PlantMicrobe Interactions v Plantmicrobe interactions diverse from the

Plant-Microbe Interactions v Plant-microbe interactions diverse – from the plant perspective: • Negative – e. g. parasitic/pathogenic • Neutral • Positive – symbiotic v This lecture important positive interactions with respect to plant abundance and distribution – related to plant nutrient and water supply: Decomposition Mycorrhizae N 2 fixation Rhizosphere the role of this interaction in the N cycle
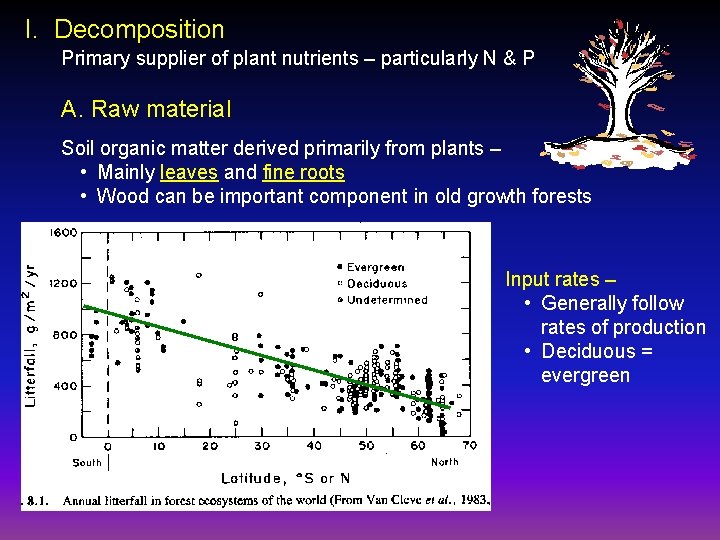
I. Decomposition Primary supplier of plant nutrients – particularly N & P A. Raw material Soil organic matter derived primarily from plants – • Mainly leaves and fine roots • Wood can be important component in old growth forests Input rates – • Generally follow rates of production • Deciduous = evergreen
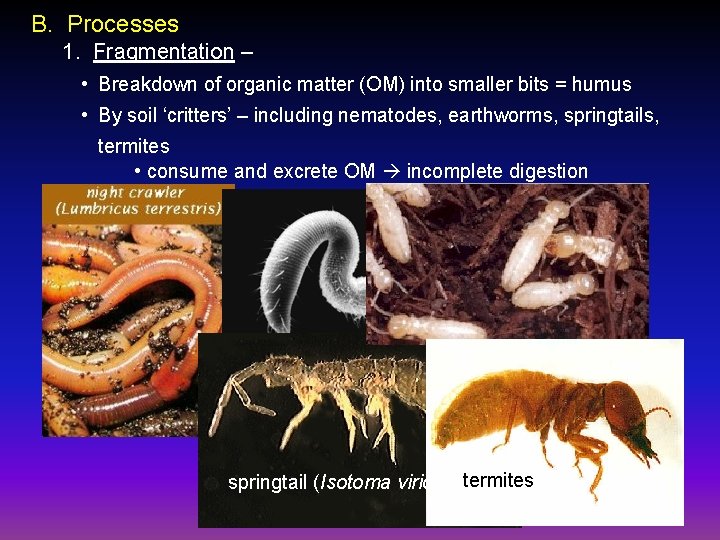
B. Processes 1. Fragmentation – • Breakdown of organic matter (OM) into smaller bits = humus • By soil ‘critters’ – including nematodes, earthworms, springtails, termites • consume and excrete OM incomplete digestion nematode springtail (Isotoma viridis) termites
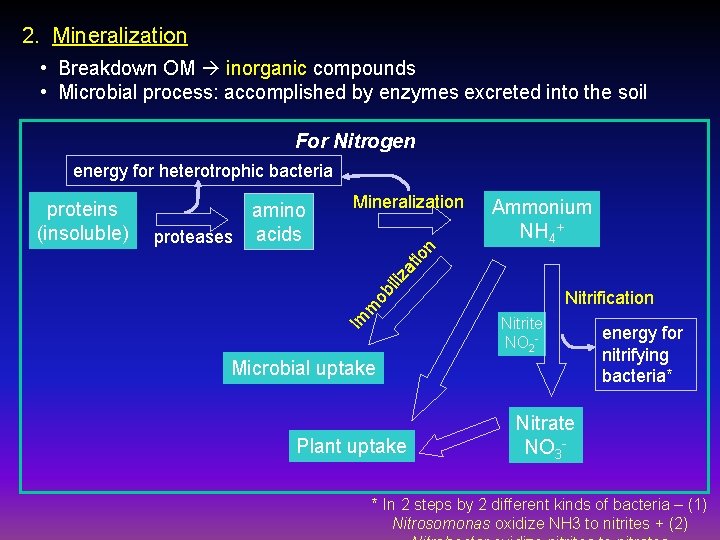
2. Mineralization • Breakdown OM inorganic compounds • Microbial process: accomplished by enzymes excreted into the soil For Nitrogen energy for heterotrophic bacteria Ammonium NH 4+ io n Mineralization iliz at amino proteases acids Nitrification ob m Im proteins (insoluble) Nitrite NO 2 - Microbial uptake Plant uptake energy for nitrifying bacteria* Nitrate NO 3 - * In 2 steps by 2 different kinds of bacteria – (1) Nitrosomonas oxidize NH 3 to nitrites + (2)

