Plant biochemistry II Lipid Anabolism Andy Howard Biochemistry

Plant biochemistry II; Lipid Anabolism Andy Howard Biochemistry Lectures, Fall 2010 8 November 2010 Plants II; lipid anabolism 11/08/2010

Plant biochemistry; lipid anabolism We’ll conclude our study of plant biochemistry n Then we’ll discuss the anabolic pathways associated with lipids n 11/08/2010 Plants II; lipid anabolism 2
What we’ll discuss n Plant biochemistry n n n CAM control Bacterial compartmentation Fatty Acid Synthesis n n n Activation Elongation Special topics n Other anabolic pathways for lipids n n Phospholipid synthesis Synthesis of other glycerol-dependent lipids 11/08/2010 Plants II; lipid anabolism 3
i. Clicker question 1 Starch can be degraded two ways. The most important difference between them is: n (a) Starch phosphorylase requires energy; amylase produces it n (b) Starch phosphorylase breaks off one sugar unit at a time; amylase splits amylopectin into oligosaccharides n (c) Amylase is a larger enzyme n (d) Starch phosphorylase is never found in animals; amylase is n (e) None of the above 11/08/2010 Plants II; lipid anabolism 4

i. Clicker quiz question 2 Why would you not expect to find crassulacean acid metabolism in tropical plants? n (a) Tropical plants do not photosynthesize. n (b) Tropical plants cannot develop the stomata that close off the chloroplast-containing cavities n (c) Water conservation is less critical in areas of high rainfall n (d) The waxy coating required to close off the leaves’ access to O 2 would dissolve in the high humidity and high temperature of the tropics n (e) None of the above 11/08/2010 Plants II; lipid anabolism 5

Answer: (c) n The primary significance of CAM is conservation of water in regions of low humidity, where evaporation rates are high and water is scarce. Neither of these conditions pertains in the tropics. 11/08/2010 Plants II; lipid anabolism 6

Control of CAM n n PEP carboxylase inhibited by malate and low p. H That prevents activity during daylight, which would lead to futile cycling and competition for CO 2 between PEP carboxylase and Ru. Bis. CO 11/08/2010 Plants II; lipid anabolism 7

Compartmentation in bacteria n n n In photosynthetic bacteria, Ru. Bis. CO is concentrated in protein microcompartment called a carboxysome Active carbonic anhydrase there: catalyzes HCO 3 - OH- + CO 2 That tends to keep the CO 2 / O 2 ratio high 11/08/2010 Plants II; lipid anabolism 8
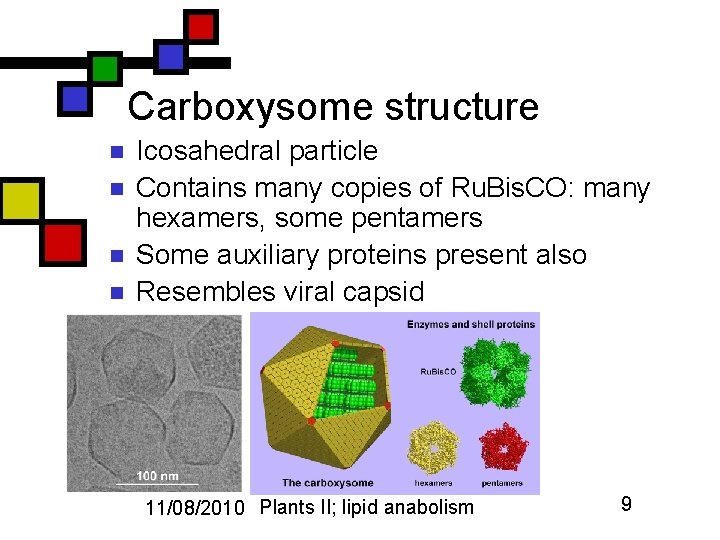
Carboxysome structure n n Icosahedral particle Contains many copies of Ru. Bis. CO: many hexamers, some pentamers Some auxiliary proteins present also Resembles viral capsid 11/08/2010 Plants II; lipid anabolism 9

Making and Breaking Lipids n n Lipid biosynthesis is a significant route to the creation of energy-storage molecules, membrane components, and hormones; Lipid catabolism is a critical energyproducing pathway, and we also need to understand degradation of functional lipids 11/08/2010 Plants II; lipid anabolism 10

