Plasma Etch Subprocesses Reactant Generation process RF Electrode

Plasma Etch Sub-processes
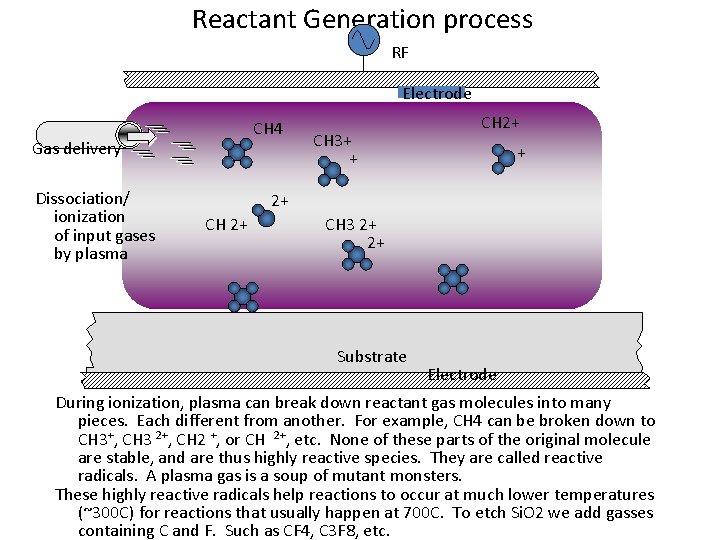
Reactant Generation process RF Electrode CH 4 Gas delivery Dissociation/ ionization of input gases by plasma CH 3+ + CH 2+ + 2+ CH 3 2+ 2+ Substrate Electrode During ionization, plasma can break down reactant gas molecules into many pieces. Each different from another. For example, CH 4 can be broken down to CH 3+, CH 3 2+, CH 2 +, or CH 2+, etc. None of these parts of the original molecule are stable, and are thus highly reactive species. They are called reactive radicals. A plasma gas is a soup of mutant monsters. These highly reactive radicals help reactions to occur at much lower temperatures (~300 C) for reactions that usually happen at 700 C. To etch Si. O 2 we add gasses containing C and F. Such as CF 4, C 3 F 8, etc.
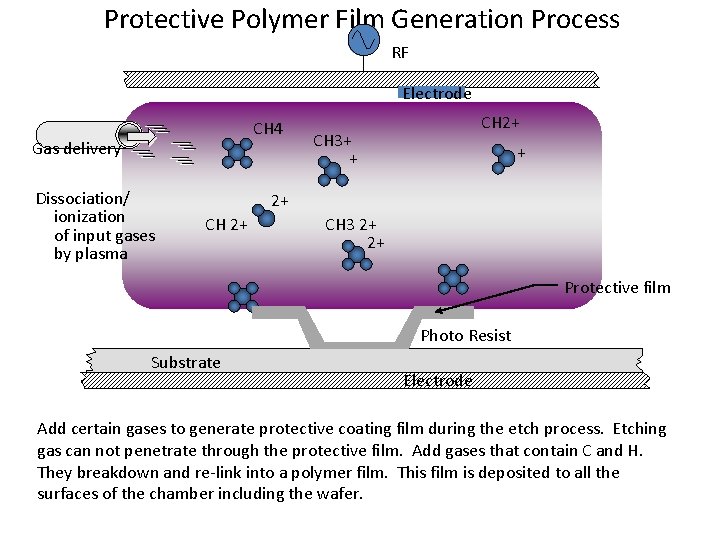
Protective Polymer Film Generation Process RF Electrode CH 4 Gas delivery Dissociation/ ionization of input gases by plasma CH 2+ CH 3+ + + 2+ CH 3 2+ 2+ Protective film Photo Resist Substrate Electrode Add certain gases to generate protective coating film during the etch process. Etching gas can not penetrate through the protective film. Add gases that contain C and H. They breakdown and re-link into a polymer film. This film is deposited to all the surfaces of the chamber including the wafer.