C. N uptake by plants – Chemical form taken up can vary 1) Nitrate (NO proteins 3) mineralization NH 4+ • Preferred by most plants, easier to take up • Even though requires conversion to NH 4+ before be used lots of energy • vs. taking up & storing NH 4+ problematic • More strongly bound to soil particles • Acidifies the soil • Not easily stored NO 3 - plant uptake 2) Ammonium (NH 4+ ) – • Used directly by plants in soils with low nitrification rates (e. g. wet soils)

3) Some plants can take up small amino acids (e. g. glycine) • Circumvents the need for N mineralization • Facilitated by mycorrhizae mineralization proteins amino acids immobilization NH 4+ nitrification microbial uptake Direct uptake NO 3 plant uptake
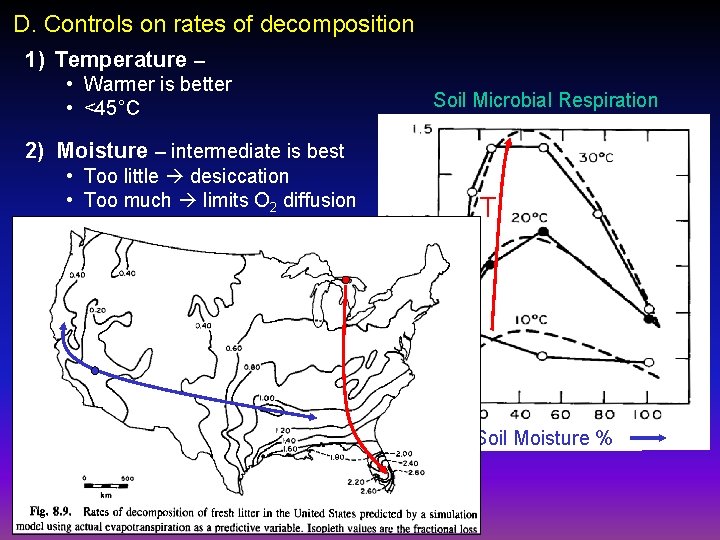
D. Controls on rates of decomposition 1) Temperature – • Warmer is better • <45°C Soil Microbial Respiration 2) Moisture – intermediate is best • Too little desiccation • Too much limits O 2 diffusion T Soil Moisture %

