Pitvarfibrillci kezelse Ritmus vagy frekvencia kontroll Zmolyi Kroly

Pitvarfibrilláció kezelése Ritmus vagy frekvencia kontroll? Zámolyi Károly Szent Ferenc Kórház, Budapest
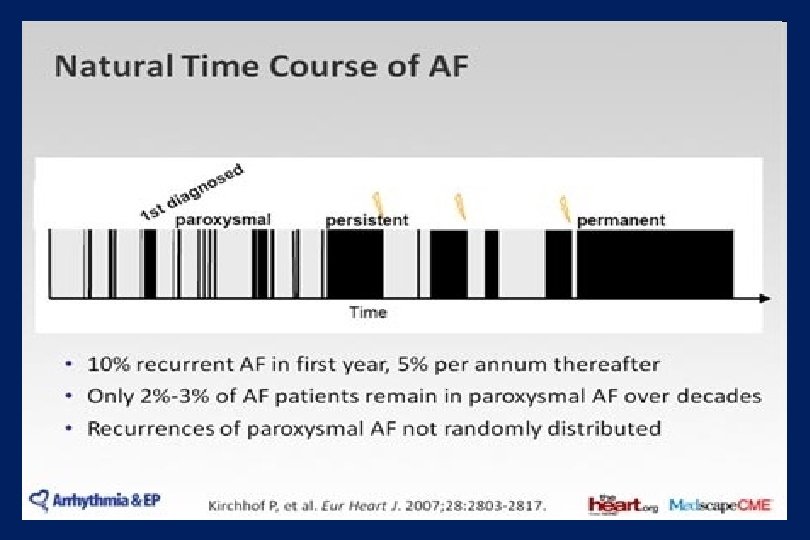
Silent

Mi a terápia célja? v Tünetek csökkentése (palpitatio, fáradtság, dyspnoe) v Tromboembolia megelőzése v Tachycardia indukálta myocardialis remodelling és szívelégtelenség megelőzése v Alapbetegség kezelése (hypertonia a legfontosabb)

Decreasing Atrial Fibrillation Burden Is an Important Goal • As with heart failure or angina, success in managing atrial fibrillation is defined as a decrease in: Frequency of episodes Duration of episodes Symptoms during episodes • Decreasing atrial fibrillation burden offers potential to successfully treat atrial fibrillation by: – Decreasing mortality – Decreasing hospitalizations – Increasing Qo. L Prystowsky EN. J Cardiovasc Electrophysiol. 2006; 17(suppl 2): S 7 -S 10; Wolf PA et al. Arch Intern Med. 1998; 158: 229 -234.
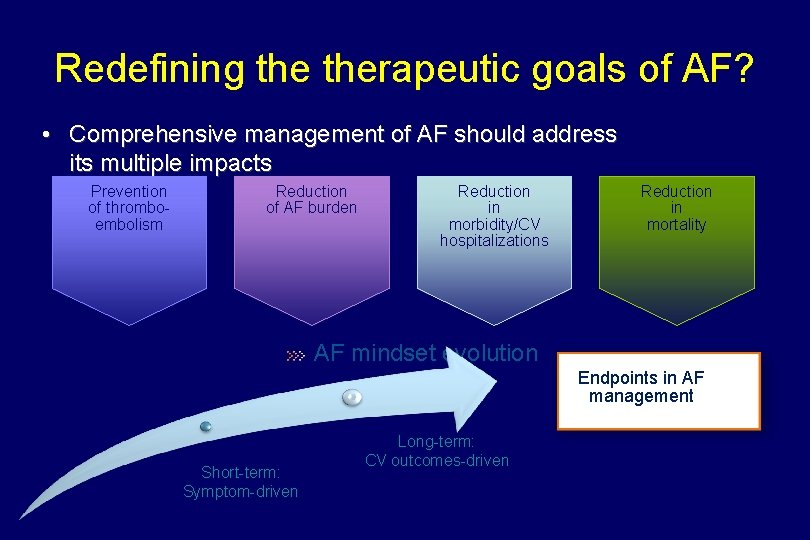
Redefining therapeutic goals of AF? • Comprehensive management of AF should address its multiple impacts Prevention of thromboembolism Reduction of AF burden Reduction in morbidity/CV hospitalizations Reduction in mortality AF mindset evolution Endpoints in AF management Short-term: Symptom-driven Long-term: CV outcomes-driven

Changing the treatment paradigm for AF in 2009 Old paradigm • • Rate vs rhythm AF recurrence=failure MD chooses Rx Focus: ECG New paradigm • Rate and rhythm • AF recurrence only important if symptomatic and severe • MD and patient enter into a therapeutic contract • Focus: patient NSR = normal sinus rhythm.

Tachycardia következményei Csökken az EF § Diastolés dysfunctio § Megnő a LVEDP, PAP, SVR § Nő az ESV, EDV § Csökkent a CO § Csökken a kontraktilis rezerv § Nő az ANP, epinephrin, norepinephrin, aldosteron § Myocyta vesztés reaktív celluláris hypertrophiával § Tachycardia indukálta cardiomyopathia Scumacher és Luderitz AJC 1998; 82: 29 N §
www. escardio. org/guidelines

Kezelési stratégia PF - ban Frekvencia kontroll és anticoagulálás Sinus ritmus fenntartás + anticoagulálás Effektivitás Biztonságosság Életminőség (AFFIRM, RACE)

Sinus ritmus fenntartás Előny • Elektromos és anatómiai remodeling megelőzése • Javulnak a tünetek • Nő a terhelési kapacitás • Javul a haemodynamika • Javul az életminőség • Visszaáll a pitvari traszport • Csökken a tromboembolia? • Prognózis?

Sinus ritmus fenntartás Hátrány • Kamrai proarrhythmia • Nő a mortalitás? • Gyógyszer okozta bradyarrhythmia • Szerv toxicitás • PF rekurrálás valószínű • Silent PF

Frekvencia kontroll Előny • Csökkennek a tünetek • Nincs proarrhythmia • Csökken a tachycardia indukálta CMP • Alacsony ár Hátrány • Tartós anticoagulálás szükséges • Rosszabb haemodynamika • Progresszív remodeling • PF permanenssé válik


The AFFIRM study • First major outcome study to compare long -term clinical benefits of rhythm control versus rate control strategies in AF patients at high–risk for stroke The AFFIRM Investigators. N Eng J Med 2002; 347(23): 1825 -33.

AFFIRM: objective • Comparing two treatment strategies in AF patients at high-risk for stroke, for preventing mortality – Rhythm control – Ventricular rate control

AFFIRM: study design • Multicenter, randomized study • N = 4060 patients • Mean follow-up: 3. 5 years (max 6 years) RHYTHM CONTROL AAD as chosen by physician: amiodarone, disopyramide, flecainide, moricizine, procainamide, propafenone, quinidine, sotalol, dofetilide, or a combination Cardioversion included as necessary RATE CONTROL Beta-blockers, calcium-channel blockers (verapamil and diltiazem), digoxin, or a combination Treatment goal: Heart rate within range 80 -110 bpm during sixminute walk test • Both treatment groups received oral anticoagulant therapy (INR 2. 0 to 3. 0): mandatory for rate control strategy, but only recommended for rhythm control strategy The AFFIRM Investigators. N Eng J Med 2002; 347(23): 1825 -33.

AFFIRM: inclusion/exclusion criteria • Inclusion criteria: – AF patient at high-risk for stroke [N = 4, 060] – Age ≥ 65 • or age < 65, plus ≥ 1 clinical risk factor* for stroke – Patient eligible for both rhythm and rate strategies • Exclusion criteria: – Lone AF – CHF patients – Absolute contraindication to warfarin * Hypertension, diabetes, CHF, TIA, prior stroke, echocardiographic parameters

AFFIRM: baseline characteristics • • • Age ~ 70 years old Women: 39% White: 89% Hypertension: 71% CAD: 38% CHF: 23% LVEF normal in > 2/3 patients AF as 1 st qualifying episode in 36% patients Qualifying episode <6 wks: 90% Am Heart J 146(6): 991 -1001

AFFIRM: drugs used in the two groups Warfarin NEJM 347(23): 1825 -1833 (85. 0) (70. 0)
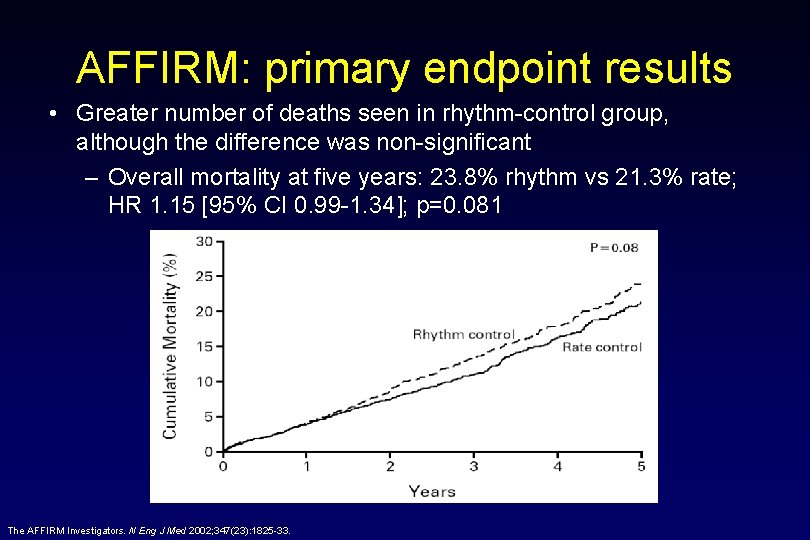
AFFIRM: primary endpoint results • Greater number of deaths seen in rhythm-control group, although the difference was non-significant – Overall mortality at five years: 23. 8% rhythm vs 21. 3% rate; HR 1. 15 [95% CI 0. 99 -1. 34]; p=0. 081 The AFFIRM Investigators. N Eng J Med 2002; 347(23): 1825 -33.

AFFIRM: composite secondary endpoint • The rates of the composite secondary endpoint* were similar p=0. 33 – Rate-control: 32. 7% – Rhythm-control: 32. 0% Death, disabling stroke, disabling anoxic encephalopathy, major bleeding, or cardiac arrest *

Adverse events • Two adverse events were significantly more frequent in the rhythm-control group: – Torsades de pointes (0. 8% vs 0. 2%, p=0. 007) – All-cause hospitalisation after baseline (80. 1% vs 73. 0%, p<0. 001) The AFFIRM Investigators. N Eng J Med 2002; 347(23): 1825 -33.
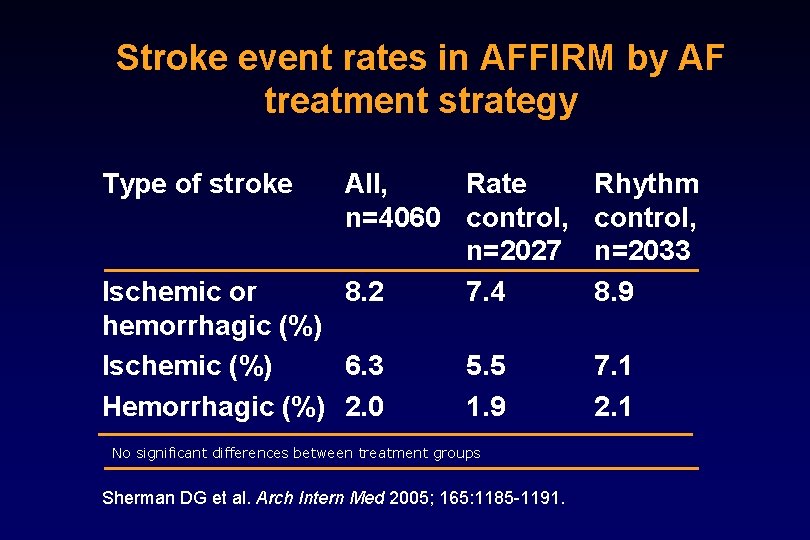
Stroke event rates in AFFIRM by AF treatment strategy Type of stroke All, Rate n=4060 control, n=2027 8. 2 7. 4 Ischemic or hemorrhagic (%) Ischemic (%) 6. 3 Hemorrhagic (%) 2. 0 5. 5 1. 9 No significant differences between treatment groups Sherman DG et al. Arch Intern Med 2005; 165: 1185 -1191. Rhythm control, n=2033 8. 9 7. 1 2. 1
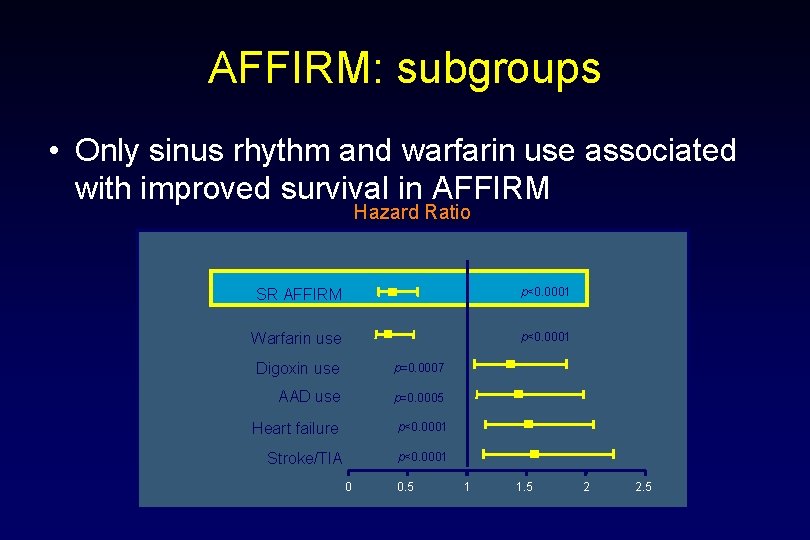
AFFIRM: subgroups • Only sinus rhythm and warfarin use associated with improved survival in AFFIRM Hazard Ratio SR AFFIRM p<0. 0001 Warfarin use p<0. 0001 Digoxin use p=0. 0007 AAD use p=0. 0005 Heart failure p<0. 0001 Stroke/TIA p<0. 0001 0 0. 5 1 1. 5 2 2. 5

AFFIRM: increased functional capacity • Mean NYHA Functional Class score was significantly better at each visit in patients in sinus rhythm Chung MK, et al. J Am Coll Cardiol 2005; 46(10): 1891 -9.

