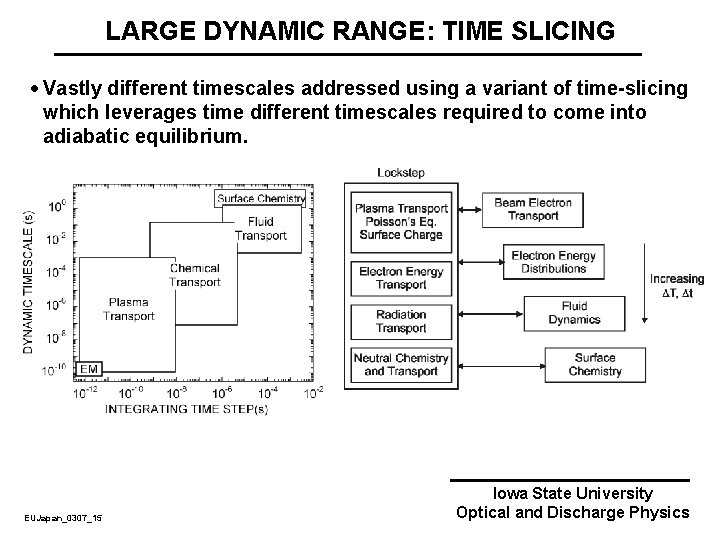
LARGE DYNAMIC RANGE: TIME SLICING · Vastly different timescales addressed using a variant of time-slicing which leverages time different timescales required to come into adiabatic equilibrium. EUJapan_0307_15 Iowa State University Optical and Discharge Physics

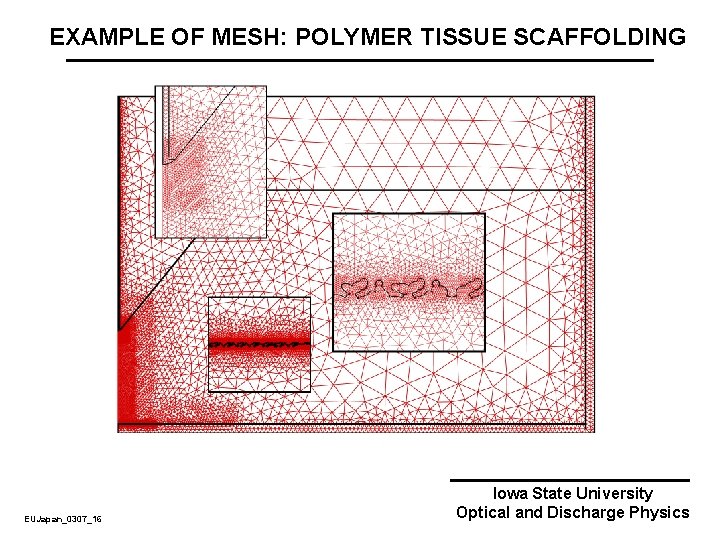
EXAMPLE OF MESH: POLYMER TISSUE SCAFFOLDING EUJapan_0307_16 Iowa State University Optical and Discharge Physics

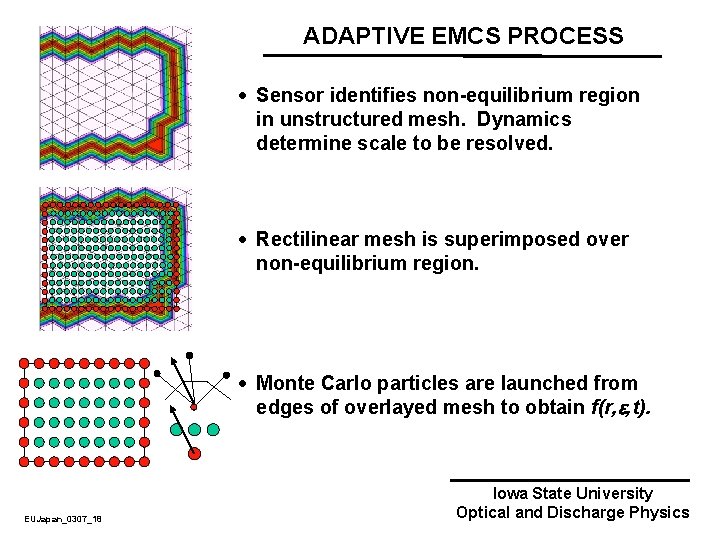
ADAPTIVE NONEQUILIBRIUM ELECTRON TRANSPORT · As the plasma evolves, regions will become “non-equilibrium” where electron transport is poorly described by fluid equations. · Large gradients in electric field. · Time rate of change of electric field is large compared to collision frequency. · To properly address transport, a kinetic approach is used for the electron energy distribution for position and time, f(r, , t). · These regions are addressed using an adaptive mesh technique with “sensors” that detect where are “non-equilibrium” conditions. · Use a kinetic Monte Carlo technique in those regions. EUJapan_0307_17 Iowa State University Optical and Discharge Physics

ADAPTIVE EMCS PROCESS · Sensor identifies non-equilibrium region in unstructured mesh. Dynamics determine scale to be resolved. · Rectilinear mesh is superimposed over non-equilibrium region. · Monte Carlo particles are launched from edges of overlayed mesh to obtain f(r, , t). EUJapan_0307_18 Iowa State University Optical and Discharge Physics
![BREAKDOWN IN HID LAMP THE IONIZATION FRONT Te Sources 10 e V e MCS BREAKDOWN IN HID LAMP: THE IONIZATION FRONT Te [Sources] 10 e. V [e] MCS](https://slidetodoc.com/presentation_image_h/2f0ddee152c78505f7f4ae69f3e949f9/image-19.jpg)
BREAKDOWN IN HID LAMP: THE IONIZATION FRONT Te [Sources] 10 e. V [e] MCS 1021 cm-3 s-1 1013 cm-3 Ionization front with steep gradients in [e] and electron impact sources moves across the gap. The EMCS sensor identifies the region of the ionization front and maps an adaptive MCS mesh onto it. MIN MAX Ar, 30 Torr, 2000 V, 100 ns EUJapan_0307_19 Animation Slide-GIF Iowa State University Optical and Discharge Physics
![BREAKDOWN IN HID LAMP THE IONIZATION FRONT Te 10 e V Sources e 1021 BREAKDOWN IN HID LAMP: THE IONIZATION FRONT Te 10 e. V [Sources] [e] 1021](https://slidetodoc.com/presentation_image_h/2f0ddee152c78505f7f4ae69f3e949f9/image-20.jpg)
BREAKDOWN IN HID LAMP: THE IONIZATION FRONT Te 10 e. V [Sources] [e] 1021 cm-3 s-1 1013 cm-3 MCS Ionization front with steep gradients in [e] and electron impact sources moves across the gap. The EMCS sensor identifies the region of the ionization front and maps an adaptive MCS mesh onto it. MIN MAX Ar, 30 Torr, 2000 V, 100 ns EUJapan_0307_19 A Animation Slide-AVI Iowa State University Optical and Discharge Physics