3) Plant factors – Litter quality a) Litter C: N ratio (= N concentration) • If C relative to N high N limits microbial growth • Immobilization favored • N to plants Decomposition rate as fn(lignin, N) Deciduous forest spp b) Plant structural material • Lignin – complex polymer, cell walls • Confers strength with flexibility – e. g. oak leaves • Relatively recalcitrant • High conc. lowers decomposition

c) Plant secondary compounds • Anti-herbivore/microbial • Common are phenolics – e. g. tannins – Aromatic ring + hydroxyl group, other compounds OH R • Control decomposition by: Bind to enzymes, blocking active sites lower mineralization N compounds bind to phenolics greater immobilization by soil Phenolics C source for microbes greater immobilization by microbes Consequence of controlling soil OM chemistry and microclimate … Plants important factor controlling spatial variation in nutrient cycling

II. Mycorrhizae A. Symbiotic relationship between plants (roots) & soil fungi • • Plant provides fungus with energy (C) Fungus enhances soil resource uptake Widespread – • • • Occurs ~80% angiosperm spp All gymnosperms Sometimes an obligate relationship
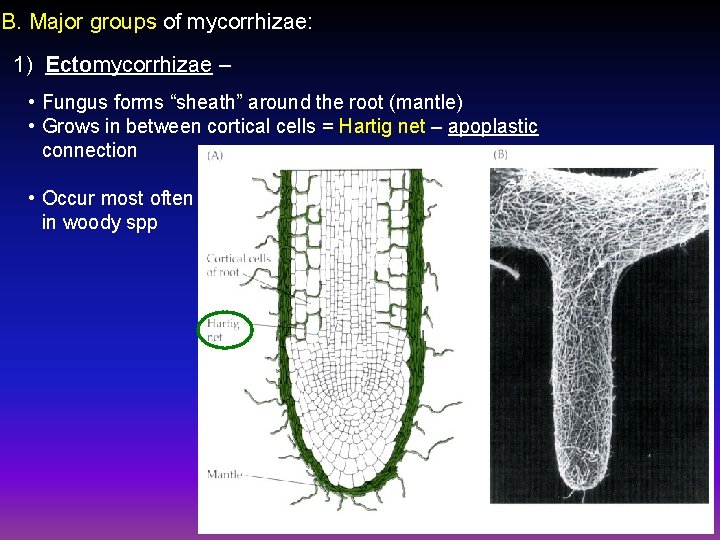
B. Major groups of mycorrhizae: 1) Ectomycorrhizae – • Fungus forms “sheath” around the root (mantle) • Grows in between cortical cells = Hartig net – apoplastic connection • Occur most often in woody spp
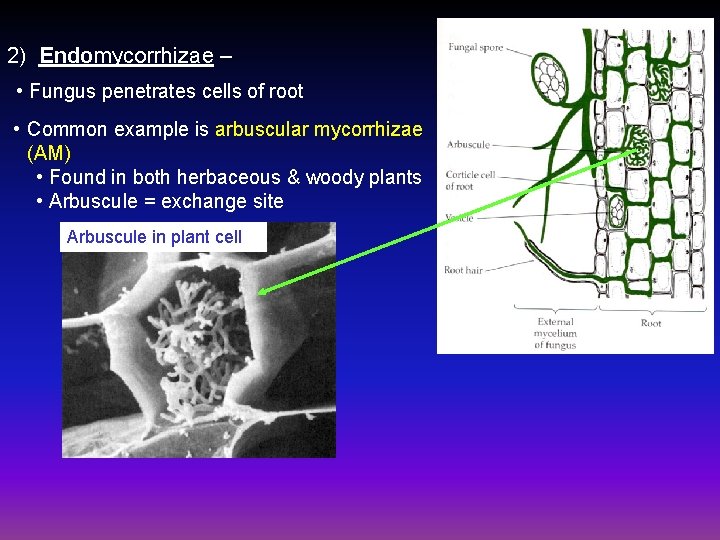
2) Endomycorrhizae – • Fungus penetrates cells of root • Common example is arbuscular mycorrhizae (AM) • Found in both herbaceous & woody plants • Arbuscule = exchange site Arbuscule in plant cell