AFFIRM: the stroke problem • In AFFIRM and other trials, anticoagulants (e. g. warfarin) used long-term in rate control groups, but discontinued early in rhythm control groups • Increasing evidence that anticoagulants must be given long-term as part of rhythm control strategy, even in absence of symptomatic episodes
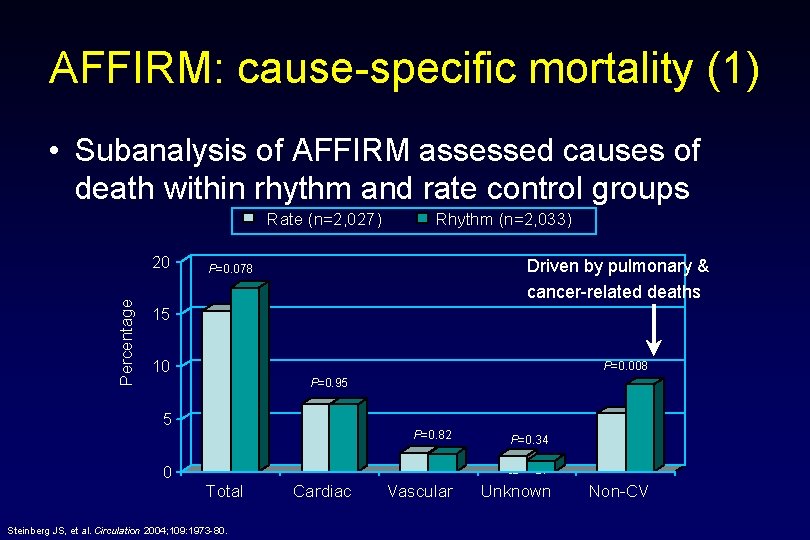
AFFIRM: cause-specific mortality (1) • Subanalysis of AFFIRM assessed causes of death within rhythm and rate control groups Rate (n=2, 027) Percentage 20 15 Rhythm (n=2, 033) Driven by pulmonary & cancer-related deaths P=0. 078 356 310 10 P=0. 008 P=0. 95 169 5 130 129 P=0. 82 37 0 Total Steinberg JS, et al. Circulation 2004; 109: 1973 -80. Cardiac 35 Vascular P=0. 34 32 113 21 Unknown Non-CV

AFFIRM: conclusions (1) • AFFIRM compared rate-control and rhythmcontrol strategies for the treatment of 4060 AF patients at high-risk for stroke • Disappointing results: More deaths occurred in the rhythm-control group than in the rate-control group (not statistically significant)

Conclusion from substudies • Maintenance of sinus rhythm is clearly a desired goal in the treatment of AF • Currently available antiarrhythmic drugs do not address this need due to their poor tolerability profiles – Of those currently available, amiodarone is the most effective • Implication: A rhythm-control strategy based on newer, more tolerable drugs will prove to be superior than simple rate control

Supporting evidence • The AFFIRM results are in general agreement with those of three other rhythm vs rate trials: – Pharmacologic Intervention in AF (PIAF) • Hohnloser SH, et al. Lancet 2000; 356: 1789 -94 – Rate Control versus Electrical Cardioversion for Persistent AF (RACE) • Van Gelder IC, et al. N Engl J Med 2002; 347: 1834 -40 – How to Treat Chronic AF (HOT CAFE) • Opolski G, et al. Chest 2004; 126: 476 -86

RAte Control vs Electrical cardioversion (RACE) Végpont Kombinált mortalitás és morbiditás Cardiovascularis mortalitás Frekvencia kontrol Ritmus kontrol 17. 2% 22. 6% NS 7. 0% 6. 7%


Overall conclusions of other rhythm vs rate trials • No differences between primary endpoints in two arms • All trials showed lower incidence of hospitalizations and adverse drug effects in the rate-control arm • PIAF and AFFIRM trials showed slightly better functional capacity in the rhythm control arm • Better exercise tolerance was achieved in the rhythm control groups of the PIAF, HOT CAFE, and AFFIRM trials

www. escardio. org/guidelines

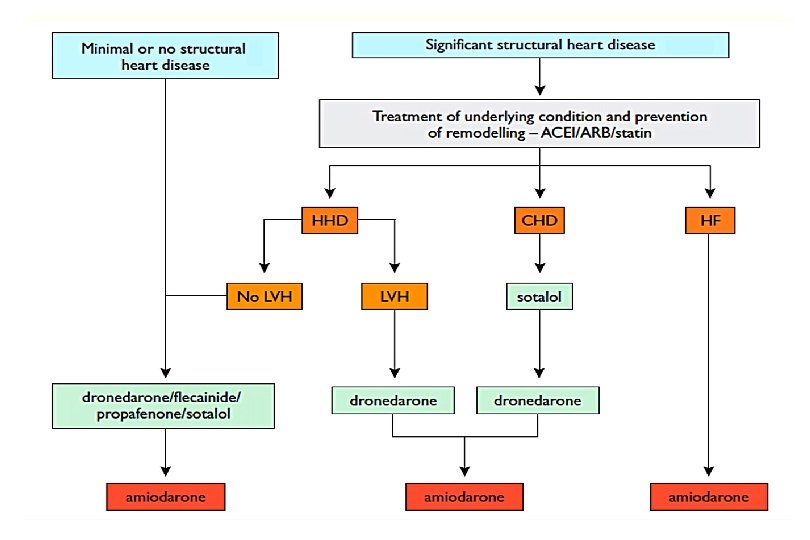
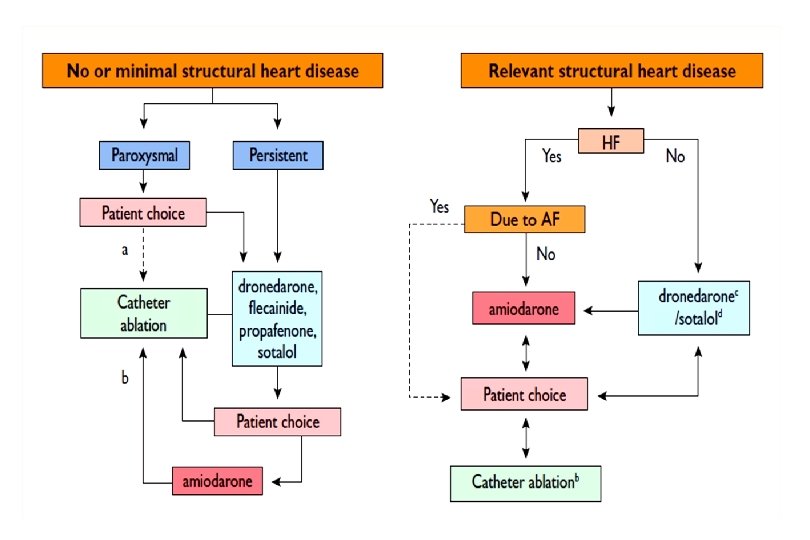
PF – frekvencia kontroll javasolt • BP nagyság > 50 mm • PF időtartam > 6 hónap • Szívelégtelenség > NYHA II • EF < 40% • Sinuscsomó betegség • Gyógyszer refrakteritás – KV > 3 • I, III intolerancia, proarrhythmia • Tünetmentesség • Proarrhythmia rizikó faktorok • Nincs anticoagulálás kontraindikáció • Öreg kor Carlsson et al. PACE 2000; 23: 891

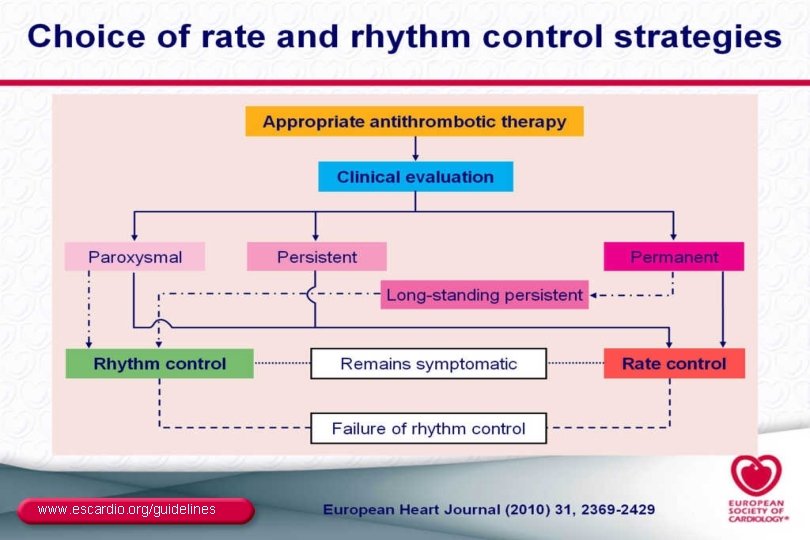
Kezelési stratégia – hogyan döntsünk? Ø A döntés legyen individuális Ø A fő szempont a beteg panasza Ø Befolyásoló tényezők: § alapbetegség § bal kamra funkció § beteg kora, terhelési kapacitása § beteg compliance § ritmuszavar természete (pl. lone típus) § korábbi kezelés eredményessége § kardioverziók száma

Ritmus kontroll • • • Egyéni döntés szükséges Újkeletű, vagy a PF első epizódja Fiatalabb betegek (< 65 év) Frekvencia kontrol ellenére fennálló tünetek Alapbetegség pl. HOCM, szívelégtelenség?

Ritmus kontroll Gyógyszerek

Antiarrhythmiás gyógyszerek Vaughan Williams osztályozás I. Osztály – Na – csatorna blokkolók A. Repolarizációt nyújtók: chinidin, disopyramid, procainamid B. Repolarizációt rövidítők: lidocain, mexiletin, tocainid, phenytoin C. Repolarizációt nem befolyásolják, ingerületvezetést rontják: flecainid, encainid, propafenon, moricizin, lorcainid, ajmalin II. Osztály – Béta – receptor blokkolók III. Osztály – Kálium – csatorna blokkolók: amiodaron, sotalol, bretylium, N-acetyl-procainamid, dofetilid, ibutilid IV. Osztály- Kálcium – csatorna blokkolók: verapamil, diltiazem Digitalis, adenosid
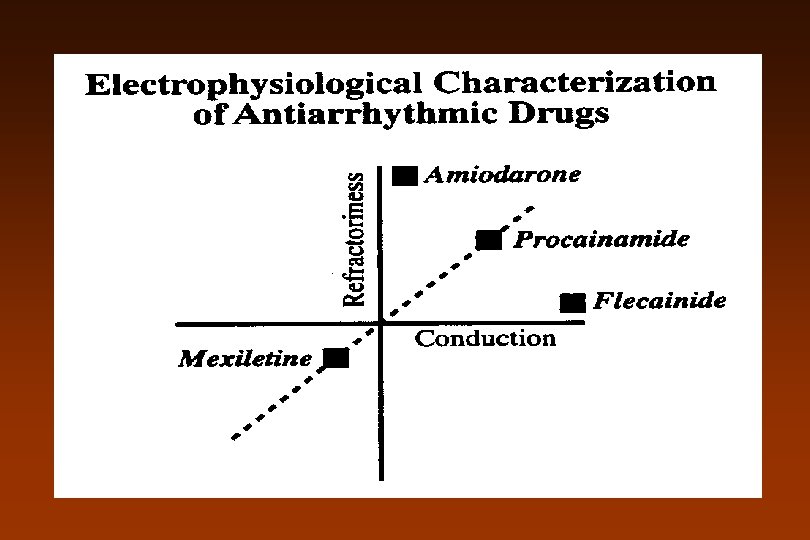
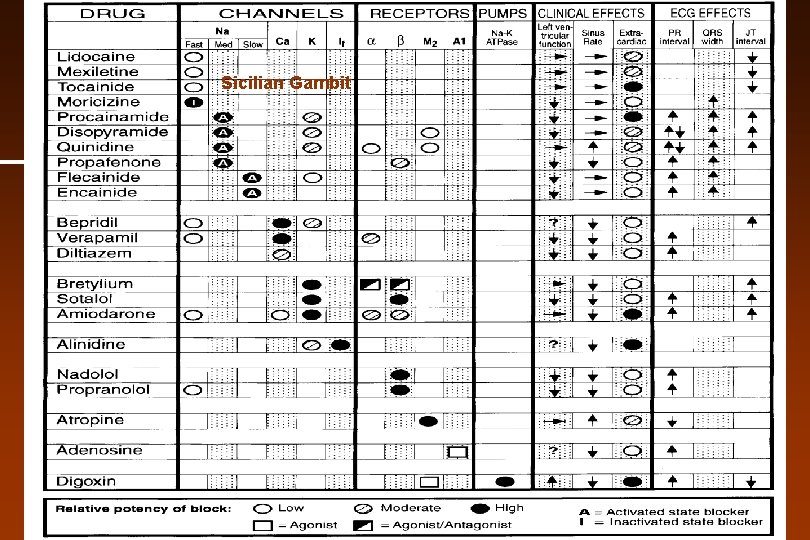
Sicilian Gambit
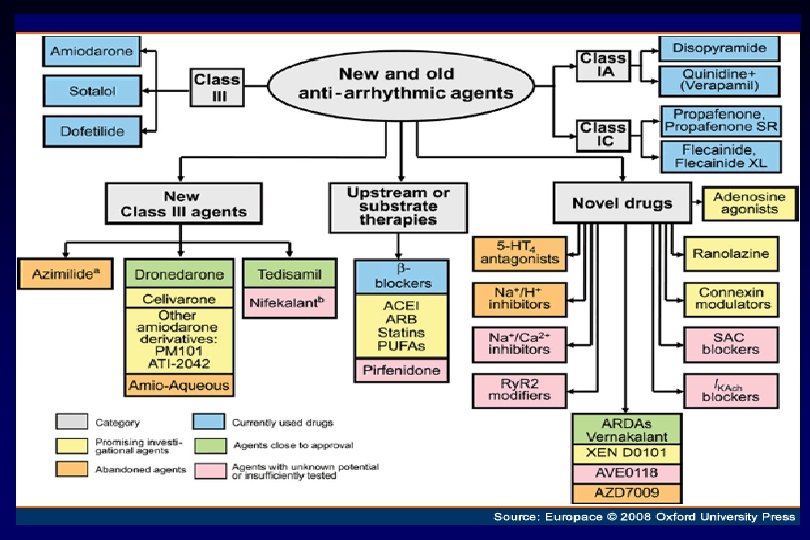
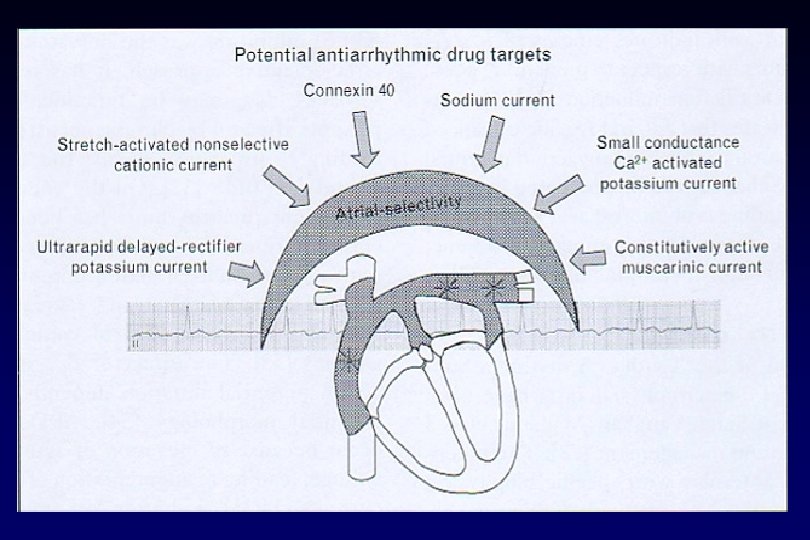