Lipids: What we won’t cover today n n n Special Cases Locations for synthesis Regulation by hormones Absorption and mobilization Ketone bodies Catabolism 11/08/2010 Plants II; lipid anabolism 11
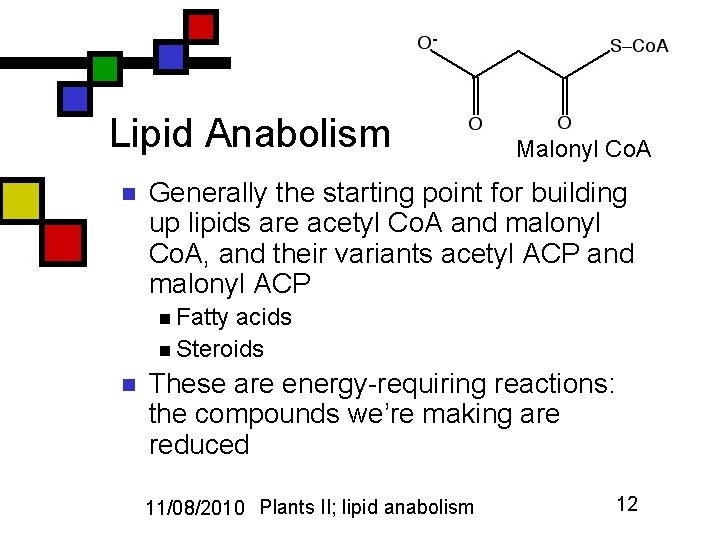
Lipid Anabolism n Malonyl Co. A Generally the starting point for building up lipids are acetyl Co. A and malonyl Co. A, and their variants acetyl ACP and malonyl ACP n Fatty acids n Steroids n These are energy-requiring reactions: the compounds we’re making are reduced 11/08/2010 Plants II; lipid anabolism 12
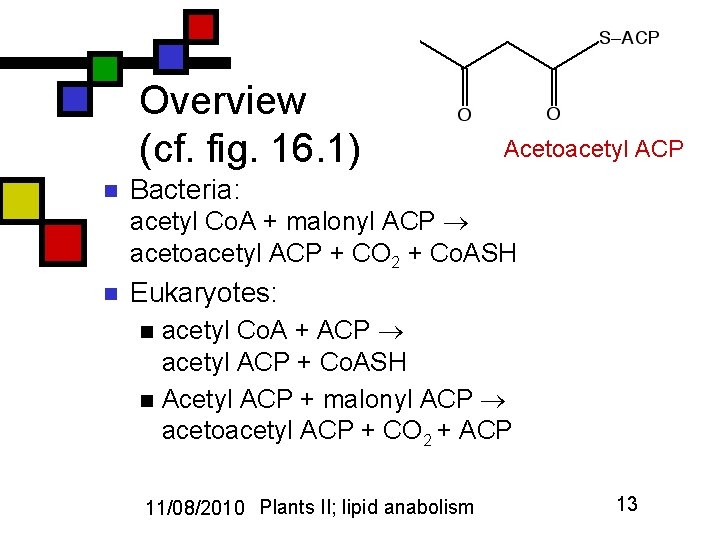
Overview (cf. fig. 16. 1) n Acetoacetyl ACP Bacteria: acetyl Co. A + malonyl ACP acetoacetyl ACP + CO 2 + Co. ASH n Eukaryotes: acetyl Co. A + ACP acetyl ACP + Co. ASH n Acetyl ACP + malonyl ACP acetoacetyl ACP + CO 2 + ACP n 11/08/2010 Plants II; lipid anabolism 13

Making malonyl Co. A n n PDB 1 W 96 (biotin carboxylase domain) 183 k. Da trimer Yeast EC 6. 4. 1. 2, 1. 8Å Acetyl Co. A incorporates an extra carboxyl via acetyl Co. A carboxylase: HCO 3 - + ATP + acetyl Co. A ADP + Pi + malonyl Co. A Biotin- and ATP-dependent 1 UYR (carboxyl-transferase ligase enzyme; similar to domain), 2. 5Å pyruvate carboxylase 162 k. Da dimer; yeast 11/08/2010 Plants II; lipid anabolism 14
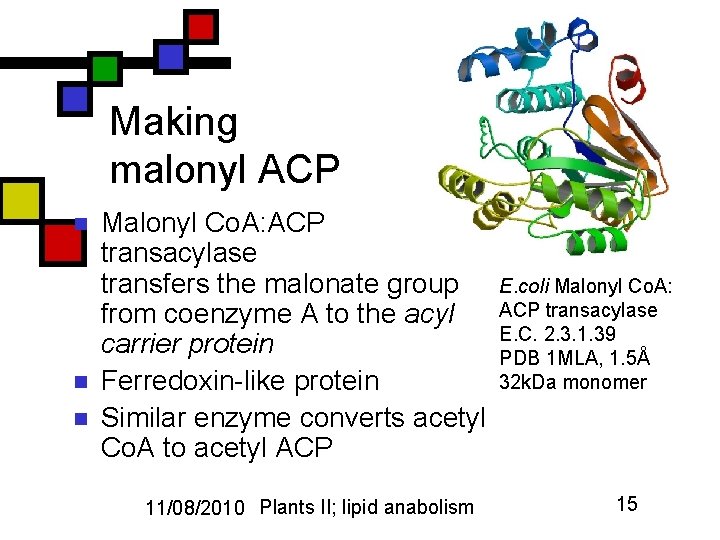
Making malonyl ACP n n n Malonyl Co. A: ACP transacylase transfers the malonate group from coenzyme A to the acyl carrier protein Ferredoxin-like protein Similar enzyme converts acetyl Co. A to acetyl ACP 11/08/2010 Plants II; lipid anabolism E. coli Malonyl Co. A: ACP transacylase E. C. 2. 3. 1. 39 PDB 1 MLA, 1. 5Å 32 k. Da monomer 15
Acyl carrier protein itself n n n Acts as a template on which acyl chain elongation can occur Simple protein: 83 amino acids, mostly helical This is actually an NMR structure PDB 1 OR 5 9. 1 k. Da monomer Streptomyces 11/08/2010 Plants II; lipid anabolism 16
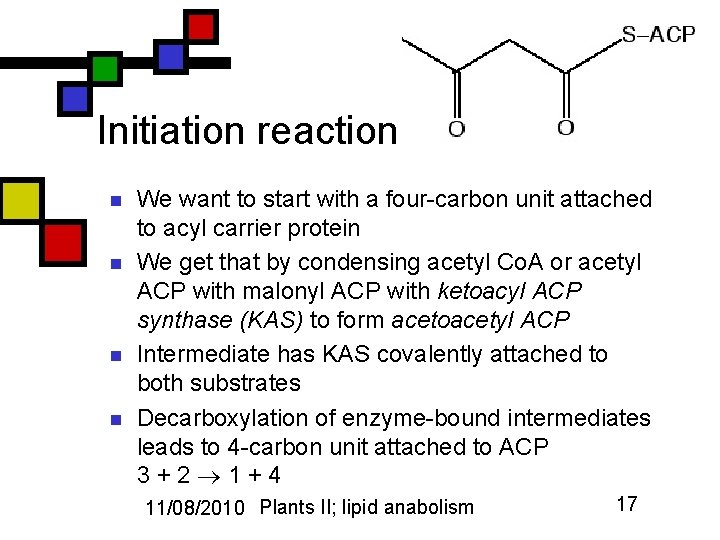
Initiation reaction n n We want to start with a four-carbon unit attached to acyl carrier protein We get that by condensing acetyl Co. A or acetyl ACP with malonyl ACP with ketoacyl ACP synthase (KAS) to form acetoacetyl ACP Intermediate has KAS covalently attached to both substrates Decarboxylation of enzyme-bound intermediates leads to 4 -carbon unit attached to ACP 3+2 1+4 11/08/2010 Plants II; lipid anabolism 17

