Plant Nutrition Basic Plant Composition Dry Weight 96

Plant Nutrition

Basic Plant Composition Dry Weight: 96% Organic: mostly carbohydrates – starch and cellulose l 4% Inorganic: non – carbon containing nutrients Essential Elements: necessary to grow – 17 types - determined by hydroponic experimentation

Macronutrients l l 9 of 17 essentials – needed in large amounts C O H N P S K Ca Mg NUTRIENT FUNCTION Carbon, Hydrogen, Oxygen Carbs, Proteins, lipids Nitrogen Nucleic Acids Proteins, Chlorophylls Potassium Cofactor, stomata control Calcium Cell stability and response Magnesium Central atom of chlorophyll Phosphorous ATP, Phospholipids, Nucleic Acids Sulfur Proteins

Micronutrients: l 8 of 17 essentials – need in very small amounts: Cl Fe Mn B Zn Cu Ni Mo NUTRIENT FUNCTION Chlorine Hydrolysis of water Iron In ETC Manganese Hydrolysis of water, Amino Acid Synthesis Boron Chlorophyll synthesis Zinc Chlorophyll synthesis Copper Lignin Synthesis Nickel Nitrogen Metabolism Molybdenum Symbiosis with N 2 Fixing Bacteria
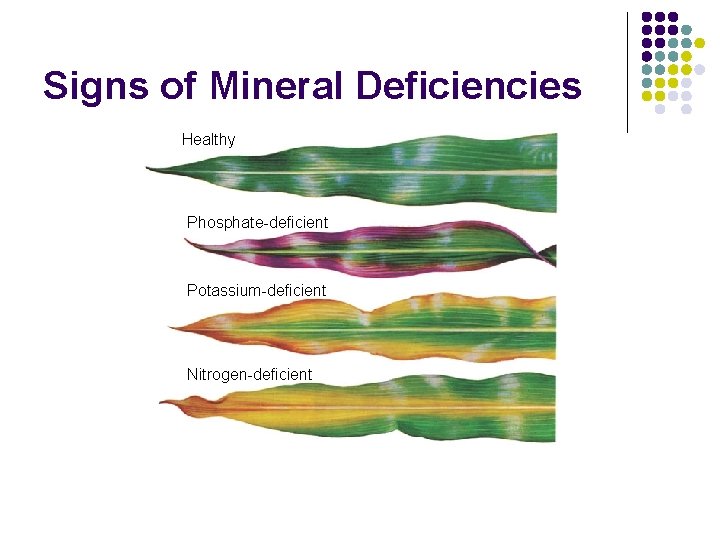
Signs of Mineral Deficiencies Healthy Phosphate-deficient Potassium-deficient Nitrogen-deficient

Role of Soil in Plant Nutrition Soil Formation: Physical Weathering Freeze/thaw cycles - water seeps into small cracks in rocks – freezes – expands – makes crack bigger - rocks break down Erosion - break down by wind and water Root Expansion - small roots in small crevices expand by lateral growth and break the rock open

Chemical Weathering: Acids dissolved in water Acids released by organisms Type of rock affects the quality of the soil Ex: Limestone (shale) – calcium carbonate – neutralization of acids - can lead to a natural buffering effect on acid rain

Soil Composition: Topsoil – rock particles, living organisms and humus (remains of partially decayed organic material – Sphagnum moss) Soil Layers – stratification - Top layer - top soil O, A, B, C, and E.

Soil Texture - based on the particles in the soil - coarse – sandy – good for drainage – too much results in an inability to retain nutrients - medium – silt - fine – clay - negatively charged – attract water molecules – good for retaining water and minerals that forms a soil solution that the plant roots extract if the water and minerals are not bound to the clay particles too tightly – anions (nitrate, phosphate, sulfate) are not bound tightly – may leach (wash away) in the presence of heavy rain – cations (potassium, calcium, magnesium) are bound tightly to the clay and are harder to extract – too much clay in the soil results in an inability to have sufficient oxygen for root metabolism - loamy –mixture of sand (40%), silt (40%) and clay (20%)

