Planar Organic Families Unsaturated compounds Contain a double

Planar Organic Families • Unsaturated compounds • Contain a double or a triple bond

Alkenes Name ends in –ene Need to know up to C-4 First 3 are gases, next 1 is a liquid Insoluble in water (do not form hydrogen bonds) • Soluble in organic solvents, e. g. , cyclohexane • Uses = plastics/polymers • •

Aldehydes • Have a C=O bond (carbonyl group) at the end of the carbon chain • Name ends in –al • Need to know up to C-4 • Lower chains soluble in water – C=O bond forms hydrogen bonds with water molecules – but solubility decreases as chains grow longer • Soluble in organic solvents, e. g. , cyclohexane

• C=O bond is highly polar so dipole-dipole forces occur between molecules • Higher boiling points than alkanes because of these dipole-dipole forces • Uses = benzaldehyde found in almond oil (gives it its scent)

Ketones • Have a C=O bond (carbonyl group) in the middle of the carbon chain • Name ends in – one • Need to know up to C-4 • Lower chains soluble in water – C=O bond forms hydrogen bonds with water molecules – but solubility decreases as chains grow longer • Soluble in organic solvents, e. g. , cyclohexane

• C=O bond is highly polar so dipole-dipole forces occur between molecules • Higher boiling points than alkanes because of these dipole-dipole forces • Uses = industrial solvents (propanone = acetone (nail polish remover))

Carboxylic acids • Have a C=O bond (carbonyl group) and a C-OH bond at the end of the carbon chain • Name ends in – oic acid • Need to know up to C-4 • Lower chains soluble in water – C=O bond forms hydrogen bonds with water molecules – but solubility decreases as chains grow longer • Soluble in organic solvents, e. g. , cyclohexane

• Hydrogen bonds occur between molecules forming dimers • Higher boiling points than alcohols because of this hydrogen bonding • Uses = salts of propanoic & butanoic acid used in food preservation, ethanoic acid used to make cellulose acetate ( photographic film, varnishes)

• Methanoic acid is in nettles & ants • Ethanoic acid is in vinegar

Esters • Have a C=O bond (carbonyl group) and a C-O bond in the middle of the carbon chain called an ester linkage • Formed from a carboxylic acid an alcohol • First part of the name comes from the alcohol (right of linkage) • Second part of the name comes from the carboxylic acid (left of linkage)

• Need to know up to C-4 • First few members are volatile liquids (low bp) • Lower chains soluble in water – C=O bond forms hydrogen bonds with water molecules – but solubility decreases as chains grow longer • Soluble in organic solvents, e. g. , cyclohexane • Low boiling points because cannot form hydrogen bonds with each other

• Uses = fruity smells, ethyl ethanoate is an excellent solvent, polyester is a long chain of esters • Formation of esters is a condensation reaction (not an elimination reaction because no new double bond formed)

Aromatics • Contain a benzene ring • Need to know benzene, methylbenzene & ethylbenzene • Insoluble in water (do not form hydrogen bonds) • Soluble in organic solvents, e. g. , cyclohexane

• Uses = used to a widely used solvent but is carcinogenic so no longer used • Not all aromatic compounds are carcinogenic – dyes, detergents, indicators (methyl orange & phenolphthaelin), aspirin, ibuprofen etc
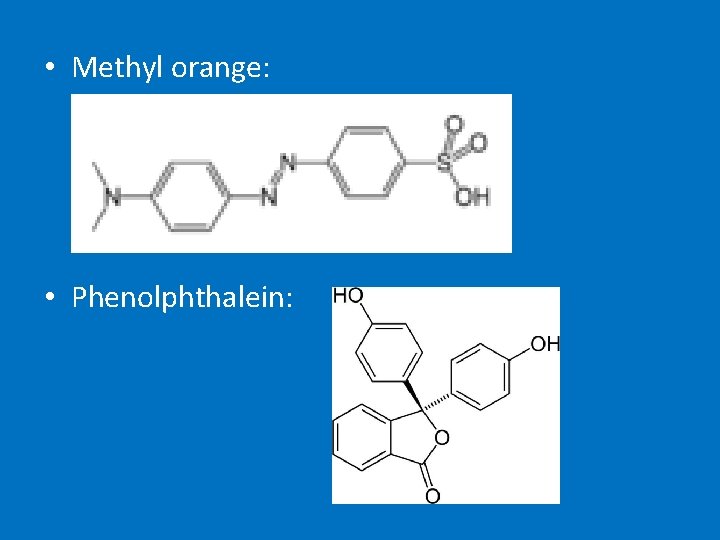
• Methyl orange: • Phenolphthalein:
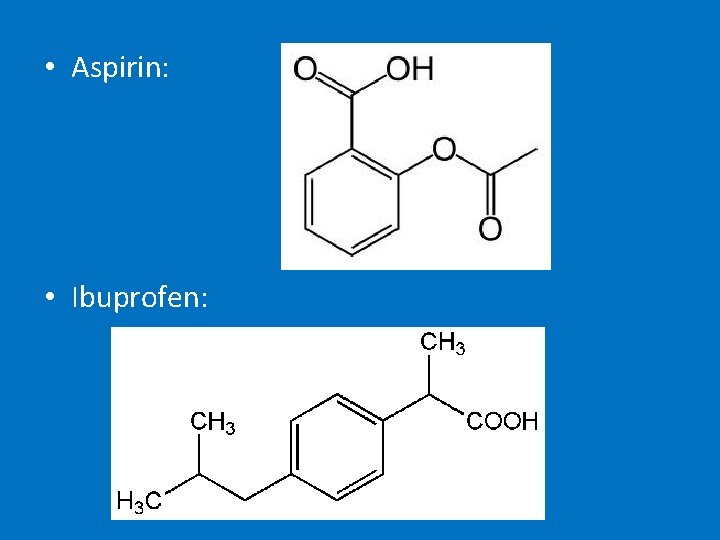
• Aspirin: • Ibuprofen:
- Slides: 16