Is this typical? Yes! n n n We’ve carboxylated acetyl Co. A to make malonyl ACP and then decarboxylated the product of malonyl ACP with acetyl Co. A / ACP This provides a favorable freeenergy change (at the expense of ATP) for the overall reaction Similar approach happens in gluconeogenesis (pyruvate oxaloacetate PEP) 11/08/2010 Plants II; lipid anabolism E. coli Ketoacyl ACP synthase PDB 1 HNJ 70 k. Da dimer; monomer shown EC 2. 3. 1. 41, 1. 46Å 18
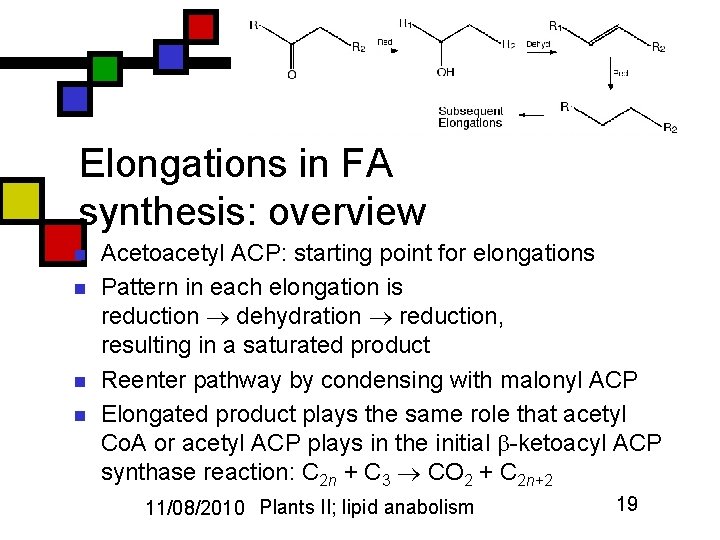
Elongations in FA synthesis: overview n n Acetoacetyl ACP: starting point for elongations Pattern in each elongation is reduction dehydration reduction, resulting in a saturated product Reenter pathway by condensing with malonyl ACP Elongated product plays the same role that acetyl Co. A or acetyl ACP plays in the initial -ketoacyl ACP synthase reaction: C 2 n + C 3 CO 2 + C 2 n+2 11/08/2010 Plants II; lipid anabolism 19

1 st step: reduce ketone sec-alcohol n n Enzyme: 3 -ketoacyl ACP reductase Ketone reacts with NADPH + H+ to produce sec-alcohol + NADP+ D-isomer of sec-alcohol always forms; by contrast, during degradation, L-isomer forms Enzyme is typical NAD(P)-dependent oxidoreductase 11/08/2010 Plants II; lipid anabolism PDB 2 C 07 125 k. Da tetramer; Monomer shown Plasmodium falciparum EC 1. 100, 1. 5Å 20

2 nd step: alcohol to enoyl ACP n n 3 -hydroxyacyl ACP dehydratase Eliminates water at beta, alpha positions to produce PDB 1 DCI 182 k. Da hexamer trans- 2 -enoyl ACP: trimer shown R–CHOH–CH 2 -CO-S-ACP Rat mitochondria R–CH=CH–CO-S-ACP + H 2 O EC 4. 2. 1. 17, 1. 5Å Note that this is a derivative of a trans-fatty acid; but it’s complexed to ACP! This form is primarily helical; there is an alternative found in Aeromonas that is an alpha-beta roll structure 11/08/2010 Plants II; lipid anabolism 21
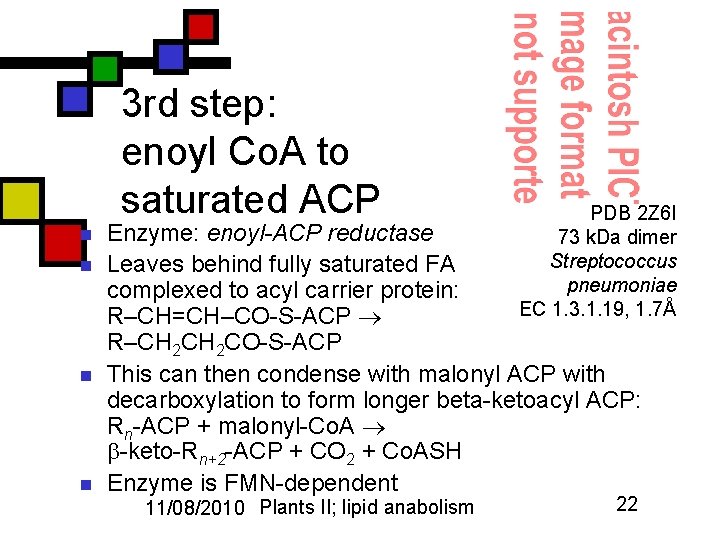
3 rd step: enoyl Co. A to saturated ACP n n PDB 2 Z 6 I 73 k. Da dimer Streptococcus pneumoniae EC 1. 3. 1. 19, 1. 7Å Enzyme: enoyl-ACP reductase Leaves behind fully saturated FA complexed to acyl carrier protein: R–CH=CH–CO-S-ACP R–CH 2 CO-S-ACP This can then condense with malonyl ACP with decarboxylation to form longer beta-ketoacyl ACP: Rn-ACP + malonyl-Co. A -keto-Rn+2 -ACP + CO 2 + Co. ASH Enzyme is FMN-dependent 11/08/2010 Plants II; lipid anabolism 22

How does this end? n n n Generally starts at C 4 and goes to C 16 or C 18. Condensing enzyme won’t fit longer FAs Completed fatty acid is cleaved from ACP by action of a thioesterase with a 3 -layer Rossmann fold 11/08/2010 Plants II; lipid anabolism Palmitoyl thioesterase I PDB 1 EI 9 31 k. Da monomer bovine EC 3. 1. 2. 22, 2. 25Å 23