Role of living organisms - decomposers: fungi, bacteria, worms (annelids, nematodes) – break down organic matter – larger organism help aerate (make air spaces) the soil - plant roots - hold soil in place - cation exchange – release organic acids which put H+ in the soil solution, the H+ replace the positive ions (Mg, K, Ca) bound to the clay particles making them available for root uptake
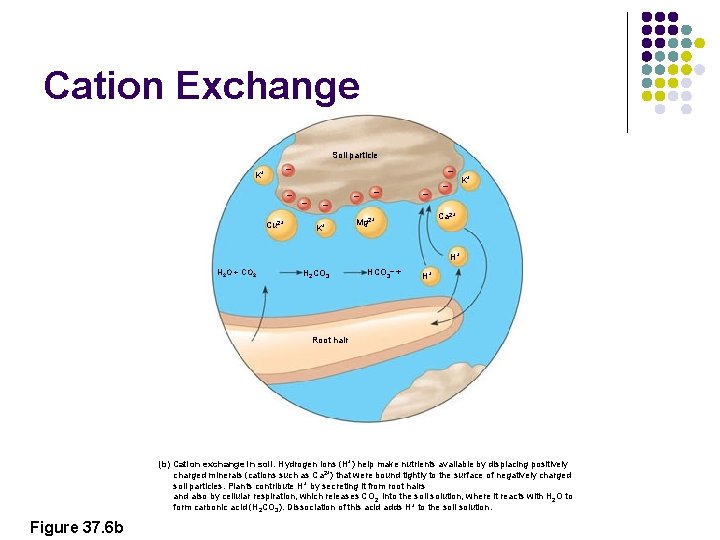
Cation Exchange Soil particle – K+ – Cu 2+ – – – K+ Ca 2+ Mg 2+ H+ H 2 O + CO 2 H 2 CO 3 HCO 3– + H+ Root hair (b) Cation exchange in soil. Hydrogen ions (H+) help make nutrients available by displacing positively charged minerals (cations such as Ca 2+) that were bound tightly to the surface of negatively charged soil particles. Plants contribute H+ by secreting it from root hairs and also by cellular respiration, which releases CO 2 into the soil solution, where it reacts with H 2 O to form carbonic acid (H 2 CO 3). Dissociation of this acid adds H+ to the soil solution. Figure 37. 6 b

Soil Amendments: changing the soil for better plant growth - too sandy, add silt - too fine, add humus or sand - lacking nutrients – add fertilizer

Types of Fertilizers: Organic – dead plant materials or poo – must be further decomposed for nutrients to be absorbed by plants – retained in the soil longer Inorganic – industrial manufactured chemicals (Ex: plant “food”) N-P-K Ratio: percentage of nitrogen, phosphorous and potassium in fertilizer - typically available to the plants almost immediately (may be a slow release) – enter the soil solution quickly - easily leached by heavy rains or excessive irrigation – run-off into ground water (wells) by percolation (methemoglobinemia – blue baby syndrome) , streams or lakes (lead to algal blooms and eutrophication) - p. H adjustments – incorrect p. H may prevent cation exchange - low p. H (acidic) add powdered lime stone - high p. H (base) add acidifiers (peat moss)

Ecological Impact of Irrigation: - taking water from somewhere else - desertification – salts in the water remain after the water has evaporated – the soil has a higher saline content and can become hyperosmotic preventing the plants from taking in water Nitrogen: - must be in ammonia or nitrate form for plants to absorb – plants mainly absorb nitrate and then convert it to ammonia in their cells - Two basic sources - decomposed materials release absorbable forms - atmospheric nitrogen is converted to these forms by nitrifying bacteria in the soil or in root nodules of plants (legumes)

Plant Nutrition and Symbiosis: - Root Nodules – Nitrogen Fixation - Mycorrhizae: “fungus roots” - plants provide sugar to fungus - fungus increases the surface area of the roots for absorption of water and minerals - also secrete growth factors that stimulate roots growth and antibiotics that protect against bacteria - fungal species is usually specific to the plant Two Types: - Ectomycorrhizae – form a sheath of mycelium and penetrate the apoplast of the plant roots - Endomycorrhizae – very fine fungal hyphae that extend into the plant root cell walls but don’t puncture the cell membrane – form branching structures called arbuscules that facilitate nutrient transfer

Strange Nutrition in Plants Epiphytes: grow on other plants – absorb water and minerals from rain, usually through their leaves EX: orchids, Parasites: roots function as haustoria (grow into the cells of other plants) and absorb sugars and nutrients – may or many not be photosynthetic: EX: mistletoe and Indian pipes Carnivores – eat animals – not for energy but for minerals – usually live in environments that are nitrogen and mineral poor – roots can’t absorb the proper minerals so they are extracted from digested animals EX: Venus fly trap, Pitcher plants, Sundews Aggressive Plants: strangler fig

EPIPHYTES Staghorn fern, an epiphyte PARASITIC PLANTS Host’s phloem Dodder Haustoria Mistletoe, a photosynthetic parasite Dodder, a nonphotosynthetic parasite Indian pipe, a nonphotosynthetic parasite CARNIVOROUS PLANTS Venus’ flytrap Pitcher plants Sundews
- Slides: 17