Plancks hypothesis and Einsteins photon theory Contents Hubris
Planck’s hypothesis, and Einstein’s photon theory. Contents: • Hubris at the turn of the century • Black Body radiation • The problem with black body radiation • Planck’s hypothesis • Einstein’s corpuscular light theory • Photons : Example 1 | Example 2 • Whiteboards

Hubris • 1900 +/- 10. • The end of physics • Tricky bits • Motion of the earth with respect to “ether” • (Einsteinian relativity) • Black body radiation • (Quantum mechanics) TOC
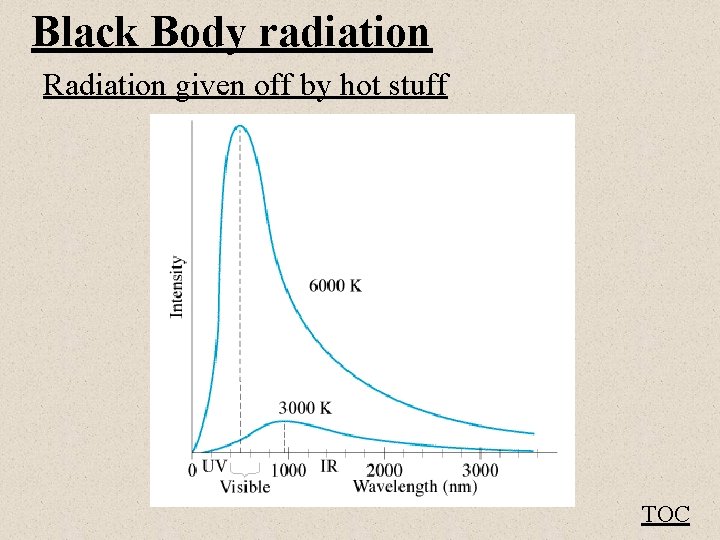
Black Body radiation Radiation given off by hot stuff TOC
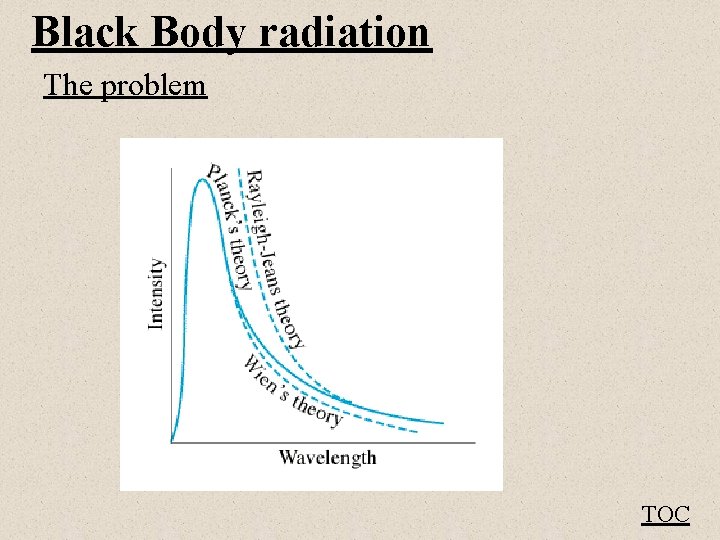
Black Body radiation The problem fig 27 -5 TOC

Black Body radiation Planck’s solution Planck proposes a quantum hypothesis Emin = hf h = Planck’s constant = 6. 626 x 10 -34 Js f = frequency of oscillations (Hz, s-1) E = nhf, n = 1, 2, 3, . . . Hypothesis fits data exactly. Planck thinks it’s just math, not reality TOC

Einstein’s photon theory Einstein: Reality is quantum Radiation is also quantized Photons = corpuscular theory E = hf v = f E = Photon energy (Joules) v = c = speed of light = 3. 00 x 108 m/s f = frequency (Hz) = wavelength (m) TOC

E = hf v = f E = Photon energy (Joules) v = c = speed of light = 3. 00 x 108 m/s f = frequency (Hz) = wavelength (m) Example 1: What is the energy of a 460 nm photon? v = f , c = f , f = c/ E = hf = hc/ TOC