The overall reaction n Acetyl Co. A + 7 Malonyl Co. A + 14 NADPH + 14 H+ 14 NADP + Palmitate + 7 CO 2 + 8 HS-Co. A + 6 H 2 O In bacteria we have separate enzymes: a type II fatty acid synthesis system In animals we have a type I FA synthesis system: a large, multi-functional enzyme including the phosphopantatheine group by which the ACP attaches 11/08/2010 Plants II; lipid anabolism 24

i. Clicker question 3 What advantage, if any, might be associated with type I fatty acid synthesis systems? n (a) None n (b) Lowered probability of undesirable oxidations of metabolites n (c) Lowered probability of undesirable reductions of metabolites n (d) Reactants remain associated with the enzymatic complex, reducing diffusive inefficiencies n (e) improved solubility of products 11/08/2010 Plants II; lipid anabolism 25

Answer: (d) n If the enzyme doesn’t have to find the substrate at the beginning of each reaction, things will proceed more readily. 11/08/2010 Plants II; lipid anabolism 26

Activating fatty acids n n n Activate stearate or palmitate via acyl Co. A synthetase: PDB 1 BS 0 R–COO + Co. ASH + ATP 42 k. Da monomer R–CO–SCo. A + AMP + PPi E. coli As usual, PPi hydrolysis drives EC 2. 3. 1. 47, 1. 65Å the reaction to the right PLP-dependent reaction Bacteria have one acyl Co. A synthetase Mammals: four isozymes for different FA lengths (small, medium, long, very long) 11/08/2010 Plants II; lipid anabolism 27

Extending and unsaturating fatty acids n n There applications for FAs with more than 18 carbons and FAs with ≥ 1 cis double bonds Elongases and desaturases exist to handle these needs (fig. 16. 7) Desaturase adds a cis-double bond; if the FA already has unsaturations, the new one is added three carbons closer to the carboxyl Elongases condense FA with malonyl Co. A; decarboxylation means we add two carbons 11/08/2010 Plants II; lipid anabolism 28

Bacterial Desaturases n n Acyl ACP desaturases in bacteria simply add a cis double bond in place of the normal trans double bond at the second phase of elongation; the cis double bond thus created remains during subsequent rounds Ferritin-like structure 11/08/2010 Plants II; lipid anabolism PDB 1 ZA 0; 30 k. Da monomer Mycobacterium tuberculosis EC 1. 14. 19. 2, 2Å 29

Eukaryotic Desaturases n n n Desaturases like stearoyl ACP desaturase in eukaryotes act on the completed saturated fatty acyl Co. A species Enzyme is ferritin-like or RNRlike Mammals can’t synthesize linoleate and they need it, so it has to be part of the diet 11/08/2010 Plants II; lipid anabolism PDB 1 OQ 9 80 k. Da dimer monomer shown castor bean EC 1. 14. 19. 2, 2. 4Å 30
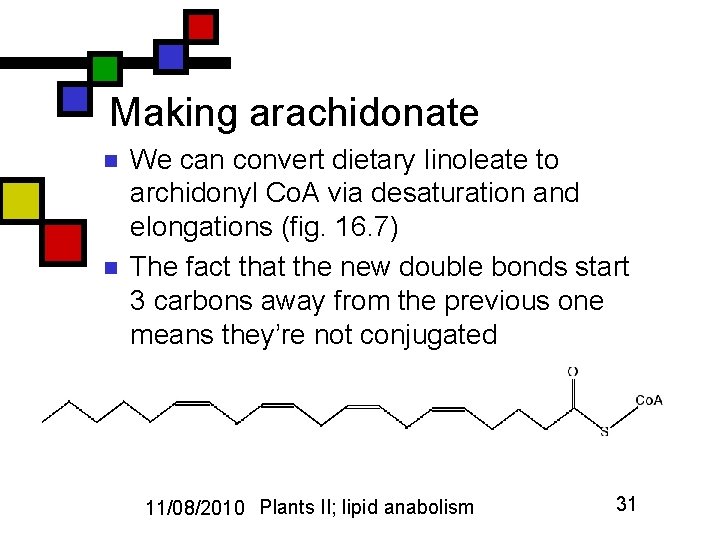
Making arachidonate n n We can convert dietary linoleate to archidonyl Co. A via desaturation and elongations (fig. 16. 7) The fact that the new double bonds start 3 carbons away from the previous one means they’re not conjugated 11/08/2010 Plants II; lipid anabolism 31

We’re done with fatty acid synthesis! n n We’ll study FA degradation Wednesday after the quiz There are several other important anabolic pathways associated with lipids, though. 11/08/2010 Plants II; lipid anabolism 32
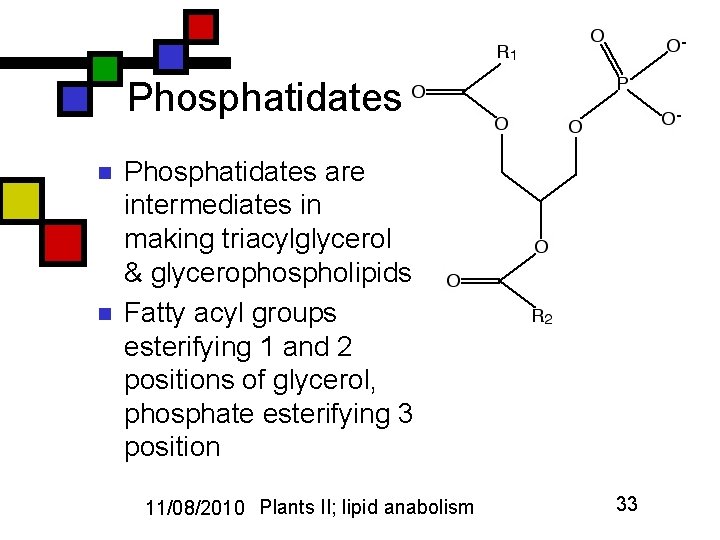
Phosphatidates n n Phosphatidates are intermediates in making triacylglycerol & glycerophospholipids Fatty acyl groups esterifying 1 and 2 positions of glycerol, phosphate esterifying 3 position 11/08/2010 Plants II; lipid anabolism 33