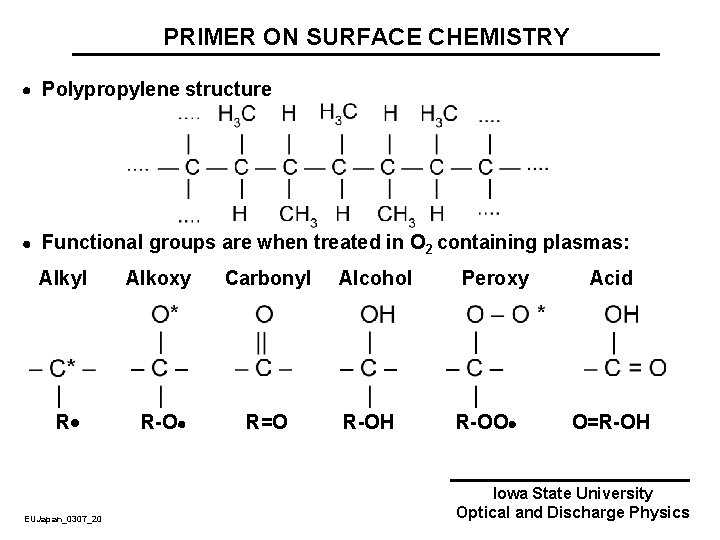
PRIMER ON SURFACE CHEMISTRY Polypropylene structure Functional groups are when treated in O 2 containing plasmas: Alkyl Alkoxy Carbonyl R R-O R=O EUJapan_0307_20 Alcohol R-OH Peroxy R-OO Acid O=R-OH Iowa State University Optical and Discharge Physics

PRIMER ON SURFACE CHEMISTRY · Ratio of O, OH, O 2 and O 3 fluxes determine surface composition. · Magnitude of fluxes and residence time determines importance of surface reactions. EUJapan_0307_21 Iowa State University Optical and Discharge Physics

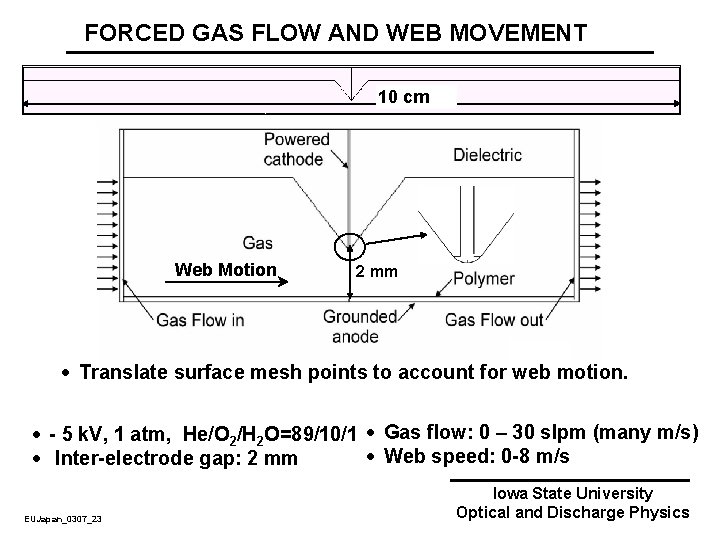
FORCED GAS FLOW AND WEB MOVEMENT Gas Flow · Polymer surfaces are continuously treated at web speeds of a few m/s with residence times in plasma of up to a few ms. · Non-air gas mixtures are often “forced flowed” through gap to customize radicals to surface. EUJapan_0307_22 Iowa State University Optical and Discharge Physics

FORCED GAS FLOW AND WEB MOVEMENT 10 cm Web Motion 2 mm · Translate surface mesh points to account for web motion. · - 5 k. V, 1 atm, He/O 2/H 2 O=89/10/1 · Gas flow: 0 – 30 slpm (many m/s) · Web speed: 0 -8 m/s · Inter-electrode gap: 2 mm EUJapan_0307_23 Iowa State University Optical and Discharge Physics

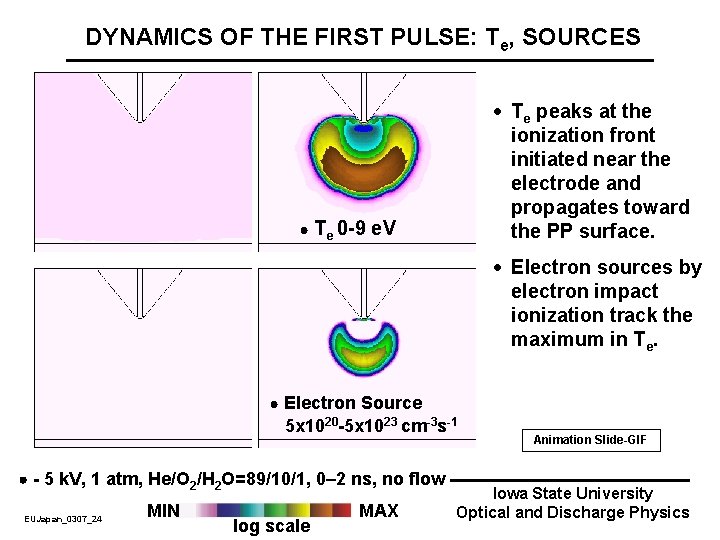
DYNAMICS OF THE FIRST PULSE: Te, SOURCES · Te peaks at the ionization front initiated near the electrode and propagates toward the PP surface. Te 0 -9 e. V · Electron sources by electron impact ionization track the maximum in Te. Electron Source 5 x 1020 -5 x 1023 cm-3 s-1 - 5 k. V, 1 atm, He/O 2/H 2 O=89/10/1, 0– 2 ns, no flow EUJapan_0307_24 MIN log scale MAX Animation Slide-GIF Iowa State University Optical and Discharge Physics
![REPETITIVELY PULSED DISCHARGES e Electron avalanche from the powered electrode The REPETITIVELY PULSED DISCHARGES – [e] · Electron avalanche from the powered electrode. · The](https://slidetodoc.com/presentation_image_h/2f0ddee152c78505f7f4ae69f3e949f9/image-26.jpg)
REPETITIVELY PULSED DISCHARGES – [e] · Electron avalanche from the powered electrode. · The pulse duration a few ns · Terminated by charging of dielectric. 1014 cm-3 0. 01 log scale 100 He/O 2/H 2 O=89/10/1, -5 k. V, 10 k. Hz, 1 atm EUJapan_0307_25 · Peak [e] of a few 1014 cm-3. Animation Slide-GIF Iowa State University Optical and Discharge Physics