E = hf v = f E = Photon energy (Joules) v = c = speed of light = 3. 00 x 108 m/s f = frequency (Hz) = wavelength (m) Example 2: A photon has an energy of 13 e. V. What is its wavelength? (Remember V = W/q, and 1 e. V is one electron moved through 1 volt) E (in Joules) = (13 e. V)(1. 602 x 10 -19 J/e. V) c = f , = c/f E = hf, f = E/h = c/f = hc/E TOC

Whiteboards: Photons 1|2|3|4 TOC
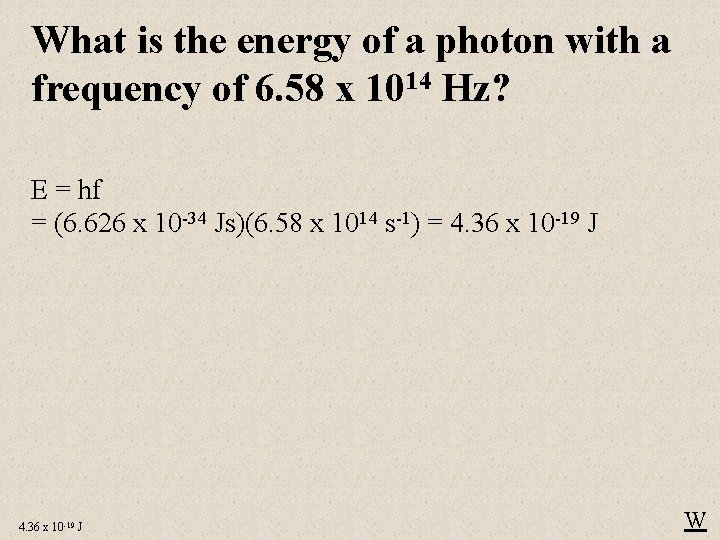
What is the energy of a photon with a frequency of 6. 58 x 1014 Hz? E = hf = (6. 626 x 10 -34 Js)(6. 58 x 1014 s-1) = 4. 36 x 10 -19 J W

What is the frequency of a photon with an energy of 5. 45 x 10 -18 J? What is its wavelength? E = hf f = E/h = (5. 45 x 10 -18 J)/(6. 626 x 10 -34 Js) = 8. 23 x 1015 Hz v = f , = v/f = (3. 00 x 108 m/s)/(8. 23 x 1015 Hz) = 36. 5 nm 8. 23 x 1015 Hz, 36. 5 nm W

What is the energy in Joules of a 314 nm photon? What is that energy in e. V? (2) E = hf = hc/ 1 e. V = 1. 602 x 10 -19 J E = hc/ = (6. 626 x 10 -34 Js)(3. 00 x 108 m/s)/(3. 14 x 10 -9 m) = 6. 33 x 10 -19 J E = (6. 33 x 10 -19 J)/(1. 602 x 10 -19 J/e. V) = 3. 95 e. V 6. 33 x 10 -19 J, 3. 95 e. V W

A photon has an energy of 6. 02 e. V. What is its wavelength? (answer in nm) (2) E = hf = hc/ 1 e. V = 1. 602 x 10 -19 J E = (6. 02 e. V)(1. 602 x 10 -19 J/e. V) = 9. 64 x 10 -19 J E = hc/ , = hc/E = (6. 626 x 10 -34 Js)(3. 00 x 108 m/s)/(9. 64 x 10 -19 J) = 2. 06 x 10 -7 m = 206 nm (nm = 1 x 10 -9 m) 206 nm W

How many photons per second stream from a 620. nm, 300. m. W laser? (2) E = hf = hc/ In one second, a 300 m. W laser emits photons with a total energy of. 300 J E = hc/ , = (6. 626 x 10 -34 Js)(3. 00 x 108 m/s)/(620. x 10 -9 m) = 3. 20613 x 10 -19 J (per photon) # photons = (. 300 J/s)/(3. 20613 x 10 -19 J/photon) = 9. 36 x 1017 photons/sec Hey, that’s a lot! 9. 36 x 1017 photons/sec W

Light Waves Wavelength Changes Color small = blue Photons Energy per photon changes E = hf X-Rays, UV, Gamma big = red Amplitude Changes Brightness small = dim big = bright # of photons changes many = bright few = dim CCD Devices, High speed film
- Slides: 15