Új gyógyszerek v Dofetilid – SAFIRE –D trial – SR fenntartás 1 év: 58% vs placebo 25% - 70% -os konverziós ráta v Dronedarone – DAFNE – 800 mg/nap effektív és biztonságos a PF megelőzésében v Ibutilide – csak iv. – mitralis betegségben és nagy pitvar esetében – p. flattern konverzió v Azimilide – ASAP trial – hatásos SV tachycardiában 40% -al csökkenti a tünetmentes PF –t és P. flatternt v Tedisamil – pitvar – szelektív szer v RSD 1235 – Ito, IKur, Ina blockoló – csak pitvarban

www. escardio. org/guidelines
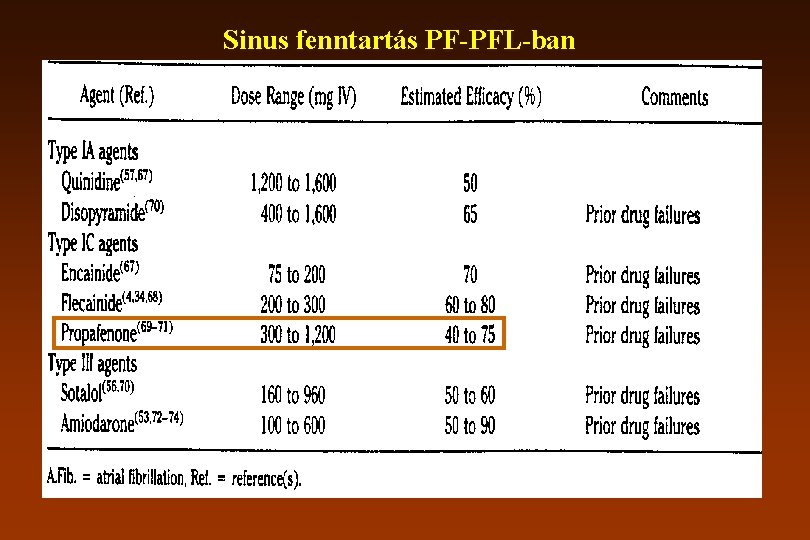
Sinus fenntartás PF-PFL-ban
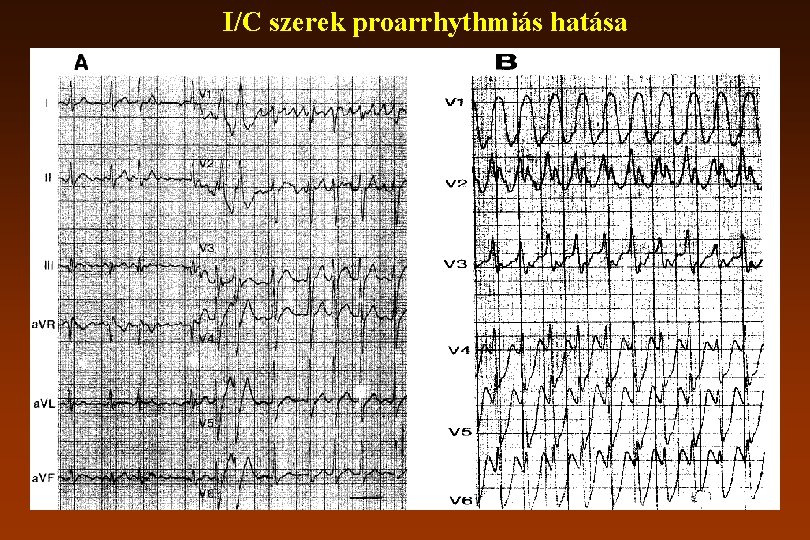
I/C szerek proarrhythmiás hatása
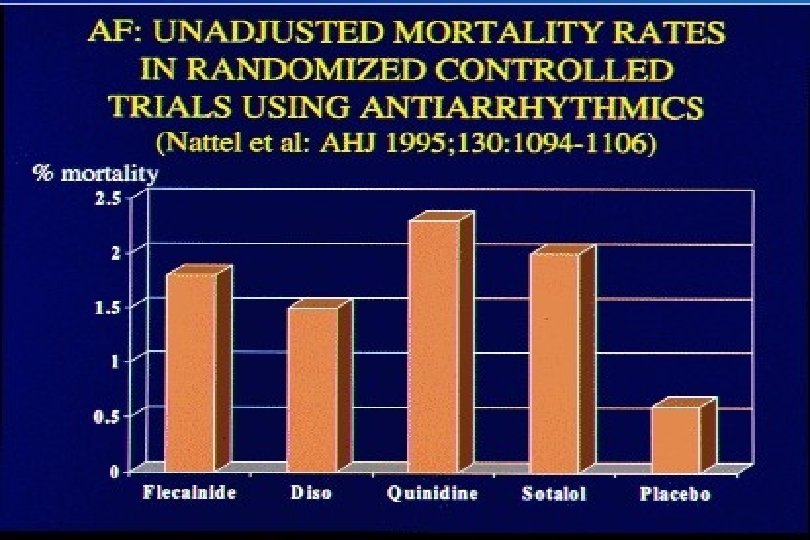
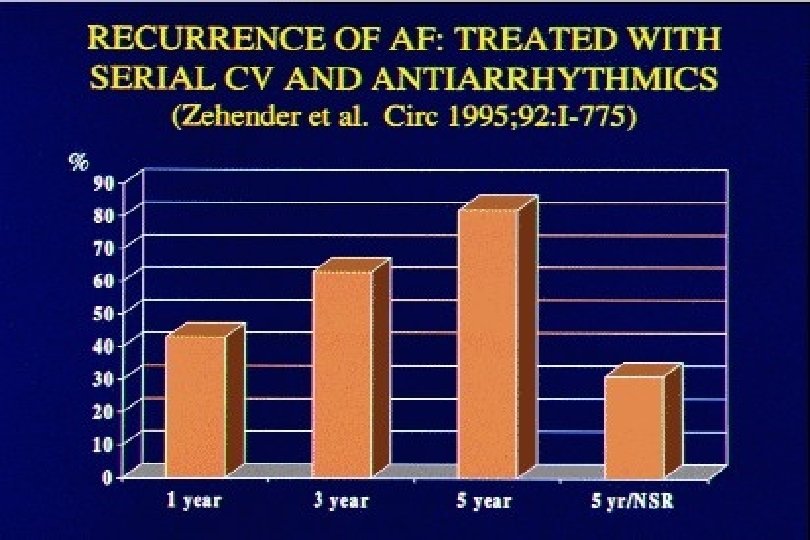
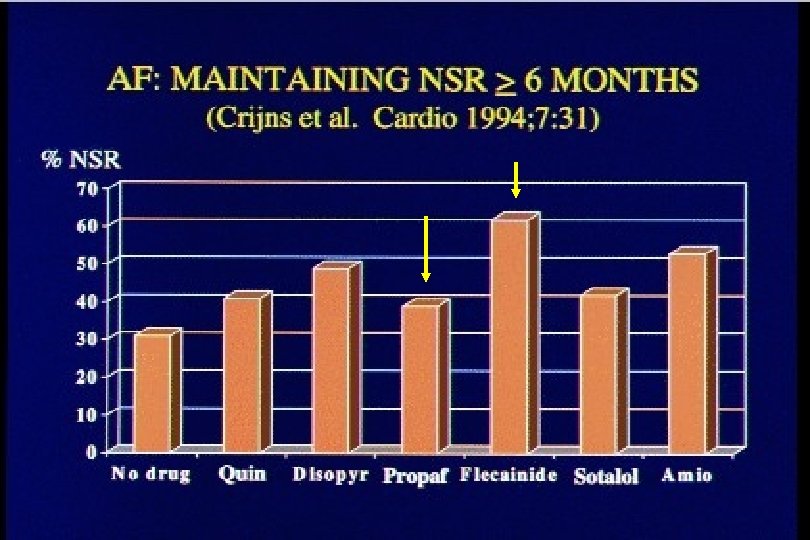
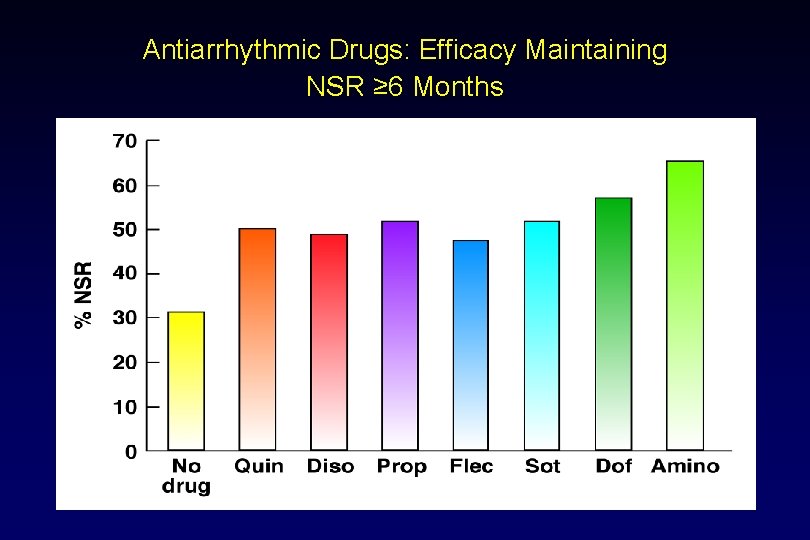
Antiarrhythmic Drugs: Efficacy Maintaining NSR ≥ 6 Months