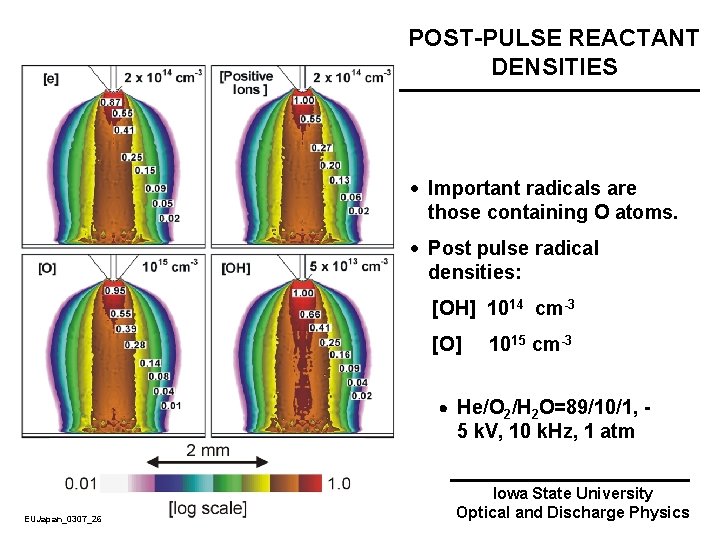
POST-PULSE REACTANT DENSITIES · Important radicals are those containing O atoms. · Post pulse radical densities: [OH] 1014 cm-3 [O] 1015 cm-3 He/O 2/H 2 O=89/10/1, - 5 k. V, 10 k. Hz, 1 atm EUJapan_0307_26 Iowa State University Optical and Discharge Physics

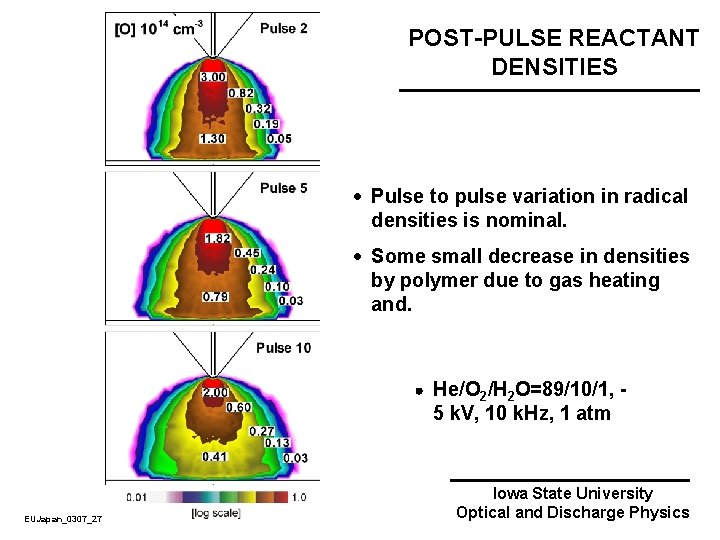
POST-PULSE REACTANT DENSITIES · Pulse to pulse variation in radical densities is nominal. · Some small decrease in densities by polymer due to gas heating and. He/O 2/H 2 O=89/10/1, - 5 k. V, 10 k. Hz, 1 atm EUJapan_0307_27 Iowa State University Optical and Discharge Physics

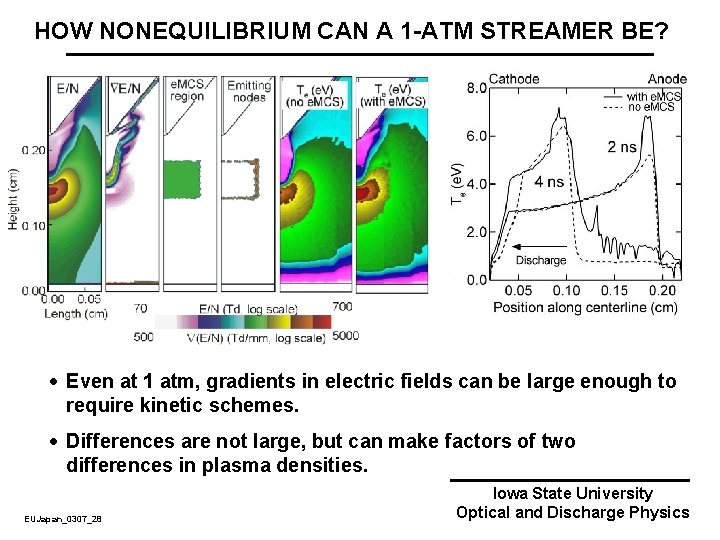
HOW NONEQUILIBRIUM CAN A 1 -ATM STREAMER BE? · Even at 1 atm, gradients in electric fields can be large enough to require kinetic schemes. · Differences are not large, but can make factors of two differences in plasma densities. EUJapan_0307_28 Iowa State University Optical and Discharge Physics