Making phosphatidates n n Glycerol-3 -phosphate acyltransferase transfers acyl Co. A to 1 position of glycerol. Glycerol-3 -P acyltransferase 3 -phosphate; prefers PDB 1 IUQ saturated chains 40 k. Da monomer Cucurbita 1 -acylglycerol-3 -phosphate EC 2. 3. 1. 15, 1. 55Å acyl transferase transfers acyl Co. A to 2 position of resulting molecule; prefers unsaturated chains 11/08/2010 Plants II; lipid anabolism 34

Making triacylglycerols and phospholipids n Phosphatidate phosphatase gets rid of the phosphate at the 3 position by hydrolysis to make 1, 2 -diacylglycerol n n A bit counterintuitive in making phospholipids: why get rid of the phosphate when you’re going to put a phosphorylated compound back at 3 position? But the groups you add already have phosphate on them! 11/08/2010 Plants II; lipid anabolism 35

Further steps in making triacylglycerols n n Diacylglycerol acyltransferase (EC 2. 3. 1. 20; no structures currently available) catalyzes reaction between 1, 2 -diacylglycerol and acyl Co. A to form triacylglycerol See fig. 16. 9, left-hand side 11/08/2010 Plants II; lipid anabolism 36

Making phospholipids from 1, 2 -diacylglycerol n n 1, 2 -diacylglycerol reacts with CDPcholine to form phosphatidylcholine with liberation of cytidine monophosphate 1, 2 -diacylglycerol reacts with CDPethanolamine to form phosphatidylethanolamine n this can be methylated 3 times to make phosphatidylcholine n S-adenosylmethionine is the methyl donor in that case 11/08/2010 Plants II; lipid anabolism 37