Sinusritmus fenntartás Propafenon 3 hónap - 65 % sinus - UK PPF PSVT study 6 hónap - 136 beteg - 67 % sinus - placebo: 35% - Stroobandt, 1997) 2 hónap - 5 o % sinus - Pritchett, 1991 12 hónap Flec - 49 % sinus Placebo - 36 % sinus (van. Gelder, 1989) 12 hónap - 2 oo beteg Flec - 77 % sinus Propafenon - 75 % sinus (Chimenti, 1996)
www. escardio. org/guidelines

www. escardio. org/guidelines

www. escardio. org/guidelines
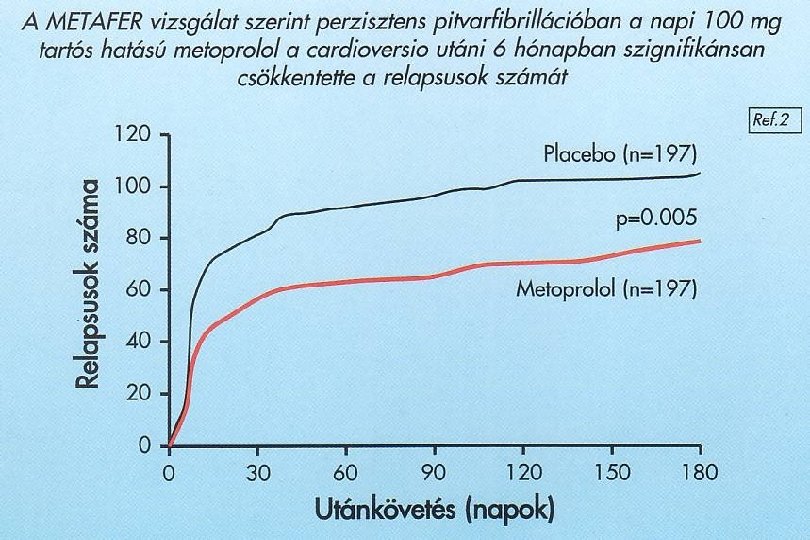

Sinus ritmus fenntartás
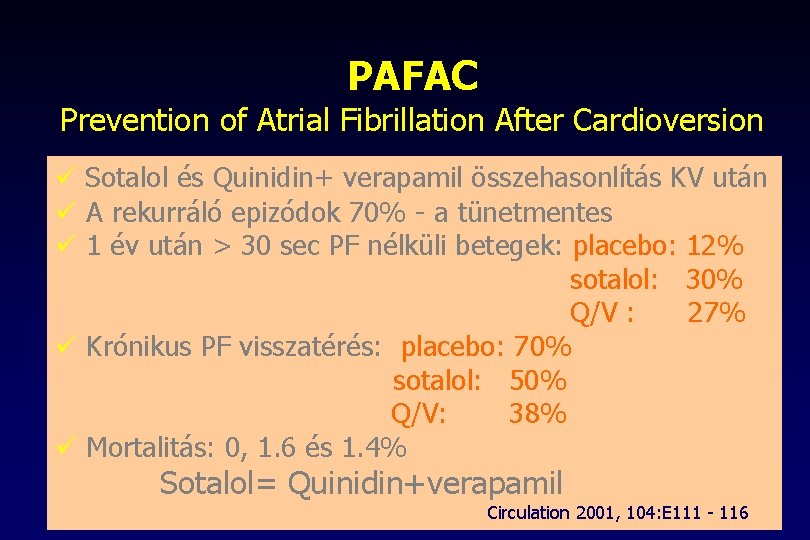
PAFAC Prevention of Atrial Fibrillation After Cardioversion ü Sotalol és Quinidin+ verapamil összehasonlítás KV után ü A rekurráló epizódok 70% - a tünetmentes ü 1 év után > 30 sec PF nélküli betegek: placebo: 12% sotalol: 30% Q/V : 27% ü Krónikus PF visszatérés: placebo: 70% sotalol: 50% Q/V: 38% ü Mortalitás: 0, 1. 6 és 1. 4% Sotalol= Quinidin+verapamil Circulation 2001, 104: E 111 - 116
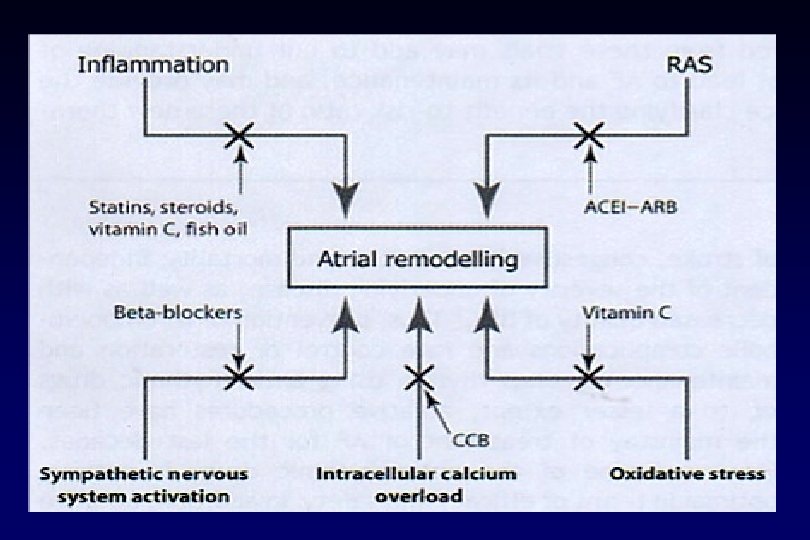

Renin-angiotensin –aldosterone rendszer - pitvarfibrillatio Ø Csökkenti az intracavitalis stresst • pitvari refrakteritás kedvező változása • korai utódepolarisatio csökken Ø Gátolja a myocardialis fibrosist • szubsztrát változás • impulzus propagatio Ø Megelőzi az apoptosist Ø Csökkenti a gyulladást Ø Csökkenti a hypertrophiát
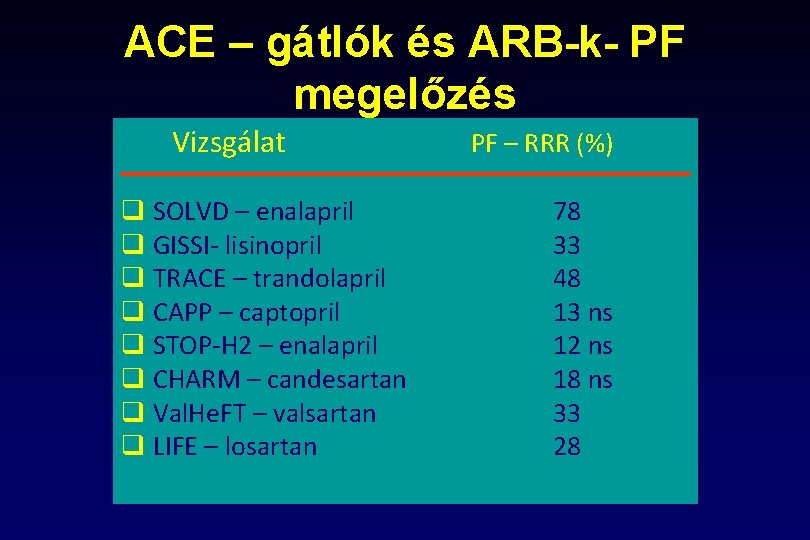
ACE – gátlók és ARB-k- PF megelőzés Vizsgálat q SOLVD – enalapril q GISSI- lisinopril q TRACE – trandolapril q CAPP – captopril q STOP-H 2 – enalapril q CHARM – candesartan q Val. He. FT – valsartan q LIFE – losartan PF – RRR (%) 78 33 48 13 ns 12 ns 18 ns 33 28
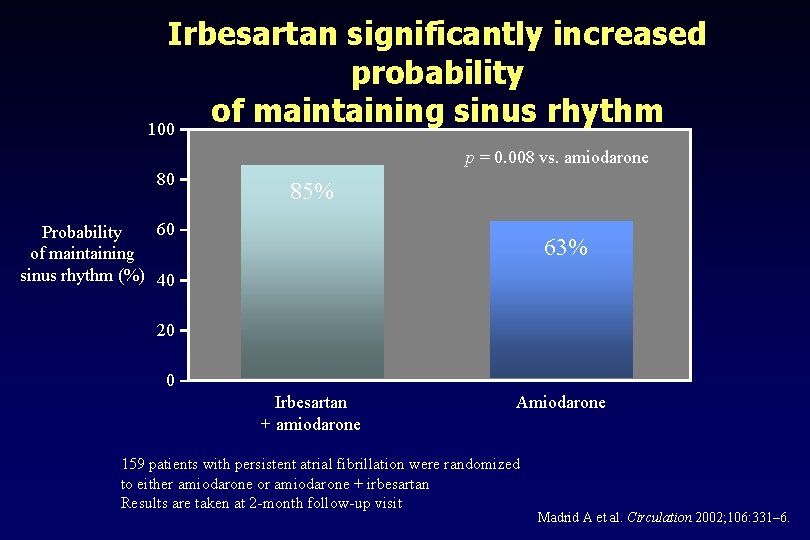
Irbesartan significantly increased probability of maintaining sinus rhythm 100 p = 0. 008 vs. amiodarone 80 85% 60 Probability of maintaining sinus rhythm (%) 40 63% 20 0 Irbesartan + amiodarone Amiodarone 159 patients with persistent atrial fibrillation were randomized to either amiodarone or amiodarone + irbesartan Results are taken at 2 -month follow-up visit Madrid A et al. Circulation 2002; 106: 331– 6.

ACEIs and ARBs reduce the risk of AF especially in patients with HF • 11 studies including 56, 308 patients – 4 heart failure – 3 hypertension – 2 after cardioversion for AF – 2 after MI • ACEIs and ARBs reduced risk of AF by 28% • No difference between the two drug classes • Risk reduction greatest in patients with HF (44%) • No significant risk reduction in hypertension, except for one trial with 29% reduction when LVH present LVH = left ventricular hypertrophy. Healey JS, et al. J Am Coll Cardiol. 2005; 45: 1832 -9.
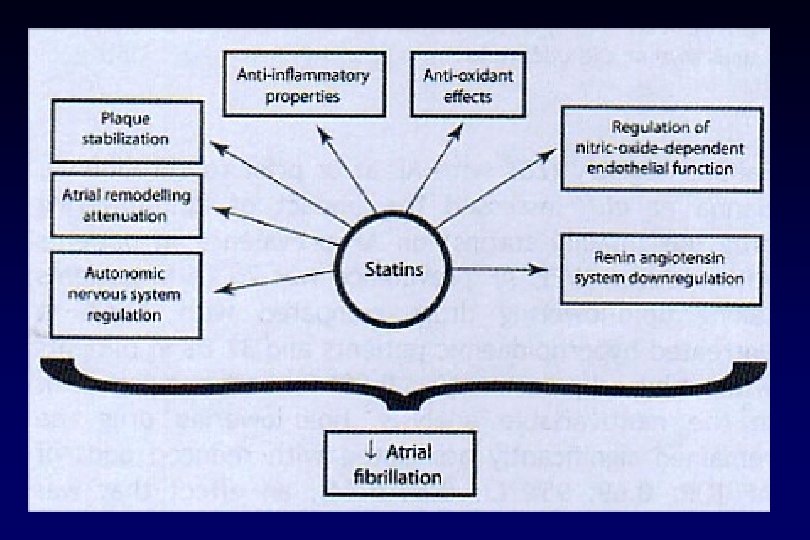
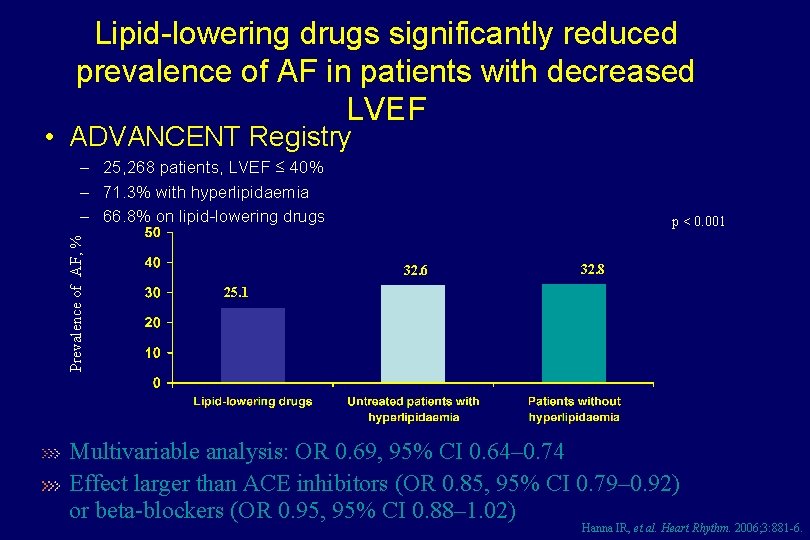
Lipid-lowering drugs significantly reduced prevalence of AF in patients with decreased LVEF • ADVANCENT Registry Prevalence of AF, % – 25, 268 patients, LVEF ≤ 40% – 71. 3% with hyperlipidaemia – 66. 8% on lipid-lowering drugs p < 0. 001 32. 6 32. 8 25. 1 Multivariable analysis: OR 0. 69, 95% CI 0. 64– 0. 74 Effect larger than ACE inhibitors (OR 0. 85, 95% CI 0. 79– 0. 92) or beta-blockers (OR 0. 95, 95% CI 0. 88– 1. 02) Hanna IR, et al. Heart Rhythm. 2006; 3: 881 -6.

„Upstream” kezelés – primer prevenció • ACEi és ARB újkeletű PF prevenciójára szívelégtelenségben és csökkent EF esetén – IIa. B • ACEi és ARB – prevenció – hypertonia, főleg BKH IIa. B • Statin – prevenció- CABG műtét után -IIa. B • Statin- prevenció – szívbetegség, főleg szívelégtelenség- IIb. B • ACEi, ARB és statin primer prevencióra nem ajánlott KV betegség nélkül – IIIC Eur Heart J 2010; 31: 2369 - 2429