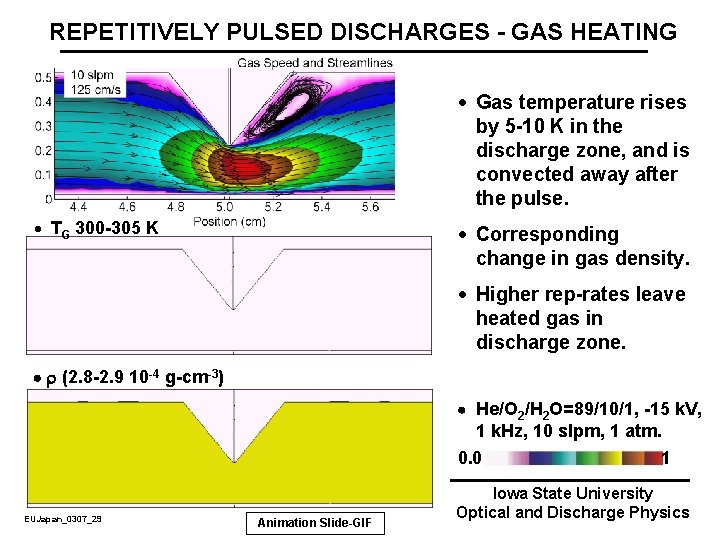
REPETITIVELY PULSED DISCHARGES - GAS HEATING · Gas temperature rises by 5 -10 K in the discharge zone, and is convected away after the pulse. · TG 300 -305 K · Corresponding change in gas density. · Higher rep-rates leave heated gas in discharge zone. r (2. 8 -2. 9 10 -4 g-cm-3) He/O 2/H 2 O=89/10/1, -15 k. V, 1 k. Hz, 10 slpm, 1 atm. 0. 0 1 EUJapan_0307_29 Animation Slide-GIF Iowa State University Optical and Discharge Physics
![O WITHOUT AND WITH FORCED GAS FLOW Without forced flow O 1015 cm3 [O] – WITHOUT AND WITH FORCED GAS FLOW Without forced flow [O] 1015 cm-3](https://slidetodoc.com/presentation_image_h/2f0ddee152c78505f7f4ae69f3e949f9/image-31.jpg)
[O] – WITHOUT AND WITH FORCED GAS FLOW Without forced flow [O] 1015 cm-3 With forced flow Flow · O atoms are generated by electron impact with every pulse. · Rapid reactions to form ozone (O + O 2 + M O 3 + M) deplete the O atoms within 10 s s of discharge pulse. · Little change in O atom distribution with and without gas flow. · He/O 2/H 2 O=89/10/1 · 10 k. Hz, 0 or 30 slpm, 1 atm. EUJapan_0307_30 0. 001 log scale Animation Slide-GIF 1 Iowa State University Optical and Discharge Physics
![O WITHOUT AND WITH FORCED GAS FLOW Without forced flow O 1015 cm3 [O] – WITHOUT AND WITH FORCED GAS FLOW Without forced flow [O] 1015 cm-3](https://slidetodoc.com/presentation_image_h/2f0ddee152c78505f7f4ae69f3e949f9/image-32.jpg)
[O] – WITHOUT AND WITH FORCED GAS FLOW Without forced flow [O] 1015 cm-3 With forced flow Flow · O atoms are generated by electron impact with every pulse. · Rapid reactions to form ozone (O + O 2 + M O 3 + M) deplete the O atoms within 10 s s of discharge pulse. · Little change in O atom distribution with and without gas flow. · He/O 2/H 2 O=89/10/1 · 10 k. Hz, 0 or 30 slpm, 1 atm. EUJapan_0307_30 A 0. 001 log scale Animation Slide-AVI 1 Iowa State University Optical and Discharge Physics
![O 3 WITHOUT AND WITH FORCED GAS FLOW Without forced flow O 3 [O 3] – WITHOUT AND WITH FORCED GAS FLOW Without forced flow [O 3]](https://slidetodoc.com/presentation_image_h/2f0ddee152c78505f7f4ae69f3e949f9/image-33.jpg)
[O 3] – WITHOUT AND WITH FORCED GAS FLOW Without forced flow [O 3] 3 x 1014 cm-3 With forced flow Flow · O 3 is relatively unreactive and accumulates from pulse to pulse. · Without forced flow, diffusion distributes O 3 up- and downstream. · With forced flow, a plume of O 3 extends downstream. · He/O 2/H 2 O=89/10/1 · 10 k. Hz, 0 or 30 slpm, 1 atm. EUJapan_0307_31 0. 001 log scale Animation Slide-GIF 1 Iowa State University Optical and Discharge Physics
![O 3 WITHOUT AND WITH FORCED GAS FLOW Without forced flow O 3 [O 3] – WITHOUT AND WITH FORCED GAS FLOW Without forced flow [O 3]](https://slidetodoc.com/presentation_image_h/2f0ddee152c78505f7f4ae69f3e949f9/image-34.jpg)
[O 3] – WITHOUT AND WITH FORCED GAS FLOW Without forced flow [O 3] 3 x 1014 cm-3 With forced flow Flow · O 3 is relatively unreactive and accumulates from pulse to pulse. · Without forced flow, diffusion distributes O 3 up- and downstream. · With forced flow, a plume of O 3 extends downstream. · He/O 2/H 2 O=89/10/1 · 10 k. Hz, 0 or 30 slpm, 1 atm. EUJapan_0307_31 A 0. 001 log scale Animation Slide-AVI 1 Iowa State University Optical and Discharge Physics
![OH WITHOUT AND WITH FORCED GAS FLOW Without forced flow OH 1014 cm3 [OH] – WITHOUT AND WITH FORCED GAS FLOW Without forced flow [OH] 1014 cm-3](https://slidetodoc.com/presentation_image_h/2f0ddee152c78505f7f4ae69f3e949f9/image-35.jpg)
[OH] – WITHOUT AND WITH FORCED GAS FLOW Without forced flow [OH] 1014 cm-3 With forced flow Flow · OH has an intermediate reactivity between O and O 3. · Some modest accumulation occurs with a small plume downstream with forced flow. · He/O 2/H 2 O=89/10/1 · 10 k. Hz, 0 or 30 slpm, 1 atm. Animation Slide-GIF EUJapan_0307_32 0. 001 log scale 1 Iowa State University Optical and Discharge Physics
![OH WITHOUT AND WITH FORCED GAS FLOW Without forced flow OH 1014 cm3 [OH] – WITHOUT AND WITH FORCED GAS FLOW Without forced flow [OH] 1014 cm-3](https://slidetodoc.com/presentation_image_h/2f0ddee152c78505f7f4ae69f3e949f9/image-36.jpg)
[OH] – WITHOUT AND WITH FORCED GAS FLOW Without forced flow [OH] 1014 cm-3 With forced flow Flow · OH has an intermediate reactivity between O and O 3. · Some modest accumulation occurs with a small plume downstream with forced flow. · He/O 2/H 2 O=89/10/1 · 10 k. Hz, 0 or 30 slpm, 1 atm. Animation Slide-AVI EUJapan_0307_32 A 0. 001 log scale 1 Iowa State University Optical and Discharge Physics