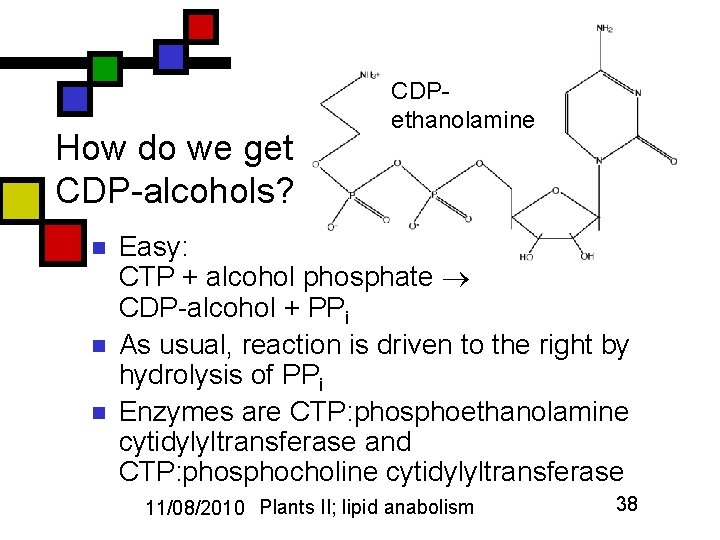
How do we get CDP-alcohols? n n n CDPethanolamine Easy: CTP + alcohol phosphate CDP-alcohol + PPi As usual, reaction is driven to the right by hydrolysis of PPi Enzymes are CTP: phosphoethanolamine cytidylyltransferase and CTP: phosphocholine cytidylyltransferase 11/08/2010 Plants II; lipid anabolism 38
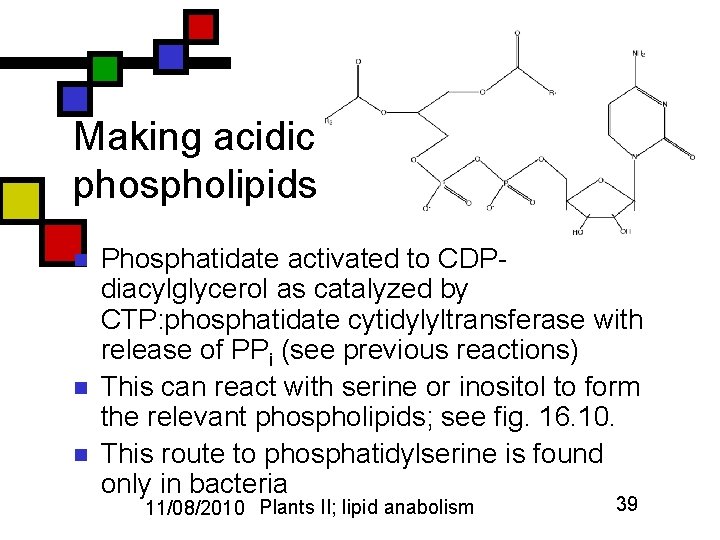
Making acidic phospholipids n n n Phosphatidate activated to CDPdiacylglycerol as catalyzed by CTP: phosphatidate cytidylyltransferase with release of PPi (see previous reactions) This can react with serine or inositol to form the relevant phospholipids; see fig. 16. 10. This route to phosphatidylserine is found only in bacteria 11/08/2010 Plants II; lipid anabolism 39
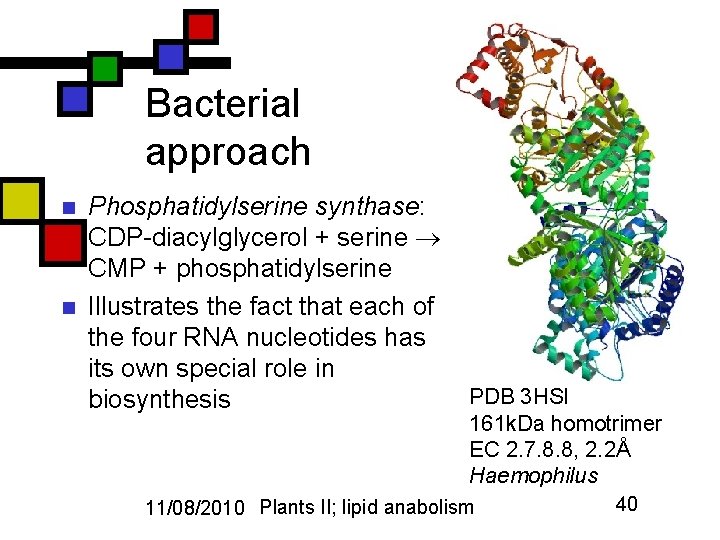
Bacterial approach n n Phosphatidylserine synthase: CDP-diacylglycerol + serine CMP + phosphatidylserine Illustrates the fact that each of the four RNA nucleotides has its own special role in biosynthesis PDB 3 HSI 161 k. Da homotrimer EC 2. 7. 8. 8, 2. 2Å Haemophilus 11/08/2010 Plants II; lipid anabolism 40
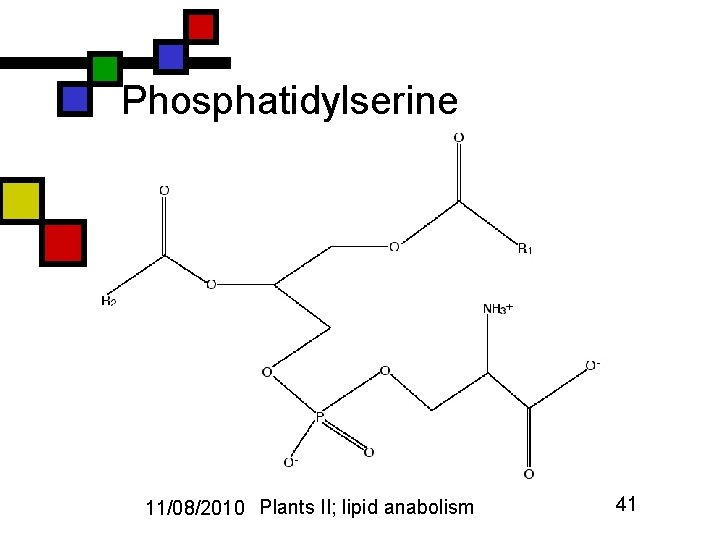
Phosphatidylserine 11/08/2010 Plants II; lipid anabolism 41
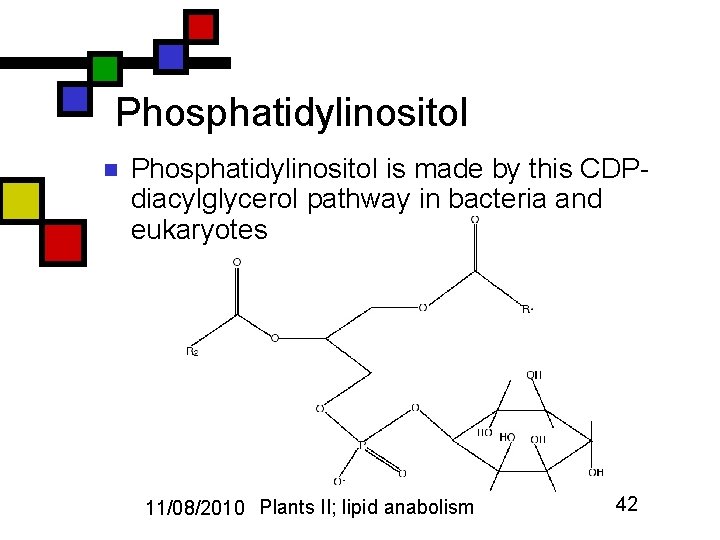
Phosphatidylinositol n Phosphatidylinositol is made by this CDPdiacylglycerol pathway in bacteria and eukaryotes 11/08/2010 Plants II; lipid anabolism 42