www. escardio. org/guidelines

2014 AHA/ACC/HRS Guideline for the Management of Patients With Atrial Fibrillation: www. escardio. org/guidelines
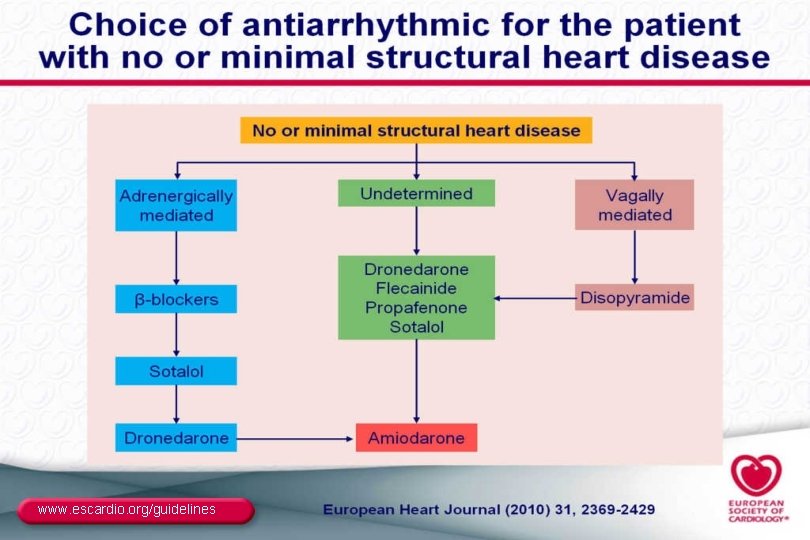
www. escardio. org/guidelines
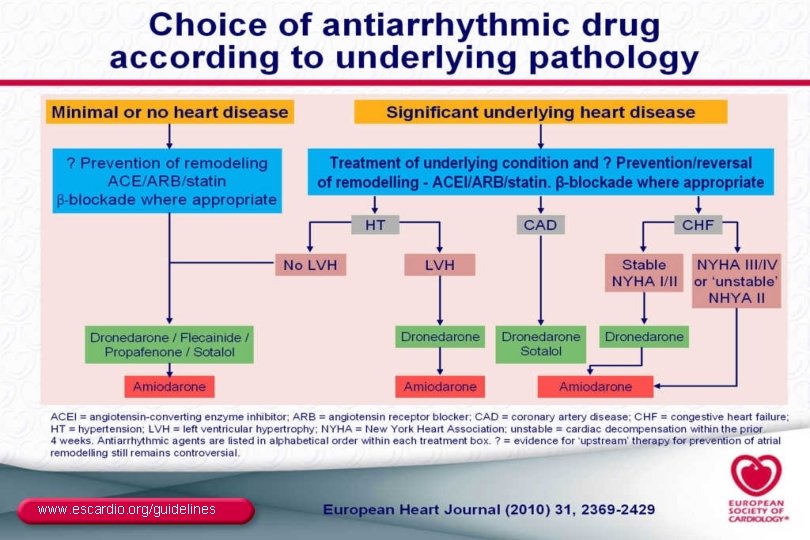
www. escardio. org/guidelines
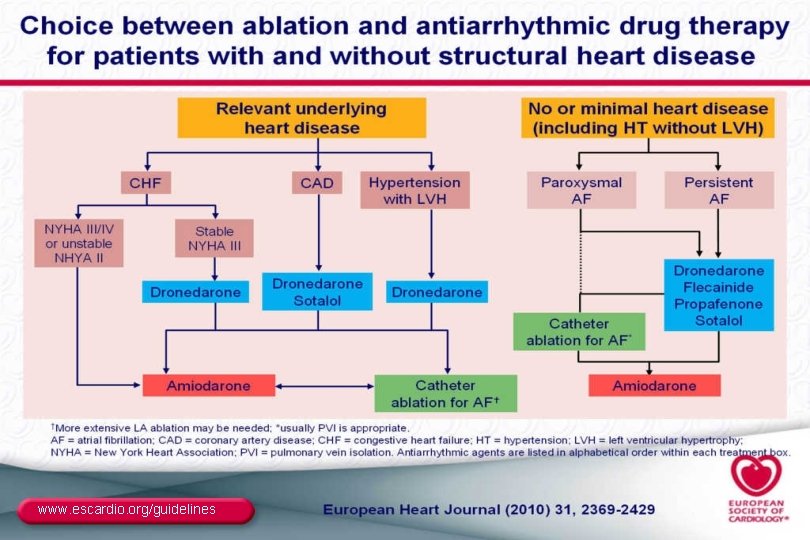
www. escardio. org/guidelines
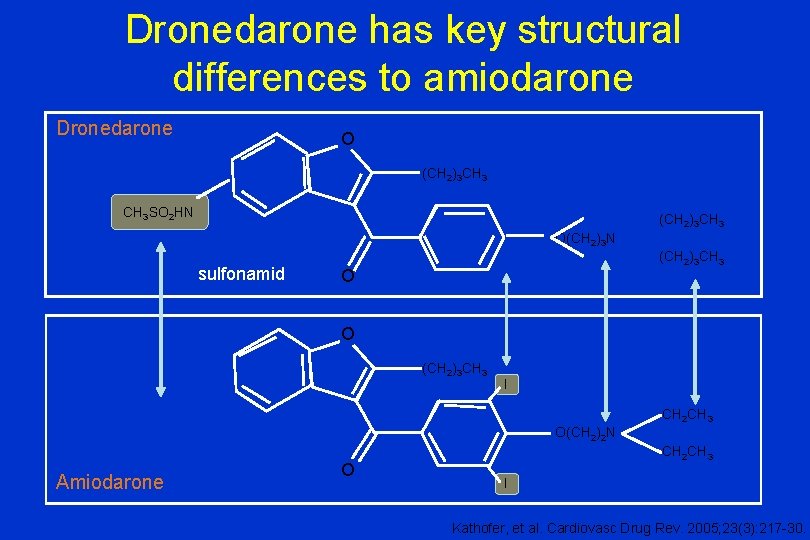
Dronedarone has key structural differences to amiodarone Dronedarone O (CH 2)3 CH 3 SO 2 HN (CH 2)3 CH 3 O(CH 2)3 N sulfonamid (CH 2)3 CH 3 O O (CH 2)3 CH 3 I CH 2 CH 3 O(CH 2)2 N Amiodarone O CH 2 CH 3 I Kathofer, et al. Cardiovasc Drug Rev. 2005; 23(3): 217 -30.


Dronedarone ü Nincs reverse-use dependens hatása az AP-ra ü Csökkenti a kamrai repolarizáció transmurális disperzióját ü III-as osztályú szerek okozta korai utódepolarizáció ellen véd ü Nem indukál torsades de pointes kamrai arrhythmiát ü Nincs szerv-toxikus hatása
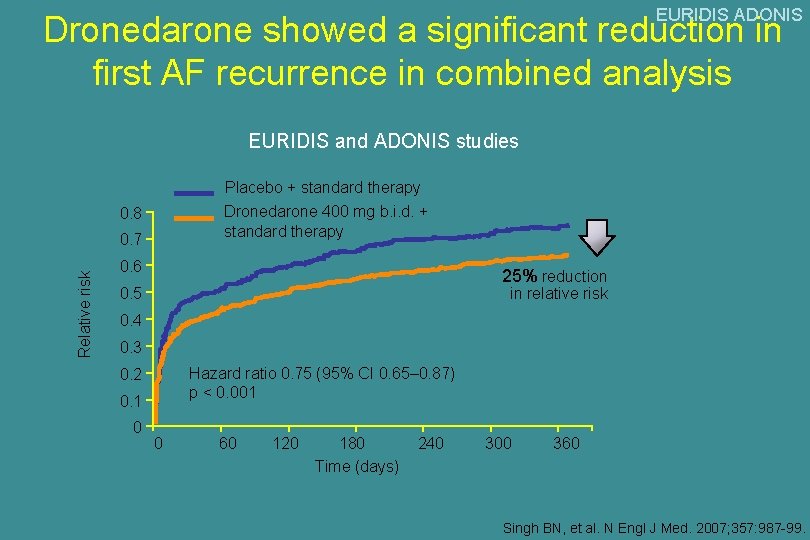
EURIDIS ADONIS Dronedarone showed a significant reduction in first AF recurrence in combined analysis EURIDIS and ADONIS studies Placebo + standard therapy Dronedarone 400 mg b. i. d. + standard therapy 0. 8 Relative risk 0. 7 0. 6 25% reduction 0. 5 in relative risk 0. 4 0. 3 Hazard ratio 0. 75 (95% CI 0. 65– 0. 87) p < 0. 001 0. 2 0. 1 0 0 60 120 180 Time (days) 240 300 360 Singh BN, et al. N Engl J Med. 2007; 357: 987 -99.
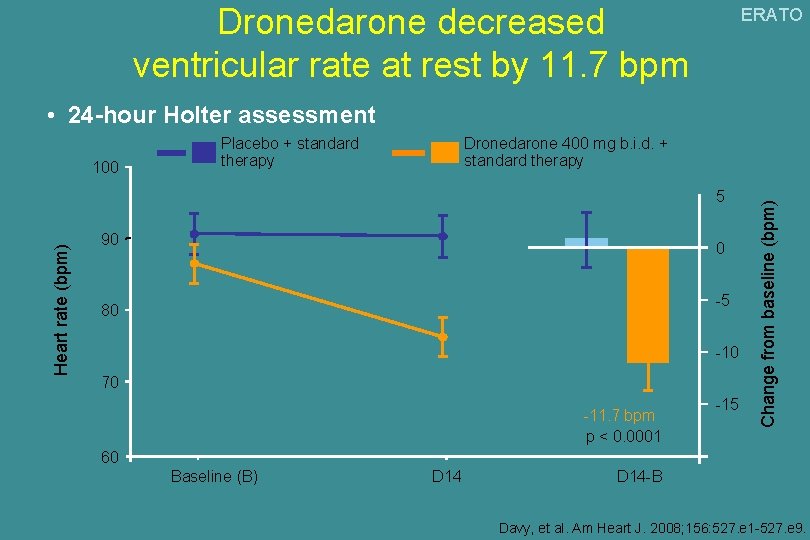
Dronedarone decreased ventricular rate at rest by 11. 7 bpm ERATO • 24 -hour Holter assessment Dronedarone 400 mg b. i. d. + standard therapy Heart rate (bpm) 5 90 0 -5 80 -10 70 -11. 7 bpm p < 0. 0001 -15 Change from baseline (bpm) 100 Placebo + standard therapy 60 Baseline (B) D 14 -B Davy, et al. Am Heart J. 2008; 156: 527. e 1 -527. e 9.
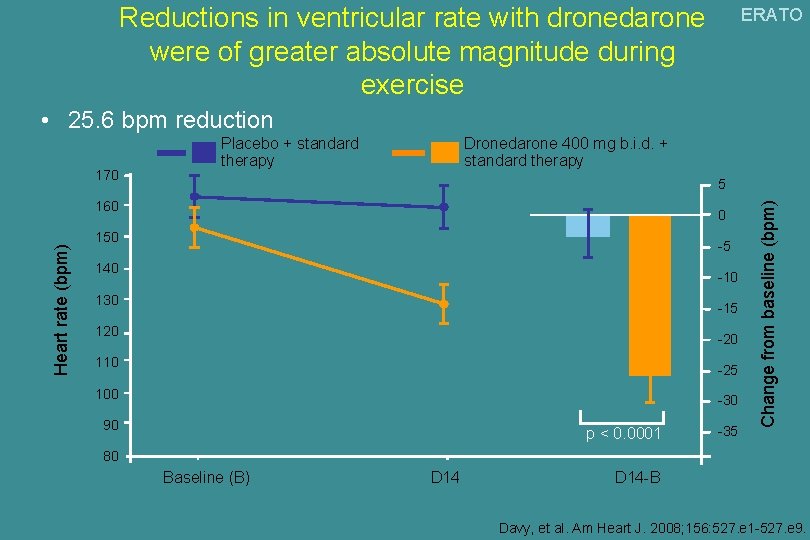
Reductions in ventricular rate with dronedarone were of greater absolute magnitude during exercise ERATO • 25. 6 bpm reduction Dronedarone 400 mg b. i. d. + standard therapy 5 Heart rate (bpm) 160 0 150 -5 140 -10 130 -15 120 -20 110 -25 100 -30 90 p < 0. 0001 -35 Change from baseline (bpm) 170 Placebo + standard therapy 80 Baseline (B) D 14 -B Davy, et al. Am Heart J. 2008; 156: 527. e 1 -527. e 9.
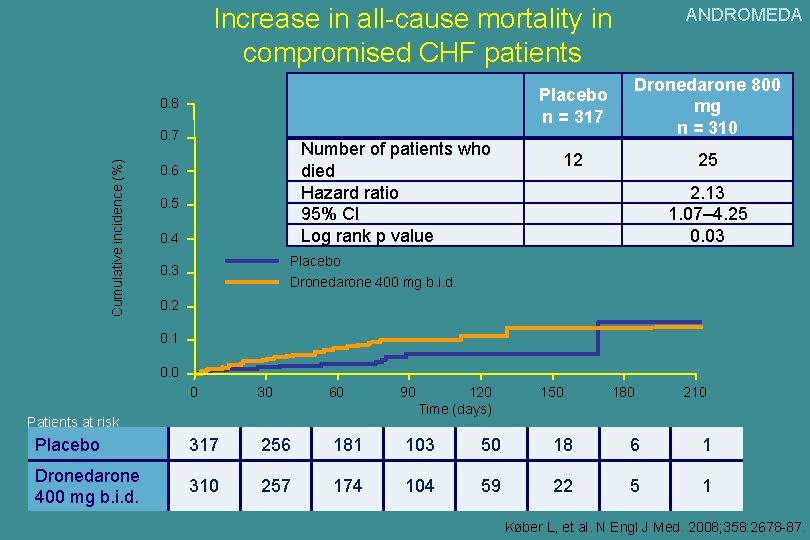
Increase in all-cause mortality in compromised CHF patients 0. 8 Cumulative incidence (%) 0. 7 Number of patients who died Hazard ratio 95% CI Log rank p value 0. 6 0. 5 0. 4 ANDROMEDA Placebo n = 317 Dronedarone 800 mg n = 310 12 25 2. 13 1. 07– 4. 25 0. 03 Placebo Dronedarone 400 mg b. i. d. 0. 3 0. 2 0. 1 0. 0 0 30 60 Placebo 317 256 181 103 50 18 6 1 Dronedarone 400 mg b. i. d. 310 257 174 104 59 22 5 1 Patients at risk 90 120 Time (days) 150 180 210 Køber L, et al. N Engl J Med. 2008; 358: 2678 -87.
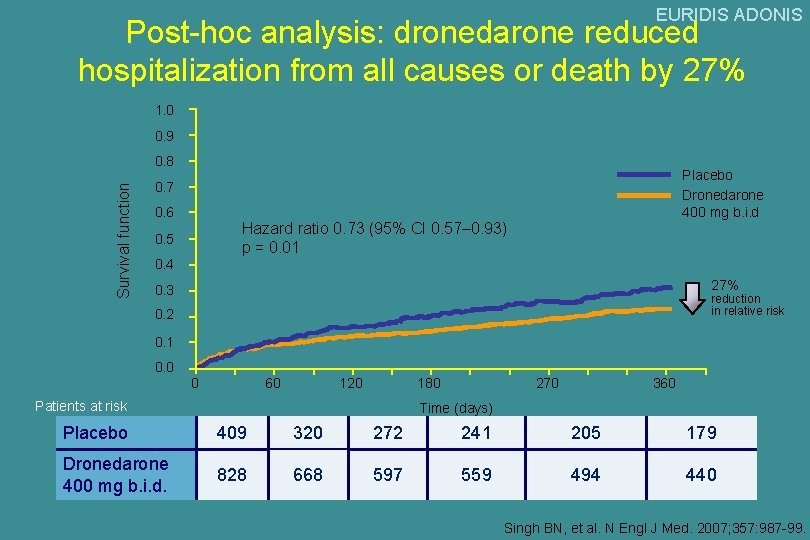
EURIDIS ADONIS Post-hoc analysis: dronedarone reduced hospitalization from all causes or death by 27% 1. 0 0. 9 Survival function 0. 8 Placebo Dronedarone 400 mg b. i. d 0. 7 0. 6 Hazard ratio 0. 73 (95% CI 0. 57– 0. 93) p = 0. 01 0. 5 0. 4 27% 0. 3 reduction in relative risk 0. 2 0. 1 0. 0 0 60 120 180 Patients at risk 270 360 Time (days) Placebo 409 320 272 241 205 179 Dronedarone 400 mg b. i. d. 828 668 597 559 494 440 Singh BN, et al. N Engl J Med. 2007; 357: 987 -99.
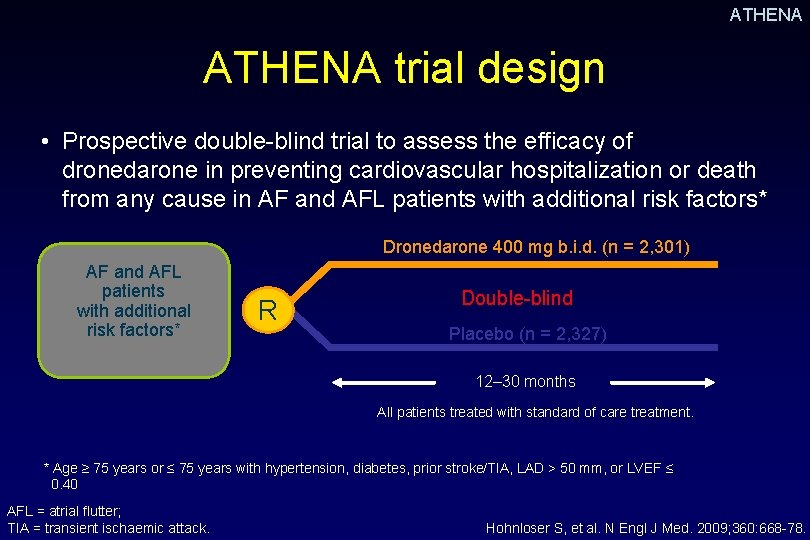
ATHENA trial design • Prospective double-blind trial to assess the efficacy of dronedarone in preventing cardiovascular hospitalization or death from any cause in AF and AFL patients with additional risk factors* Dronedarone 400 mg b. i. d. (n = 2, 301) AF and AFL patients with additional risk factors* R Double-blind Placebo (n = 2, 327) 12– 30 months All patients treated with standard of care treatment. * Age ≥ 75 years or ≤ 75 years with hypertension, diabetes, prior stroke/TIA, LAD > 50 mm, or LVEF ≤ 0. 40 AFL = atrial flutter; TIA = transient ischaemic attack. Hohnloser S, et al. N Engl J Med. 2009; 360: 668 -78.