C. Function of mycorrhizae: 1) Roles in plant-soil interface – a) Increase surface area & reach for absorption of soil water & nutrients b) Increase mobility and uptake of soil P c) Provides plant with access to organic N d) Protect roots from toxic heavy metals e) Protect roots from pathogens 2) Effect of soil nutrient levels on mycorrhizae • Intermediate soil P concentrations favorable • Extremely low P – poor fungal infection • Hi P – plants suppress fungal growth – taking up P directly • N saturation

III. N 2 Fixation N 2 abundant – chemically inert N 2 must be fixed = converted into chemically usable form • Lightning • High temperature or pressure (humans) • Biologically fixed v Nitrogenase – enzyme catalyzes N 2 NH 3 v Expensive process – ATP, Molybdenum v Anaerobic – requires special structures

A. Occurs only in prokaryotes: • Bacteria (e. g. Rhizobium, Frankia) • Cyanobacteria (e. g. Nostoc, Anabaena) Ø Ø Ø Free-living in soil/water – heterocysts Symbiotic with plants – root nodules Loose association with plants Anabaena with heterocysts Symbiosis with plants – Mutualism • Prokaryote receives carbohydrates • Plant may allocate up to 30% of its C to the symbiont • Plant provides anaerobic site – nodules • Plant receives N

Examples of plant–N 2 -fixing symbiotic systems – 1) Legumes (Fabaceae) • Widespread • bacteria = e. g. , Rhizobium spp. • Those with N 2 -fixing symbionts form root “nodules” – anaerobic sites that “house” bacteria alpine clover soybean root

Problem of O 2 toxicity – • Symbionts regulate O 2 in the nodule with leghemoglobin • Different part synthesized by the bacteria and legume Cross-section of nodules of soybean nodules

2) Non-legume symbiotic plants – • “Actinorhizal”= associated with actinomycetes (N 2 -fixing bacteria) • genus Frankia • Usually woody species – e. g. Alders, Ceanothus • Bacteria in root or small vesicles Ceanothus velutinus - snowbrush Ceanothus roots, with Frankia vesicles Buffaloberry (Shepherdia argentea) - actinorhizal shrub (Arizona)

B. Ecological importance of N 2 fixation 1) Important in “young” ecosystems – • Young soils low in organic matter, N

2) Plant-level responses to increased soil N conc: Some plants (facultative N-fixers) respond to soil N concentration • Plant shifts to direct N uptake • N fixation • Number of nodules decreases