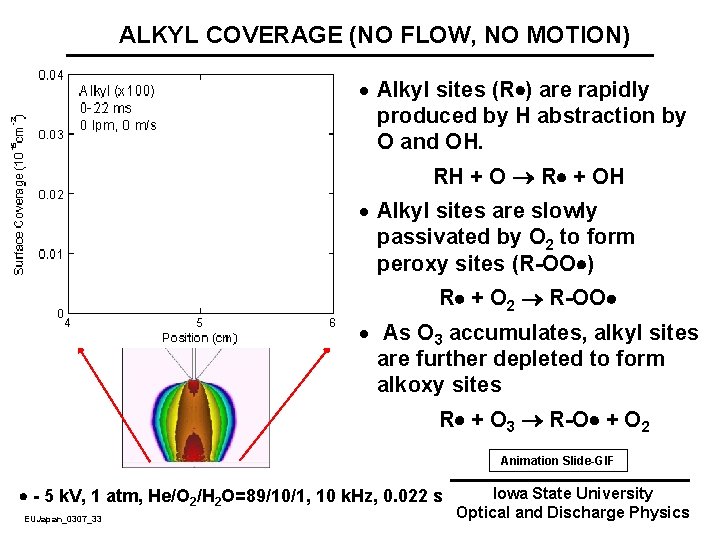
ALKYL COVERAGE (NO FLOW, NO MOTION) · Alkyl sites (R ) are rapidly produced by H abstraction by O and OH. RH + O R + OH · Alkyl sites are slowly passivated by O 2 to form peroxy sites (R-OO ) R + O 2 R-OO · As O 3 accumulates, alkyl sites are further depleted to form alkoxy sites R + O 3 R-O + O 2 Animation Slide-GIF - 5 k. V, 1 atm, He/O 2/H 2 O=89/10/1, 10 k. Hz, 0. 022 s EUJapan_0307_33 Iowa State University Optical and Discharge Physics

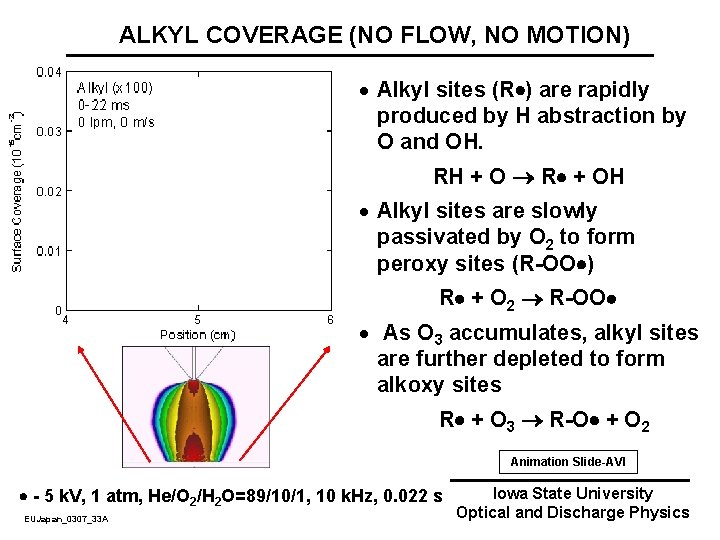
ALKYL COVERAGE (NO FLOW, NO MOTION) · Alkyl sites (R ) are rapidly produced by H abstraction by O and OH. RH + O R + OH · Alkyl sites are slowly passivated by O 2 to form peroxy sites (R-OO ) R + O 2 R-OO · As O 3 accumulates, alkyl sites are further depleted to form alkoxy sites R + O 3 R-O + O 2 Animation Slide-AVI - 5 k. V, 1 atm, He/O 2/H 2 O=89/10/1, 10 k. Hz, 0. 022 s EUJapan_0307_33 A Iowa State University Optical and Discharge Physics

PEROXY COVERAGE (NO FLOW, NO MOTION) · Peroxy sites are produced following each pulse by O 2 reactions with alkyl sites R + O 2 R-OO · Peroxy sites are relatively unreactive and only slowly are depleted by H abstraction R-OO • + RH R-OOH + R • · As a result, peroxy sites accumulate pulse to pulse. Animation Slide-GIF - 5 k. V, 1 atm, He/O 2/H 2 O=89/10/1, 10 k. Hz, 0. 022 s EUJapan_0307_34 Iowa State University Optical and Discharge Physics

PEROXY COVERAGE (NO FLOW, NO MOTION) · Peroxy sites are produced following each pulse by O 2 reactions with alkyl sites R + O 2 R-OO · Peroxy sites are relatively unreactive and only slowly are depleted by H abstraction R-OO • + RH R-OOH + R • · As a result, peroxy sites accumulate pulse to pulse. Animation Slide-AVI - 5 k. V, 1 atm, He/O 2/H 2 O=89/10/1, 10 k. Hz, 0. 022 s EUJapan_0307_34 A Iowa State University Optical and Discharge Physics