Study end-points • Primary end-point – First cardiovascular hospitalization or death • Secondary end-points – Death – Cardiovascular death – Cardiovascular hospitalization Hohnloser S, et al. N Engl J Med. 2009; 360: 668 -78.

ATHENA Inclusion and Exclusion Criteria Inclusion criteria • High-risk patients with a history of paroxysmal or persistent AF/AFL • Aged ≥ 75 years with or without additional risk factors • Aged ≥ 70 years and ≥ 1 risk factor (hypertension; diabetes; prior stroke/TIA; LA ≥ 50 mm; LVEF <0. 40) Exclusion criteria • Permanent AF • Unstable hemodynamic situation recently decompensated CHF) (i. e. • CHF NYHA class IV • Bradycardia <50 bpm and/or PR >0. 28 sec • Sick sinus syndrome • Calculated GFR at baseline <10 ml/min • Originally the protocol had allowed patients <70 years of age with additional risk factors into the study • The protocol was subsequently amended to include only patients ≥ 70 years of age • Potassium <3. 5 mmol/L • Concomitant antiarrhythmic drug Rx • Severe illness limiting life expectancy • Pregnancy or breastfeeding • Refusal or inability to give informed consent Hohnloser SH, et al. J Cardiovasc Electrophysiol 2008; 19: 69 -73.
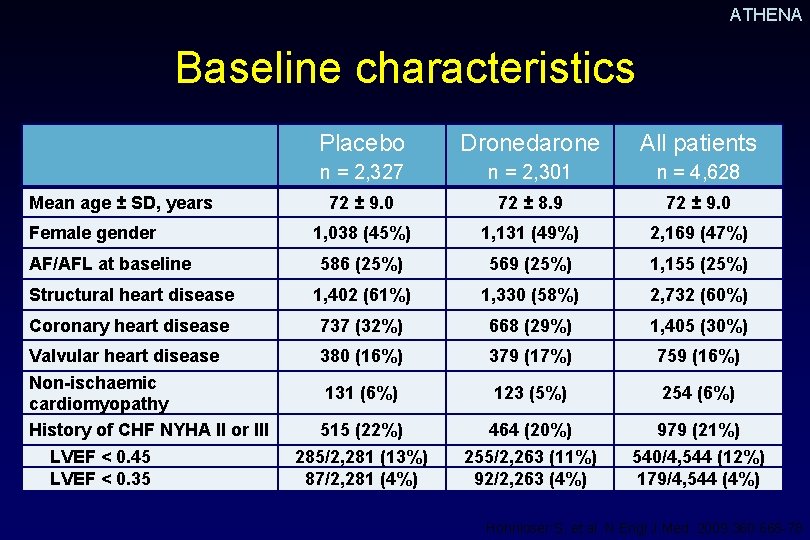
ATHENA Baseline characteristics Placebo Dronedarone All patients n = 2, 327 n = 2, 301 n = 4, 628 72 ± 9. 0 72 ± 8. 9 72 ± 9. 0 1, 038 (45%) 1, 131 (49%) 2, 169 (47%) 586 (25%) 569 (25%) 1, 155 (25%) Structural heart disease 1, 402 (61%) 1, 330 (58%) 2, 732 (60%) Coronary heart disease 737 (32%) 668 (29%) 1, 405 (30%) Valvular heart disease Non-ischaemic cardiomyopathy History of CHF NYHA II or III LVEF < 0. 45 LVEF < 0. 35 380 (16%) 379 (17%) 759 (16%) 131 (6%) 123 (5%) 254 (6%) 515 (22%) 285/2, 281 (13%) 87/2, 281 (4%) 464 (20%) 255/2, 263 (11%) 92/2, 263 (4%) 979 (21%) 540/4, 544 (12%) 179/4, 544 (4%) Mean age ± SD, years Female gender AF/AFL at baseline Hohnloser S, et al. N Engl J Med. 2009; 360: 668 -78.
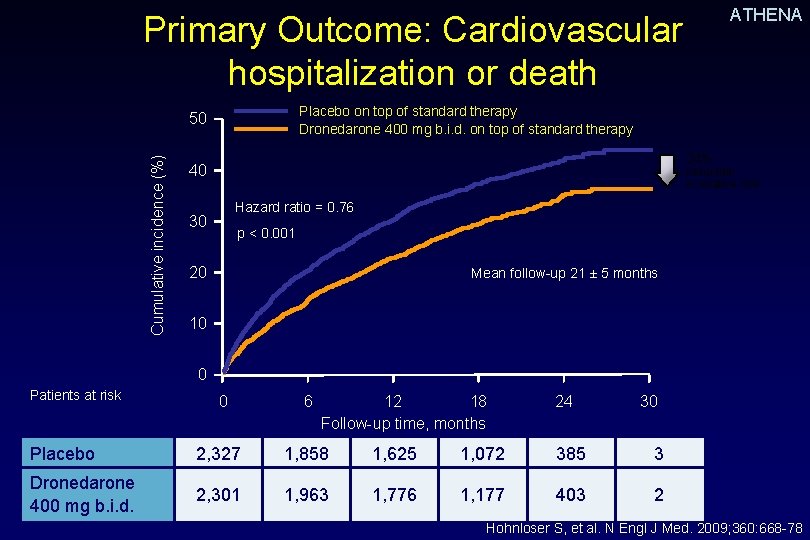
ATHENA Primary Outcome: Cardiovascular hospitalization or death Placebo on top of standard therapy Dronedarone 400 mg b. i. d. on top of standard therapy Cumulative incidence (%) 50 24% 40 reduction in relative risk Hazard ratio = 0. 76 30 p < 0. 001 20 Mean follow-up 21 ± 5 months 10 0 Patients at risk 0 6 12 18 Follow-up time, months 24 30 Placebo 2, 327 1, 858 1, 625 1, 072 385 3 Dronedarone 400 mg b. i. d. 2, 301 1, 963 1, 776 1, 177 403 2 Hohnloser S, et al. N Engl J Med. 2009; 360: 668 -78.
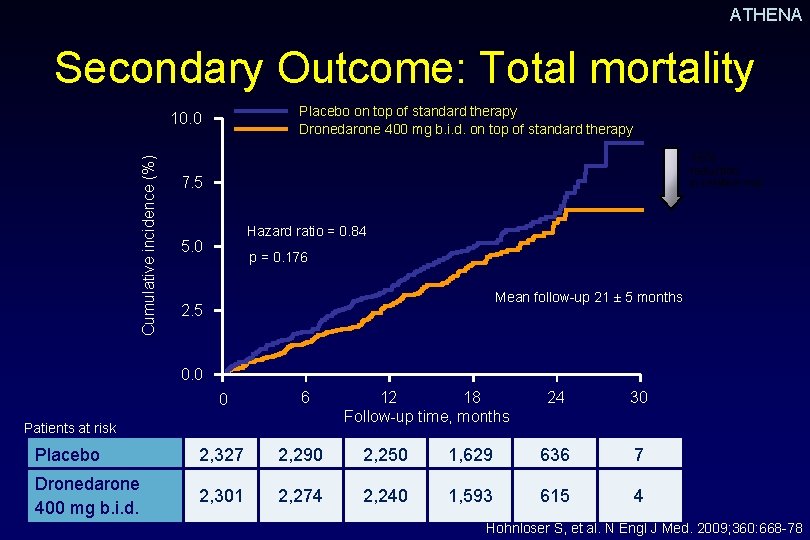
ATHENA Secondary Outcome: Total mortality Placebo on top of standard therapy Dronedarone 400 mg b. i. d. on top of standard therapy Cumulative incidence (%) 10. 0 16% reduction in relative risk 7. 5 Hazard ratio = 0. 84 5. 0 p = 0. 176 Mean follow-up 21 ± 5 months 2. 5 0. 0 0 6 Patients at risk 12 18 Follow-up time, months 24 30 Placebo 2, 327 2, 290 2, 250 1, 629 636 7 Dronedarone 400 mg b. i. d. 2, 301 2, 274 2, 240 1, 593 615 4 Hohnloser S, et al. N Engl J Med. 2009; 360: 668 -78.
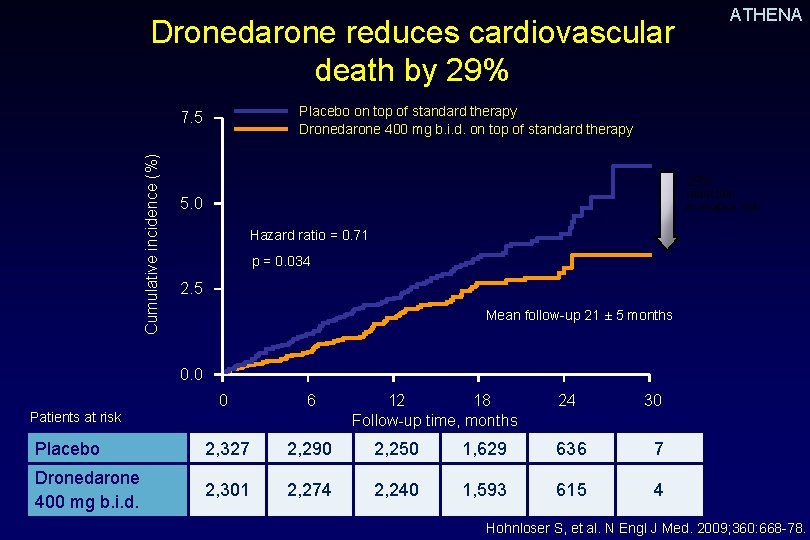