3) Competition: N fixers-plant community interactions N 2 -fixing plants higher P, light, Mo, and Fe requirements Poor competitors • Competitive exclusion less earlier in succession • Though - N 2 fixers in “mature” ecosystems Example N-fixing plants important in early stages of succession: • Lupines, alders, clovers, Dryas
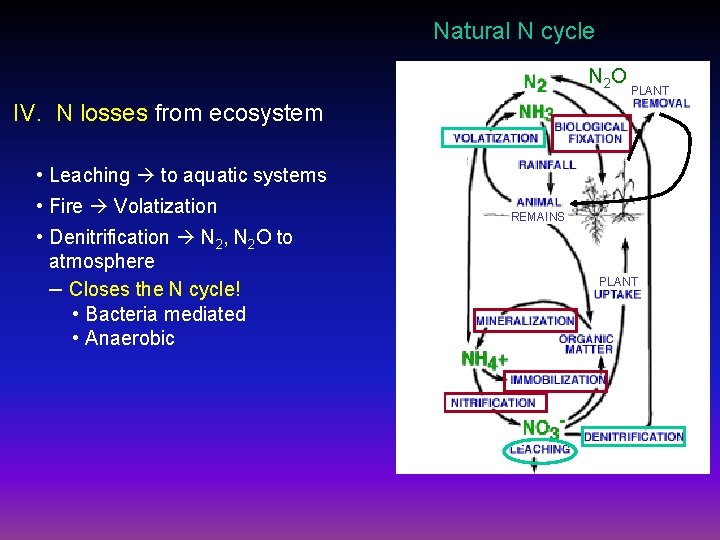
Natural N cycle N 2 O PLANT IV. N losses from ecosystem • Leaching to aquatic systems • Fire Volatization • Denitrification N 2, N 2 O to atmosphere – Closes the N cycle! • Bacteria mediated • Anaerobic REMAINS PLANT
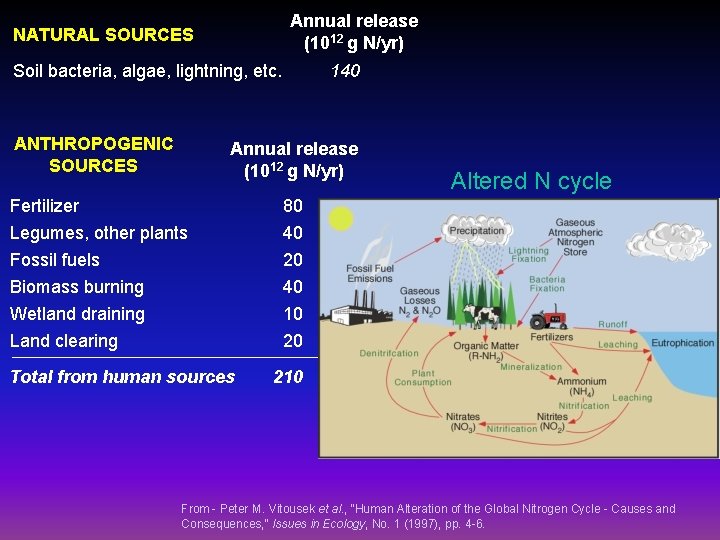
Annual release (1012 g N/yr) NATURAL SOURCES Soil bacteria, algae, lightning, etc. ANTHROPOGENIC SOURCES 140 Annual release (1012 g N/yr) Fertilizer Legumes, other plants 80 40 Fossil fuels Biomass burning Wetland draining 20 40 10 Land clearing 20 Total from human sources Altered N cycle 210 From - Peter M. Vitousek et al. , "Human Alteration of the Global Nitrogen Cycle - Causes and Consequences, " Issues in Ecology, No. 1 (1997), pp. 4 -6.
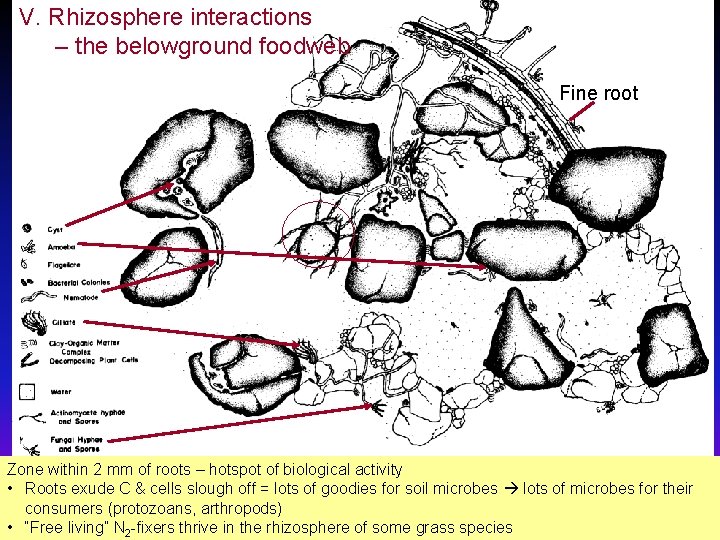
V. Rhizosphere interactions – the belowground foodweb Fine root Zone within 2 mm of roots – hotspot of biological activity • Roots exude C & cells slough off = lots of goodies for soil microbes lots of microbes for their consumers (protozoans, arthropods) • “Free living” N 2 -fixers thrive in the rhizosphere of some grass species

Summary • Plant–microbial interactions play key roles in plant nutrient dynamics Ø Decomposition – Ø mineralization, nitrification … Ø immobilization, denitrification … Ø Rhizosphere – soil foodweb Ø Mycorrhizae – plant-fungi symbiosis Ø N fixation – plant-bacteria symbiosis • Highly adapted root morphology and physiology to accommodate these interactions • N cycle, for example, significantly altered by human activities
- Slides: 25