Making phosphatidylserine n n n Alternative approach to phosphatidylserine found in eukaryotes: make phosphatidylethanolamine, then phosphatidylethanolamine: serine transferase swaps serine for ethanolamine When we do it that way, we can recover phosphatidylethanolamine back by a decarboxylation (or another exchange) Ethanolamine is just serine without COO- ! 11/08/2010 Plants II; lipid anabolism 43

Where does this happen? n n Mostly in the endoplasmic reticulum in eukaryotes Biosynthesis enzymes are membrane bound but have their active sites facing the cytosol so they can pick up the water-soluble metabolites from which they can build up phospholipids and other lipids 11/08/2010 Plants II; lipid anabolism 44

Making eicosanoids n Classes of eicosanoids: n Prostaglandins and thromboxanes n Leukotrienes n n Remember that we make arachidonate from linoleoyl Co. A; eiconsanoids made from arachidonate Reactions involve formation of oxygencontaining rings; thus the enzymes are cyclooxygenases 11/08/2010 Plants II; lipid anabolism 45

What eiconsanoids do n n They’re like hormones, but they act very locally: within µm of the cell in which they’re produced Involved in platelet aggregation, blood clots, constriction of smooth muscles Mediate pain sensitivity, inflammation, swelling Therefore enzymes that interconvert them are significant drug targets! 11/08/2010 Plants II; lipid anabolism 46
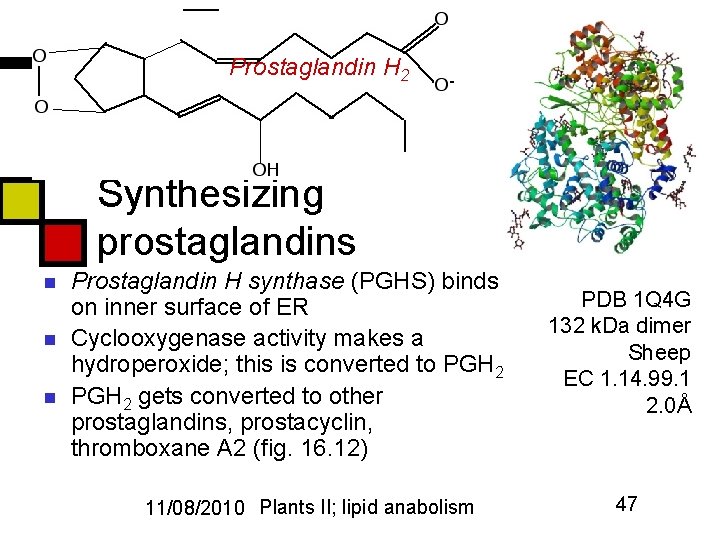
Prostaglandin H 2 Synthesizing prostaglandins n n n Prostaglandin H synthase (PGHS) binds on inner surface of ER Cyclooxygenase activity makes a hydroperoxide; this is converted to PGH 2 gets converted to other prostaglandins, prostacyclin, thromboxane A 2 (fig. 16. 12) 11/08/2010 Plants II; lipid anabolism PDB 1 Q 4 G 132 k. Da dimer Sheep EC 1. 14. 99. 1 2. 0Å 47
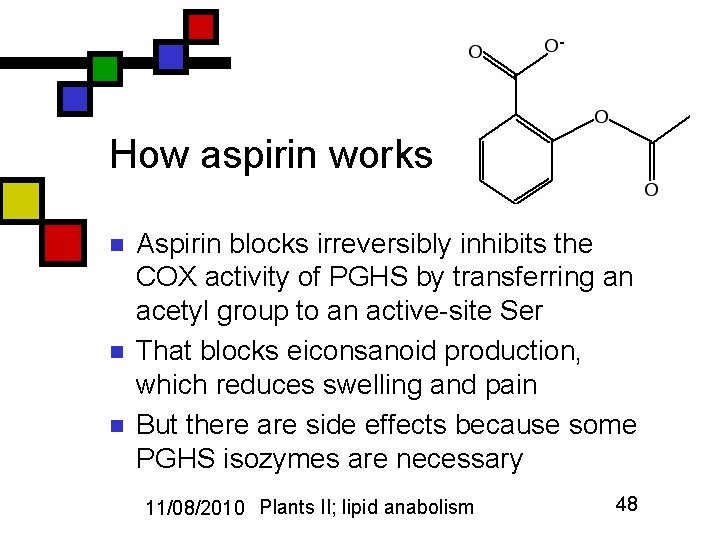
How aspirin works n n n Aspirin blocks irreversibly inhibits the COX activity of PGHS by transferring an acetyl group to an active-site Ser That blocks eiconsanoid production, which reduces swelling and pain But there are side effects because some PGHS isozymes are necessary 11/08/2010 Plants II; lipid anabolism 48

Cyclooxygenase inhibition n n Cox-1 is constitutive and regulates secretion of mucin in the stomach Cox-2 is inducible and promotes inflammation, pain, fever Aspirin inhibits both: the mucin-secretion inhibition means that causes bleeding or ulcers in the stomach lining Other nonsteroidal anti-inflammatories (NSAIDs) besides aspirin compete with arachidonate rather than binding covalently to COX-1 and COX-2 11/08/2010 Plants II; lipid anabolism 49

Could we find a COX-2 inhibitor? n n n This would eliminate the stomach irritation that aspirin causes Some structure-based inhibitors have been developed They work as expected; but They also increase risk of cardiovascular disease Prof. Prancan (Rush U) discussed these issues in his February 2007 colloquium 11/08/2010 Plants II; lipid anabolism 50