ATHENA Dronedarone reduces cardiovascular death by 29% Placebo on top of standard therapy Dronedarone 400 mg b. i. d. on top of standard therapy Cumulative incidence (%) 7. 5 29% reduction in relative risk 5. 0 Hazard ratio = 0. 71 p = 0. 034 2. 5 Mean follow-up 21 ± 5 months 0. 0 0 6 Placebo 2, 327 2, 290 2, 250 1, 629 636 7 Dronedarone 400 mg b. i. d. 2, 301 2, 274 2, 240 1, 593 615 4 Patients at risk 12 18 Follow-up time, months 24 30 Hohnloser S, et al. N Engl J Med. 2009; 360: 668 -78.
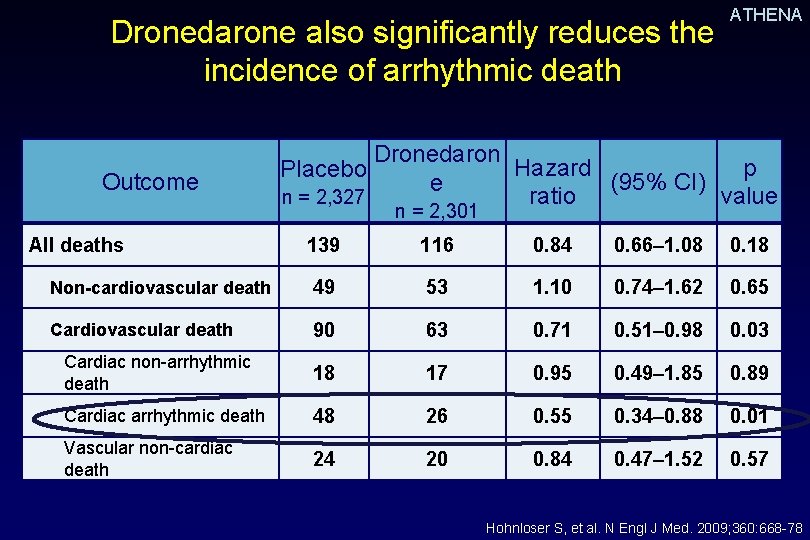
Dronedarone also significantly reduces the incidence of arrhythmic death Outcome ATHENA Dronedaron Hazard p Placebo (95% CI) e n = 2, 327 ratio value n = 2, 301 All deaths 139 116 0. 84 0. 66– 1. 08 0. 18 Non-cardiovascular death 49 53 1. 10 0. 74– 1. 62 0. 65 Cardiovascular death 90 63 0. 71 0. 51– 0. 98 0. 03 Cardiac non-arrhythmic death 18 17 0. 95 0. 49– 1. 85 0. 89 Cardiac arrhythmic death 48 26 0. 55 0. 34– 0. 88 0. 01 Vascular non-cardiac death 24 20 0. 84 0. 47– 1. 52 0. 57 Hohnloser S, et al. N Engl J Med. 2009; 360: 668 -78.
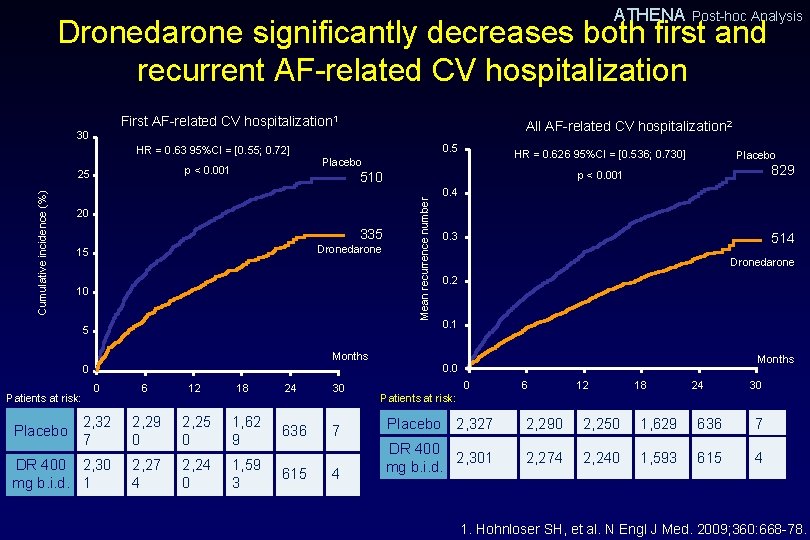
ATHENA Post-hoc Analysis Dronedarone significantly decreases both first and recurrent AF-related CV hospitalization First AF-related CV hospitalization 1 30 All AF-related CV hospitalization 2 0. 5 HR = 0. 63 95%CI = [0. 55; 0. 72] p < 0. 001 20 335 Dronedarone 15 10 5 Months 0 0 6 12 2, 32 7 2, 29 0 2, 25 0 1, 62 9 DR 400 2, 30 mg b. i. d. 1 2, 27 4 2, 24 0 1, 59 3 Patients at risk: Placebo 18 24 30 636 7 615 4 Placebo 829 p < 0. 001 510 Mean recurrence number Cumulative incidence (%) 25 HR = 0. 626 95%CI = [0. 536; 0. 730] Placebo 0. 4 0. 3 514 Dronedarone 0. 2 0. 1 Months 0. 0 0 6 12 18 24 30 Patients at risk: Placebo 2, 327 2, 290 2, 250 1, 629 636 7 DR 400 2, 301 mg b. i. d. 2, 274 2, 240 1, 593 615 4 1. Hohnloser SH, et al. N Engl J Med. 2009; 360: 668 -78.
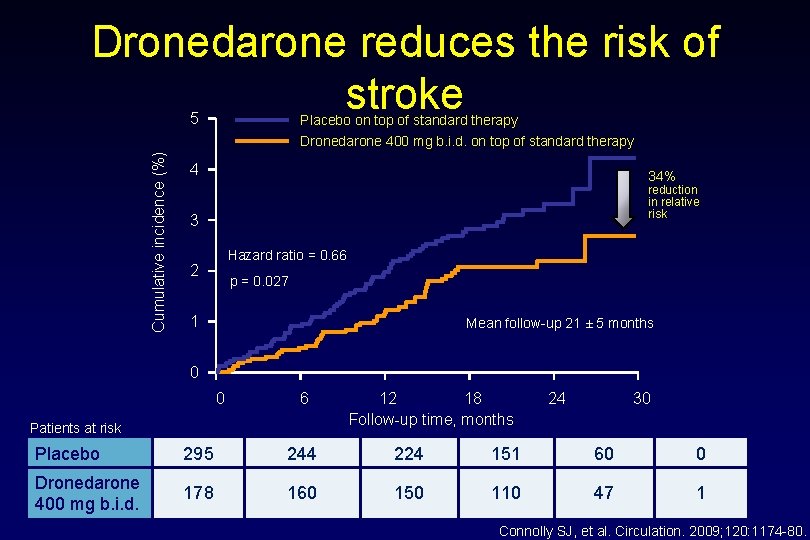
Dronedarone reduces the risk of stroke Cumulative incidence (%) 5 Placebo on top of standard therapy Dronedarone 400 mg b. i. d. on top of standard therapy 4 34% reduction in relative risk 3 Hazard ratio = 0. 66 2 p = 0. 027 1 Mean follow-up 21 ± 5 months 0 0 6 Patients at risk 12 18 Follow-up time, months 24 30 Placebo 295 244 224 151 60 0 Dronedarone 400 mg b. i. d. 178 160 150 110 47 1 Connolly SJ, et al. Circulation. 2009; 120: 1174 -80.
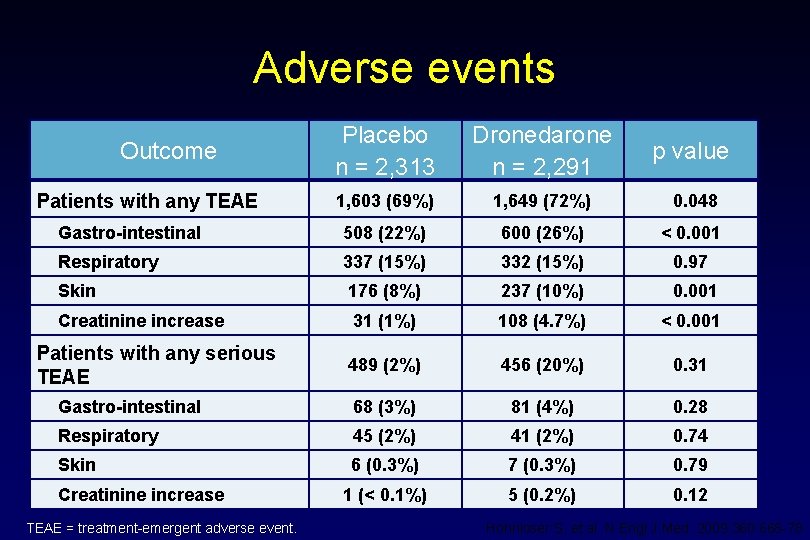
Adverse events Placebo n = 2, 313 Dronedarone n = 2, 291 p value 1, 603 (69%) 1, 649 (72%) 0. 048 Gastro-intestinal 508 (22%) 600 (26%) < 0. 001 Respiratory 337 (15%) 332 (15%) 0. 97 Skin 176 (8%) 237 (10%) 0. 001 Creatinine increase 31 (1%) 108 (4. 7%) < 0. 001 489 (2%) 456 (20%) 0. 31 Gastro-intestinal 68 (3%) 81 (4%) 0. 28 Respiratory 45 (2%) 41 (2%) 0. 74 Skin 6 (0. 3%) 7 (0. 3%) 0. 79 1 (< 0. 1%) 5 (0. 2%) 0. 12 Outcome Patients with any TEAE Patients with any serious TEAE Creatinine increase TEAE = treatment-emergent adverse event. Hohnloser S, et al. N Engl J Med. 2009; 360: 668 -78.
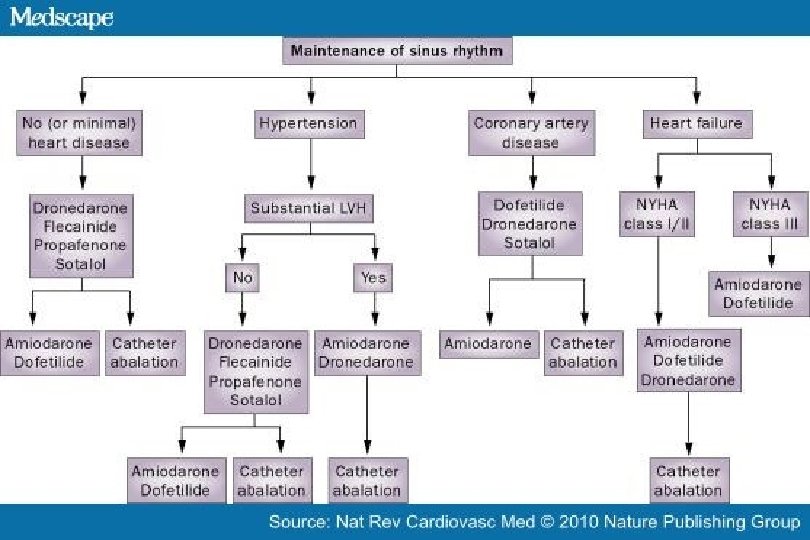
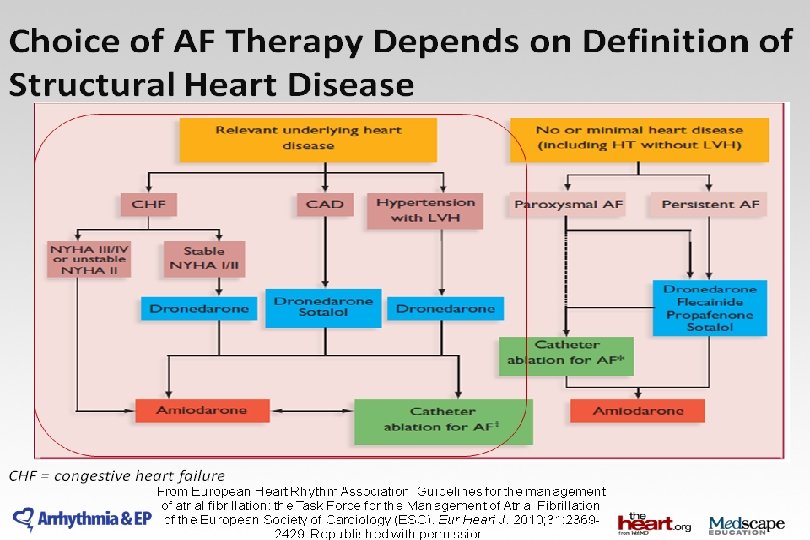
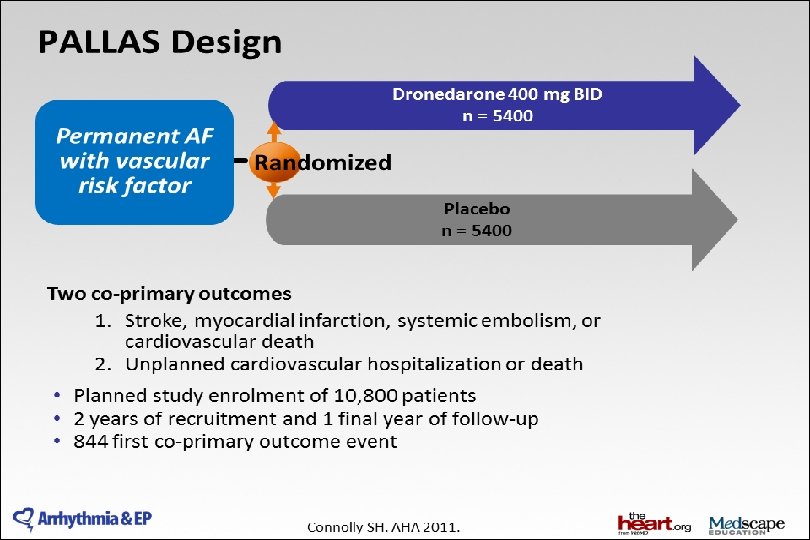
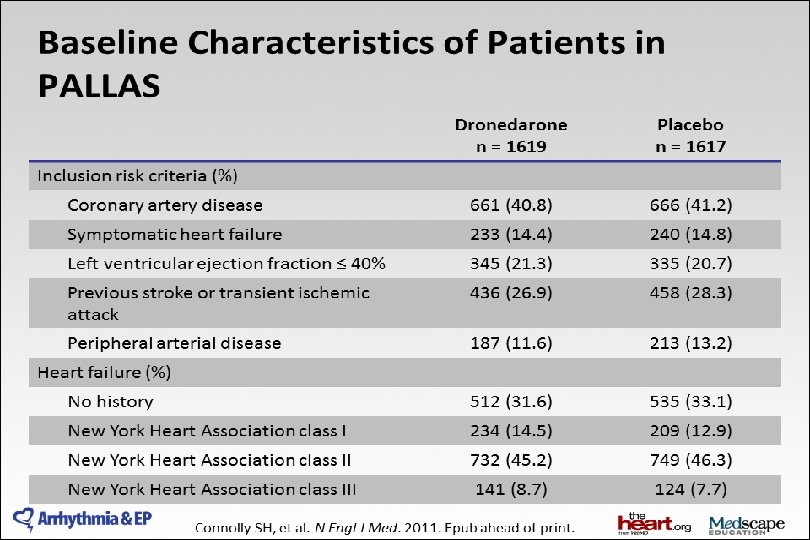
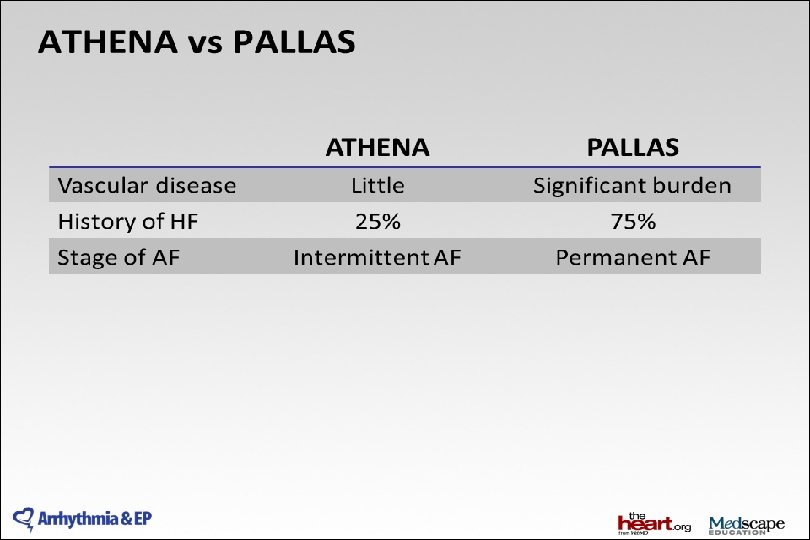
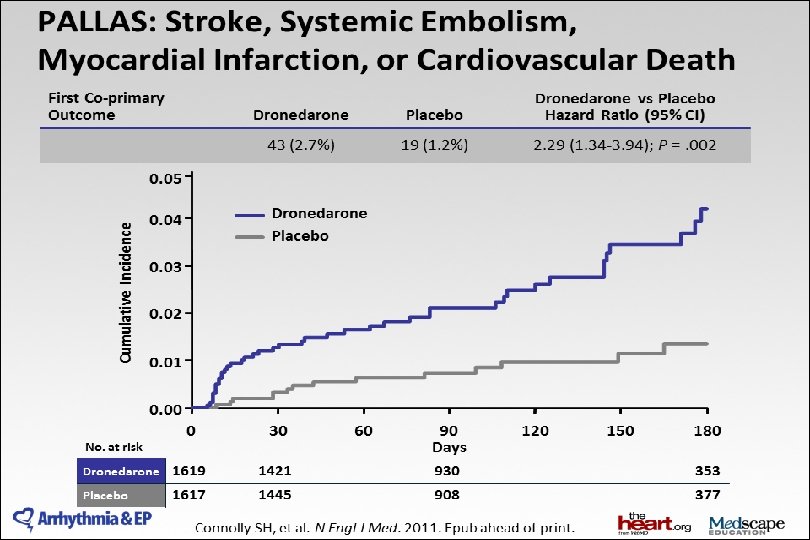
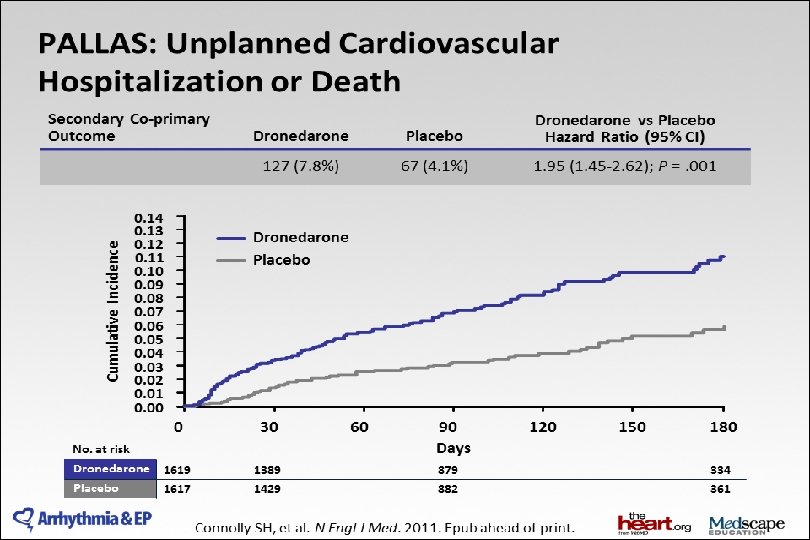
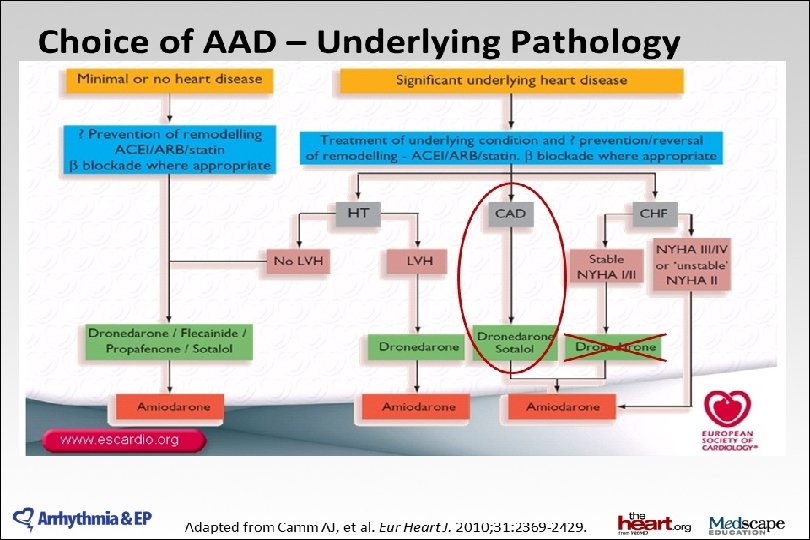
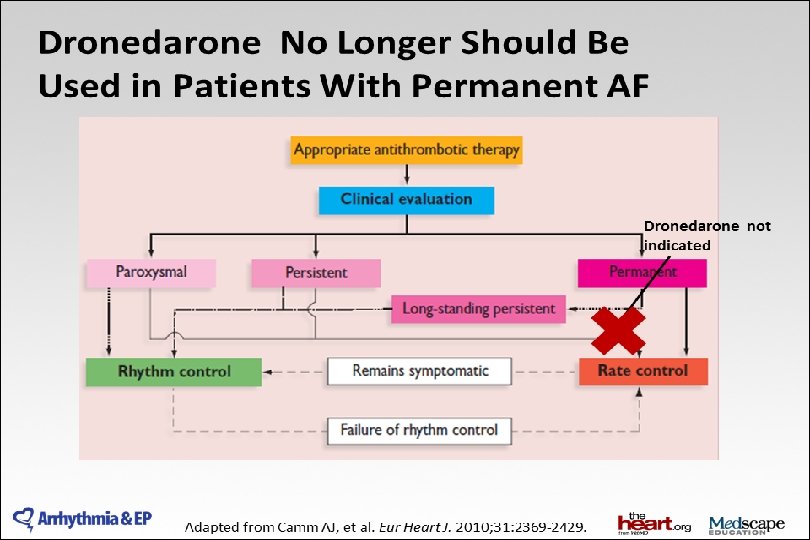