ALKYL, PEROXY COVERAGE WITH FLOW · Alkyl (R ) sites are still rapidly produced and passivated with each pulse. · The plume of OH radicals downstream produce a tail of R sites. · Peroxy sites accumulate downstream. 1 atm, He/O 2/H 2 O=89/10/1, 10 k. Hz, 0. 022 s, 30 slpm Flow EUJapan_0307_35 Animation Slide-GIF Iowa State University Optical and Discharge Physics

ALKYL, PEROXY COVERAGE WITH FLOW · Alkyl (R ) sites are still rapidly produced and passivated with each pulse. · The plume of OH radicals downstream produce a tail of R sites. · Peroxy sites accumulate downstream. 1 atm, He/O 2/H 2 O=89/10/1, 10 k. Hz, 0. 022 s, 30 slpm Flow EUJapan_0307_35 A Animation Slide-AVI Iowa State University Optical and Discharge Physics

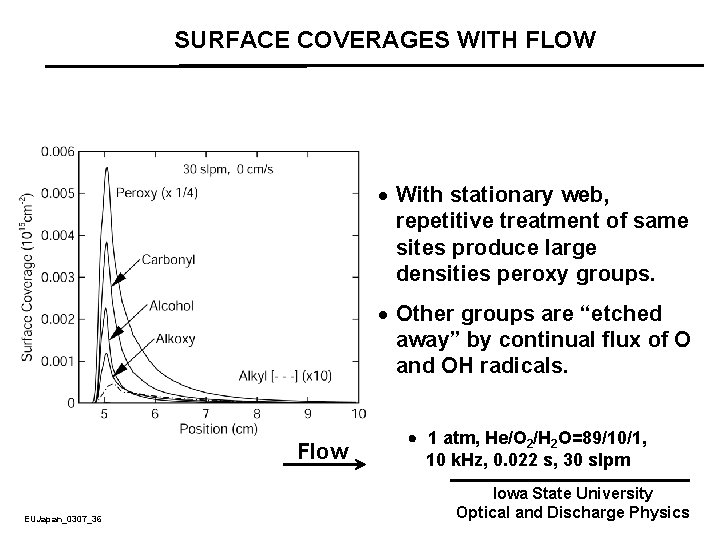
SURFACE COVERAGES WITH FLOW · With stationary web, repetitive treatment of same sites produce large densities peroxy groups. · Other groups are “etched away” by continual flux of O and OH radicals. Flow EUJapan_0307_36 1 atm, He/O 2/H 2 O=89/10/1, 10 k. Hz, 0. 022 s, 30 slpm Iowa State University Optical and Discharge Physics

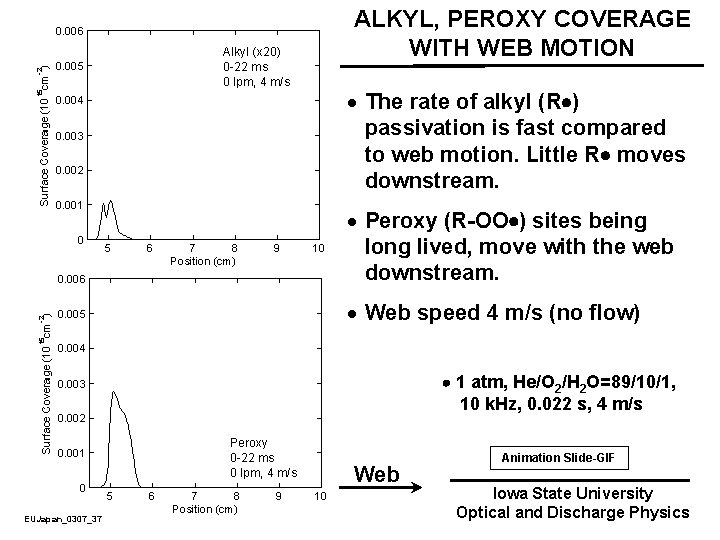
ALKYL, PEROXY COVERAGE WITH WEB MOTION · The rate of alkyl (R ) passivation is fast compared to web motion. Little R moves downstream. · Peroxy (R-OO ) sites being long lived, move with the web downstream. · Web speed 4 m/s (no flow) 1 atm, He/O 2/H 2 O=89/10/1, 10 k. Hz, 0. 022 s, 4 m/s Web EUJapan_0307_37 Animation Slide-GIF Iowa State University Optical and Discharge Physics

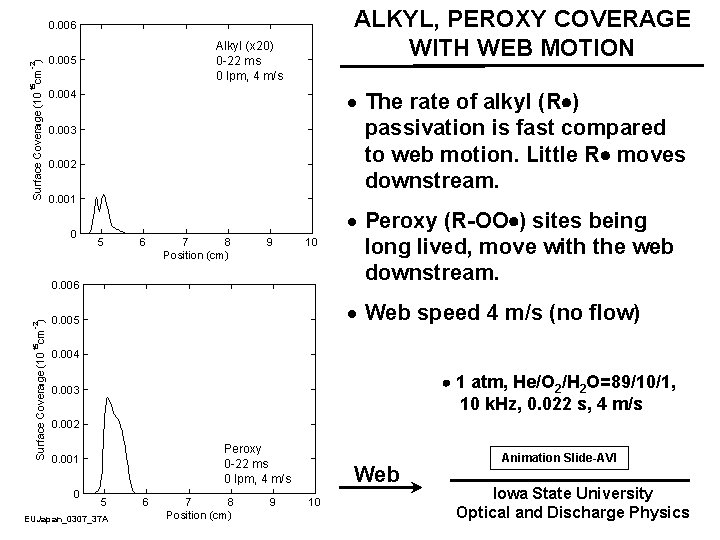
ALKYL, PEROXY COVERAGE WITH WEB MOTION · The rate of alkyl (R ) passivation is fast compared to web motion. Little R moves downstream. · Peroxy (R-OO ) sites being long lived, move with the web downstream. · Web speed 4 m/s (no flow) 1 atm, He/O 2/H 2 O=89/10/1, 10 k. Hz, 0. 022 s, 4 m/s Web EUJapan_0307_37 A Animation Slide-AVI Iowa State University Optical and Discharge Physics