Leukotrienes n n Lipoxygenases convert arachidonate to these compounds, which contain 3 conjugated double bonds These compounds interact with GPCRs Involved in inflammatory and allergic reactions Also involved in the pathophysiology of asthma Leukotriene B 4 11/08/2010 Plants II; lipid anabolism PDB 2 P 0 M 146 k. Da dimer rabbit EC 1. 13. 11. 33 2. 4Å 51
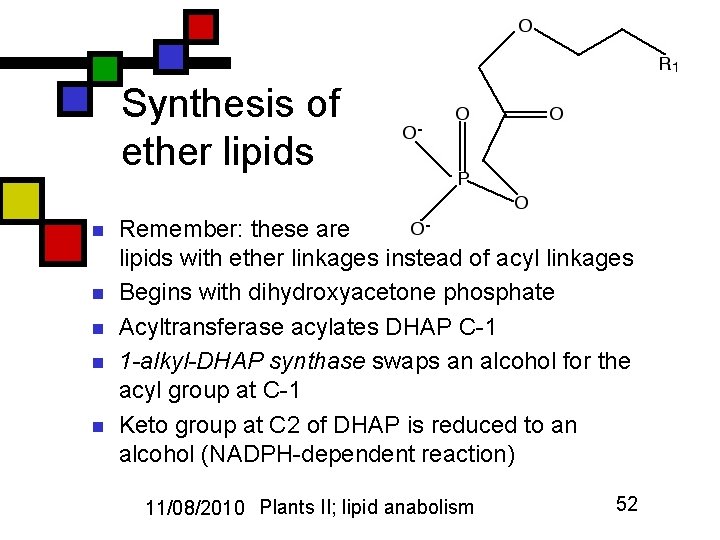
Synthesis of ether lipids n n n Remember: these are lipids with ether linkages instead of acyl linkages Begins with dihydroxyacetone phosphate Acyltransferase acylates DHAP C-1 1 -alkyl-DHAP synthase swaps an alcohol for the acyl group at C-1 Keto group at C 2 of DHAP is reduced to an alcohol (NADPH-dependent reaction) 11/08/2010 Plants II; lipid anabolism 52
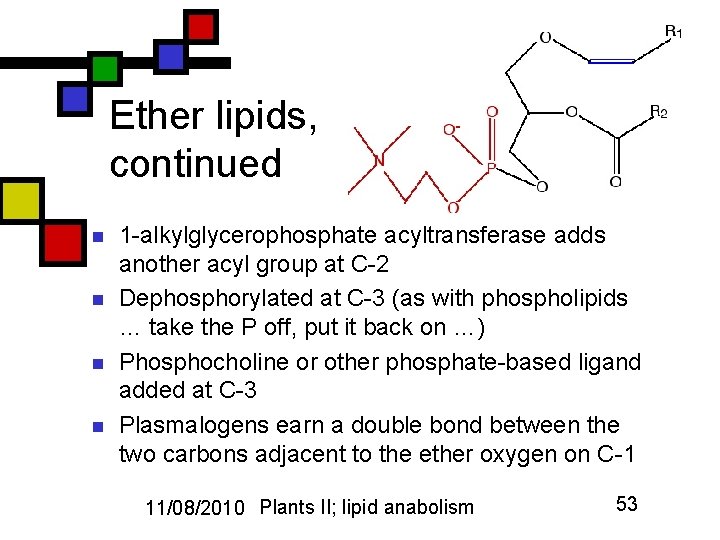
Ether lipids, continued n n 1 -alkylglycerophosphate acyltransferase adds another acyl group at C-2 Dephosphorylated at C-3 (as with phospholipids … take the P off, put it back on …) Phosphocholine or other phosphate-based ligand added at C-3 Plasmalogens earn a double bond between the two carbons adjacent to the ether oxygen on C-1 11/08/2010 Plants II; lipid anabolism 53
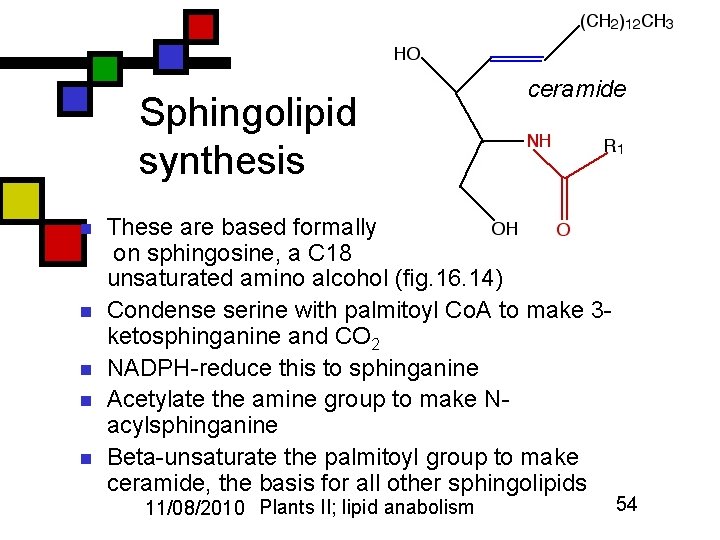
Sphingolipid synthesis n n n ceramide These are based formally on sphingosine, a C 18 unsaturated amino alcohol (fig. 16. 14) Condense serine with palmitoyl Co. A to make 3 ketosphinganine and CO 2 NADPH-reduce this to sphinganine Acetylate the amine group to make Nacylsphinganine Beta-unsaturate the palmitoyl group to make ceramide, the basis for all other sphingolipids 11/08/2010 Plants II; lipid anabolism 54
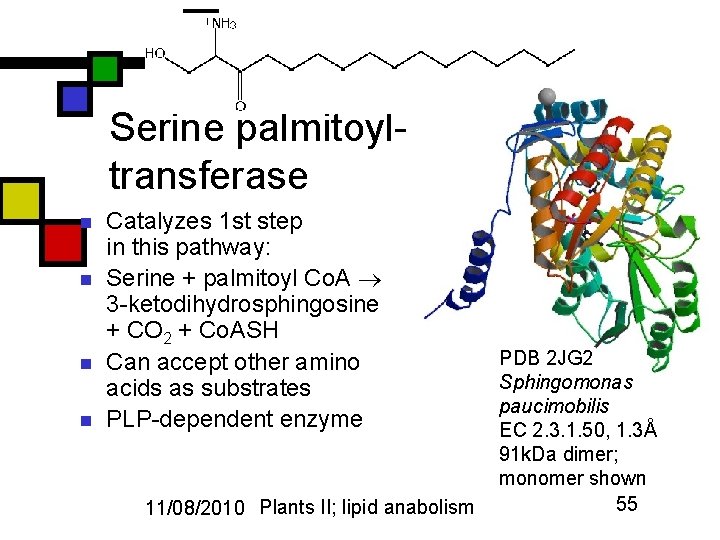
Serine palmitoyltransferase n n Catalyzes 1 st step in this pathway: Serine + palmitoyl Co. A 3 -ketodihydrosphingosine + CO 2 + Co. ASH Can accept other amino acids as substrates PLP-dependent enzyme 11/08/2010 Plants II; lipid anabolism PDB 2 JG 2 Sphingomonas paucimobilis EC 2. 3. 1. 50, 1. 3Å 91 k. Da dimer; monomer shown 55
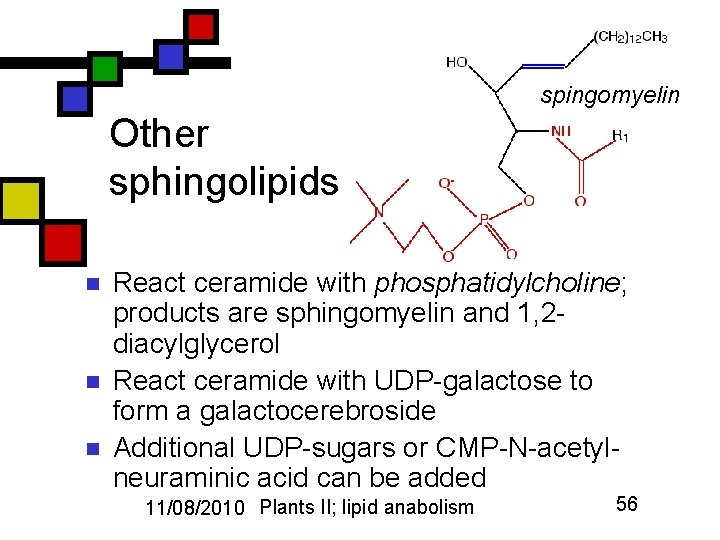
spingomyelin Other sphingolipids n n n React ceramide with phosphatidylcholine; products are sphingomyelin and 1, 2 diacylglycerol React ceramide with UDP-galactose to form a galactocerebroside Additional UDP-sugars or CMP-N-acetylneuraminic acid can be added 11/08/2010 Plants II; lipid anabolism 56
- Slides: 56