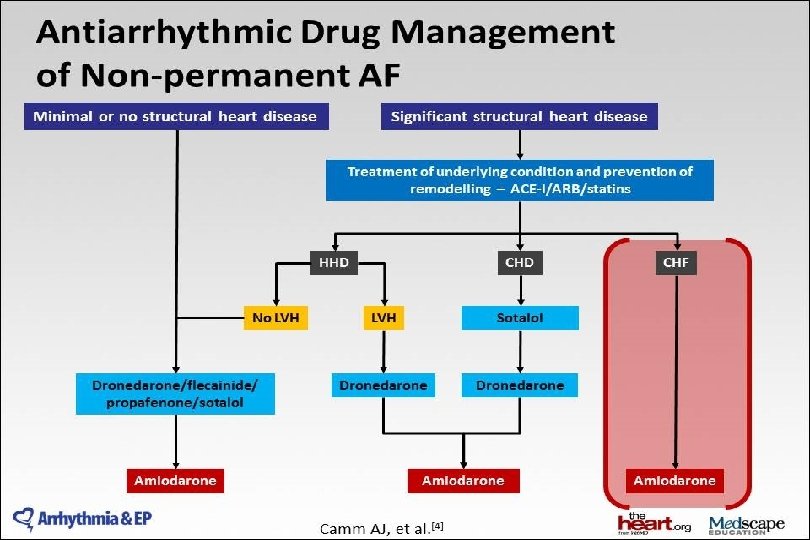
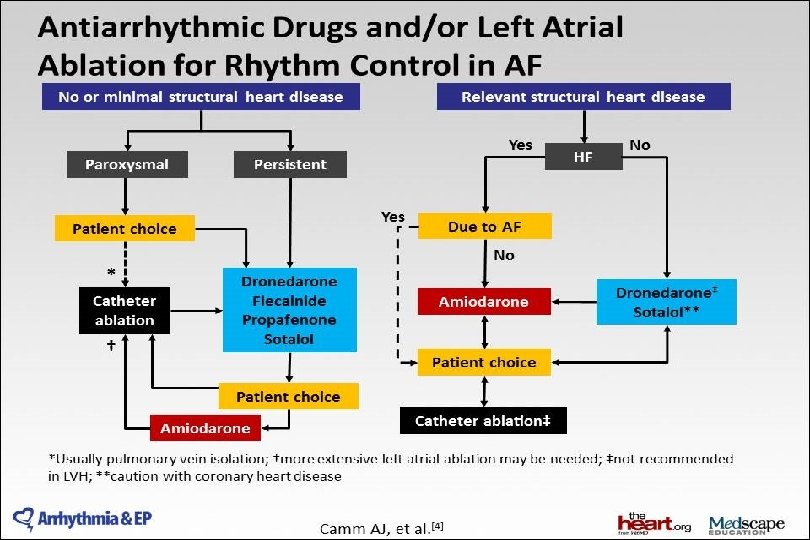

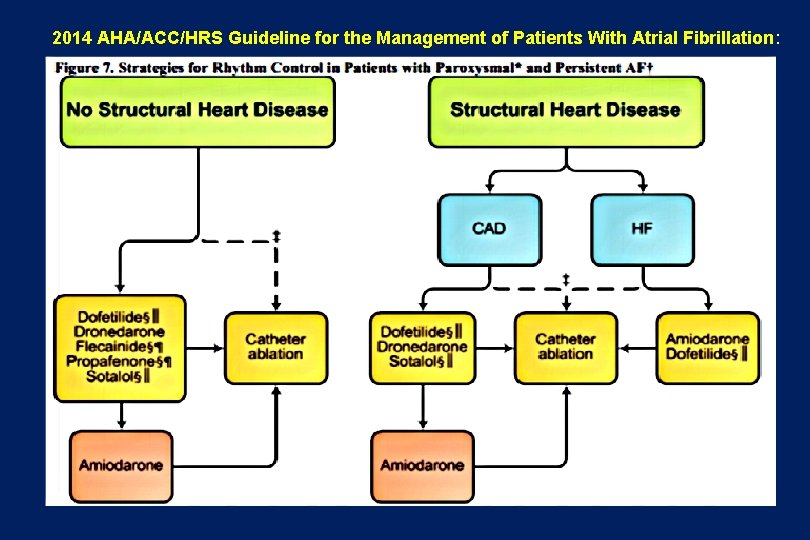
2014 AHA/ACC/HRS Guideline for the Management of Patients With Atrial Fibrillation:

Köszönöm a megtisztelő figyelmet !

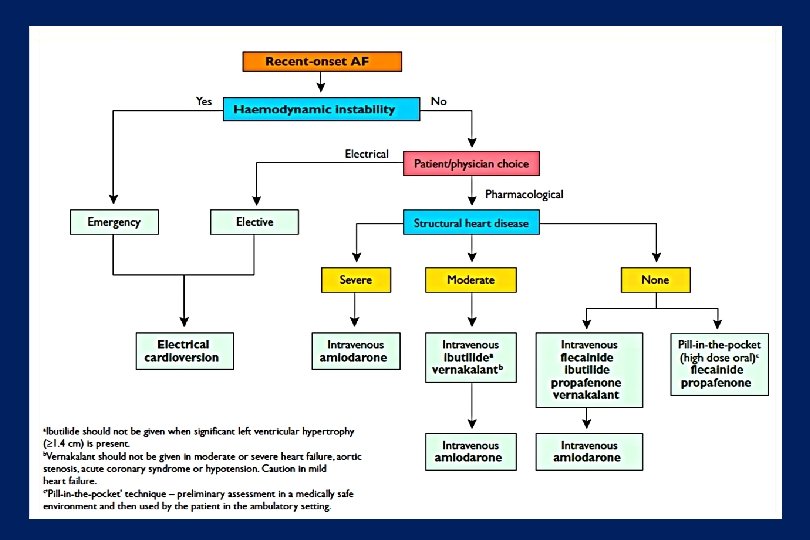
- Slides: 113