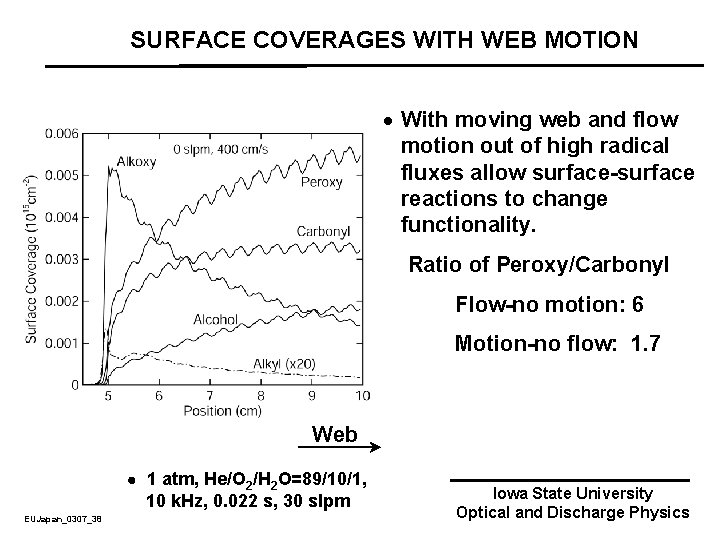
SURFACE COVERAGES WITH WEB MOTION · With moving web and flow motion out of high radical fluxes allow surface-surface reactions to change functionality. Ratio of Peroxy/Carbonyl Flow-no motion: 6 Motion-no flow: 1. 7 Web 1 atm, He/O 2/H 2 O=89/10/1, 10 k. Hz, 0. 022 s, 30 slpm EUJapan_0307_38 Iowa State University Optical and Discharge Physics

SURFACE COVERAGES WITH FLOW AND WEB MOTION · With flow and motion in same direction, sites move “under plume” of radicals. · Effectively larger fluence increases radical processes. Ratio of Peroxy/Carbonyl Flow-no motion: 6 Motion-no flow: 1. 7 Motion-flow Flow Web 1 atm, He/O 2/H 2 O=89/10/1, 10 k. Hz, 0. 022 s, 30 slpm EUJapan_0307_39 2. 8 · Choice of flow and motion provides control over functional groups. Iowa State University Optical and Discharge Physics

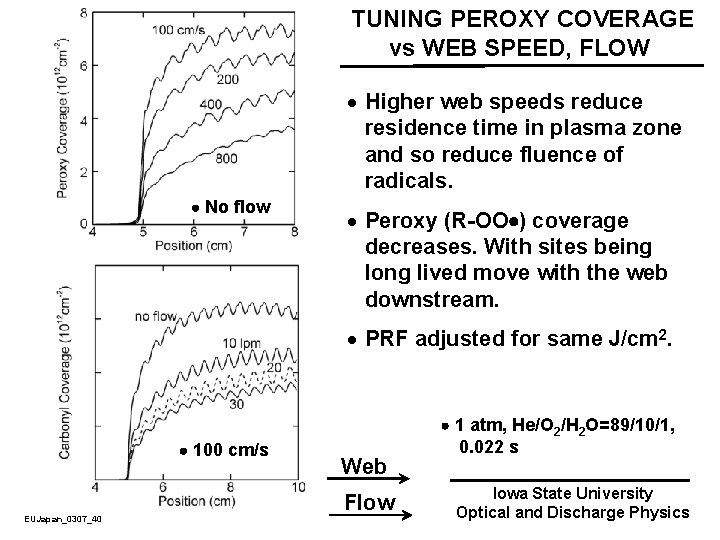
TUNING PEROXY COVERAGE vs WEB SPEED, FLOW · Higher web speeds reduce residence time in plasma zone and so reduce fluence of radicals. No flow · Peroxy (R-OO ) coverage decreases. With sites being long lived move with the web downstream. · PRF adjusted for same J/cm 2. 100 cm/s EUJapan_0307_40 Web Flow 1 atm, He/O 2/H 2 O=89/10/1, 0. 022 s Iowa State University Optical and Discharge Physics

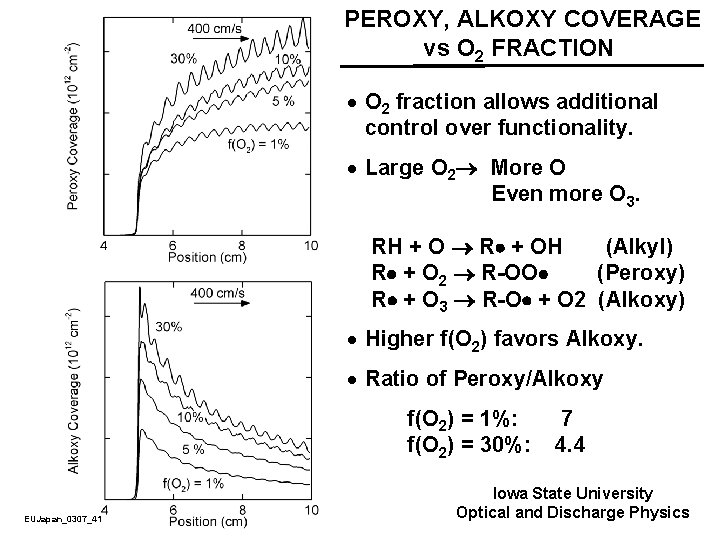
PEROXY, ALKOXY COVERAGE vs O 2 FRACTION · O 2 fraction allows additional control over functionality. · Large O 2 More O Even more O 3. RH + O R + OH (Alkyl) R + O 2 R-OO (Peroxy) R + O 3 R-O + O 2 (Alkoxy) · Higher f(O 2) favors Alkoxy. · Ratio of Peroxy/Alkoxy f(O 2) = 1%: f(O 2) = 30%: EUJapan_0307_41 7 4. 4 Iowa State University Optical and Discharge Physics