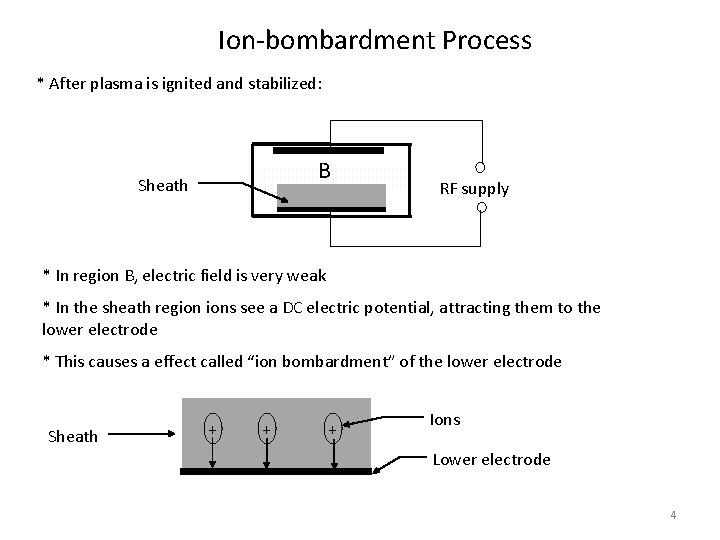
Ion-bombardment Process * After plasma is ignited and stabilized: B Sheath RF supply * In region B, electric field is very weak * In the sheath region ions see a DC electric potential, attracting them to the lower electrode * This causes a effect called “ion bombardment” of the lower electrode Sheath + + + Ions Lower electrode 4
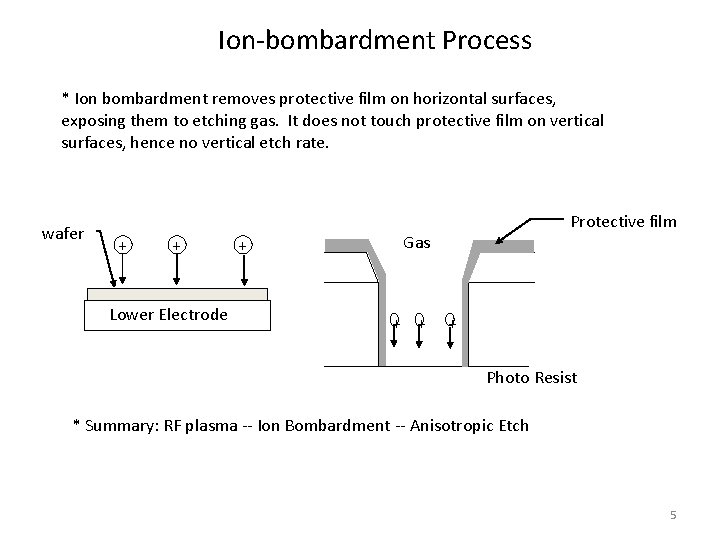
Ion-bombardment Process * Ion bombardment removes protective film on horizontal surfaces, exposing them to etching gas. It does not touch protective film on vertical surfaces, hence no vertical etch rate. wafer + + Lower Electrode + Protective film Gas + + + Photo Resist * Summary: RF plasma -- Ion Bombardment -- Anisotropic Etch 5
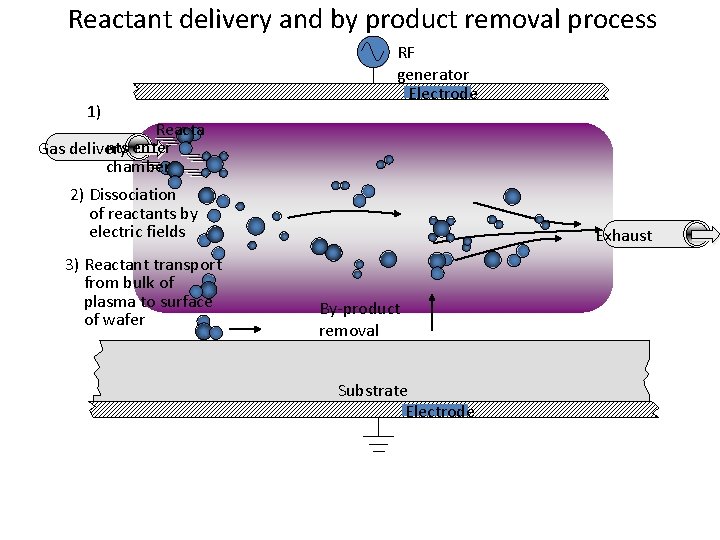
Reactant delivery and by product removal process 1) RF generator Electrode Reacta nts enter Gas delivery chamber 2) Dissociation of reactants by electric fields 3) Reactant transport from bulk of plasma to surface of wafer Exhaust By-product removal Substrate Electrode

The slowest sub-process bottlenecks the overall process. It thus determines the reaction rate. It becomes the dominate sub-process– the overall process behaves like it.
- Slides: 7