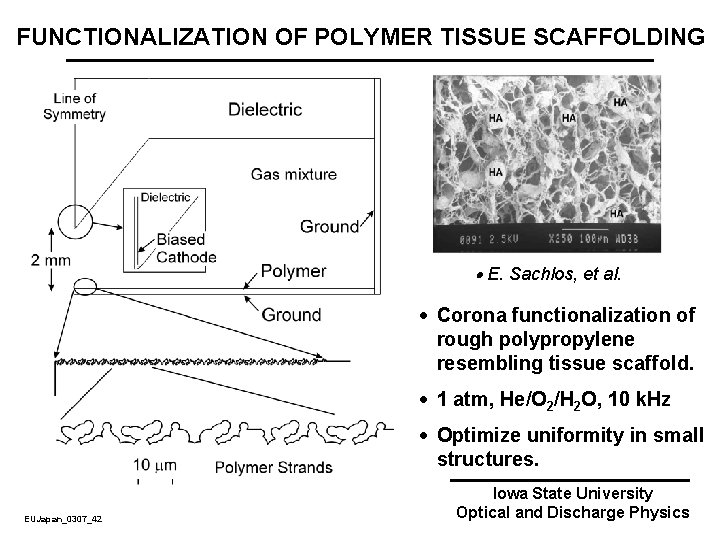
FUNCTIONALIZATION OF POLYMER TISSUE SCAFFOLDING E. Sachlos, et al. · Corona functionalization of rough polypropylene resembling tissue scaffold. · 1 atm, He/O 2/H 2 O, 10 k. Hz · Optimize uniformity in small structures. EUJapan_0307_42 Iowa State University Optical and Discharge Physics

OPTIMIZE CHEMISTRY, UNIFORMITY WITH GAS MIXTURE · Balance of peroxy (R-OO ), alkoxy (R-O ) and alcohol (R-OH) groups can be controlled by composition of fluxes. · Example: He/O 2/H 2 O e + O 2 O + e e + H 2 O H + OH + e O + O 2 + M O 3 + M · Large f(O 2), small f(H 2 O): Small OH fluxes, large O 3 fluxes Small f(O 2), large f(H 2 O): Large OH fluxes, small O 3 fluxes · Impact on polypropylene surface chemistry RH + OH R + O 2 R + O 3 R + OH EUJapan_0307_43 R + OH R + H 2 O R-O + O 2 R-OH (slow rate) (fast rate) (slow rate but a lot of O 2) (fast rate) Iowa State University Optical and Discharge Physics

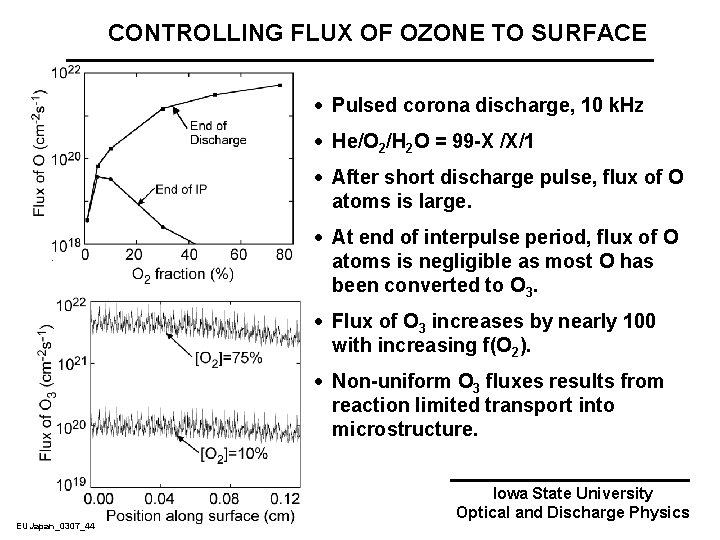
CONTROLLING FLUX OF OZONE TO SURFACE · Pulsed corona discharge, 10 k. Hz · He/O 2/H 2 O = 99 -X /X/1 · After short discharge pulse, flux of O atoms is large. · At end of interpulse period, flux of O atoms is negligible as most O has been converted to O 3. · Flux of O 3 increases by nearly 100 with increasing f(O 2). · Non-uniform O 3 fluxes results from reaction limited transport into microstructure. EUJapan_0307_44 Iowa State University Optical and Discharge Physics

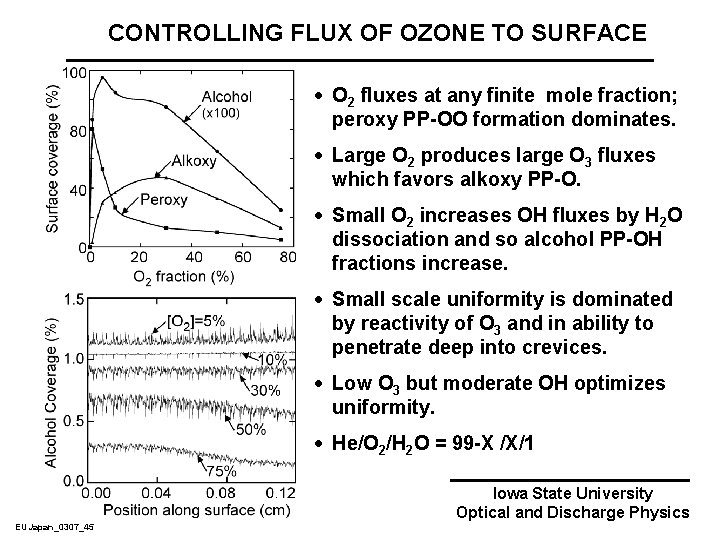
CONTROLLING FLUX OF OZONE TO SURFACE · O 2 fluxes at any finite mole fraction; peroxy PP-OO formation dominates. · Large O 2 produces large O 3 fluxes which favors alkoxy PP-O. · Small O 2 increases OH fluxes by H 2 O dissociation and so alcohol PP-OH fractions increase. · Small scale uniformity is dominated by reactivity of O 3 and in ability to penetrate deep into crevices. · Low O 3 but moderate OH optimizes uniformity. · He/O 2/H 2 O = 99 -X /X/1 EUJapan_0307_45 Iowa State University Optical and Discharge Physics

CONCLUDING REMARKS · Functionalization of low value materials for high value applications has great promise. · Even at atmospheric pressure, fluxes of radicals can be tailored to provide desired functionality. · Modeling of industrial processes requires some care to details: flow, motion of web, repetition rates, photon-surface-interactions. EUJapan_0307_45 Iowa State University Optical and Discharge Physics