Pituitary Causes of female infertility By Dr Mohammad


Pituitary Causes of female infertility By : Dr Mohammad Wali Naseri MD Associate Prof of Internal Medicine, Division of Endocrinology Metabolism and Diabetes, Kabul University of Medical Sciences

• The eye don’t see what the mind doesn’t know
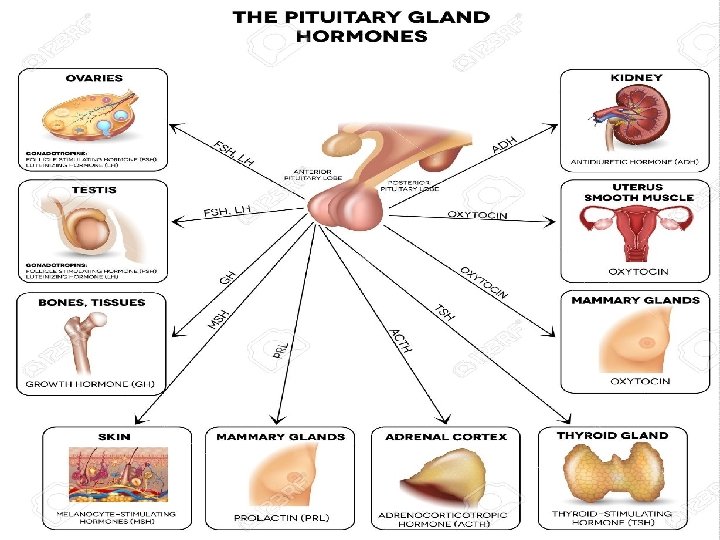

Pituitary Causes of female infertility Pituitary: Sheehan syndrome Tumors: Pituitary adenoma, metastatic Empty sella syndrome Inappropriate gonadal steroid feedback: ○ estrogen excess: obesity/ tumors ○ estrogen deficiency: aromatase deficiency/ ER gene mutation ○ androgen excess: adrenal or ovarian ○ PCOS Testicular feminization (46 XY) - absence androgen receptors

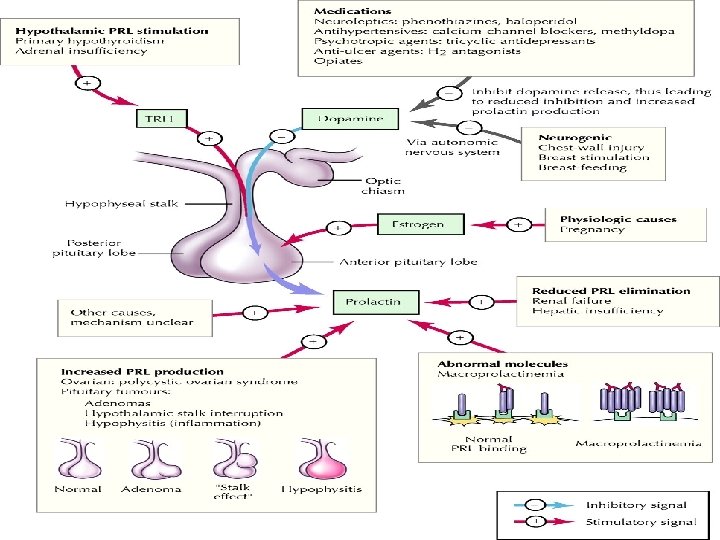
Functions of prolactin • Important role in a variety of reproductive functions • Essential factor for normal production of breast milk following child birth • Hyperprolactinaemia disrupts normal pulsatile secretion of gonadotrophic-releasing hormone, altered LH and FSH secretion and impaired gonadal steroidogenesis, leads to infertility and gonadal dysfunction

Control of prolactin secretion • Secretion is mainly under inhibitory control by hypothalamic dopamine • Circadian variation. Levels rise after the onset of sleep, nocturnal peak of 2 x daytime concentration

Epidemiology • Prolactinomas are the commonest functioning pituitary tumor. • Post-mortem studies show micro adenomas in 10% of the population. • During life, microprolactinomas are commoner than macroprolactinomas, and there is a ♀ preponderance of microprolactinomas.

Clinical features Hyperprolactinaemia (micro adenomas and macro adenomas) • Galactorrhoea (up to 90% ♀, <10% ♂). • Disturbed gonadal function in ♀ presents with menstrual disturbance (up to 95%)—amenorrhea, oligomenorrhoea, or with infertility and reduced libido. • Disturbed gonadal function in ♂ presents with loss of libido and/ or erectile dysfunction. Presentation with reduced fertility and oligospermia or gynaecomastia is unusual.

Clinical features • Hyperprolactinemia is associated with a long-term risk of decreased BMD. • Hyperprolactinemia inhibits Gn. RH release, leading to decreased LH secretion. • There may be a direct action of PRL on the ovary to interfere with LH and FSH signaling which inhibits estradiol and progesterone secretion and also follicle maturation.

Mass effects (macro adenomas only) • Headaches and visual field defects (uni- or bitemporal defects). • Hypopituitarism. • Invasion of the cavernous sinus may lead to cranial nerve palsies. • Occasionally, very invasive tumors may erode bone and present with a CSF leak or s meningitis.

Causes of hyperprolactinaemia Physiological: • Pregnancy. • Sexual intercourse. • Nipple stimulation/suckling. • Neonatal. • Stress.

Causes of hyperprolactinaemia Pituitary tumor: • Prolactinomas. • Mixed GH/PRL-secreting tumor. • Macro adenoma compressing stalk • Empty sella.

Causes of hyperprolactinemia • Hypothalamic disease—mass compressing stalk (craniopharyngioma, meningioma, neurofibromatosis). • Infiltration—sarcoidosis, Langerhans cell histiocytosis. • Stalk section—head injury, surgery. • Cranial irradiation.

Causes of hyperprolactinaemia Drug treatment: • Dopamine receptor antagonists (metoclopramide, domperidone). • Neuroleptics* (perphenazine, flupentixol, fluphenazine, haloperidol, thioridazine, chlorpromazine, trifluoperazine, risperidone, sulpiride). • Antidepressants(tricyclics, selective serotonin reuptake inhibitors, monoamine oxidase inhibitors, sulpiride, amisulpride, imipramine, clomipramine, amitriptyline, pargyline, clorgiline). • Cardiovascular drugs—verapamil, methyldopa, reserpine. • Opiates. • Cocaine. • Protease inhibitors—e. g. ritonavir, indinavir, zidovudine. • Oestrogens. • Others—bezafibrate, omeprazole, H 2 antagonists.

Causes of hyperprolactinemia Metabolic: • Hypothyroidism—TRH increases PRL. • Chronic renal failure—reduced PRL clearance. • Severe liver disease—disordered hypothalamic regulation

Causes of hyperprolactinemia Other: • PCOS—can make differential diagnosis of menstrual problems difficult. • Chest wall lesions—zoster, burns, trauma(stimulation of suckling reflex). • No cause found: • ‘Idiopathic hyperprolactinemia.

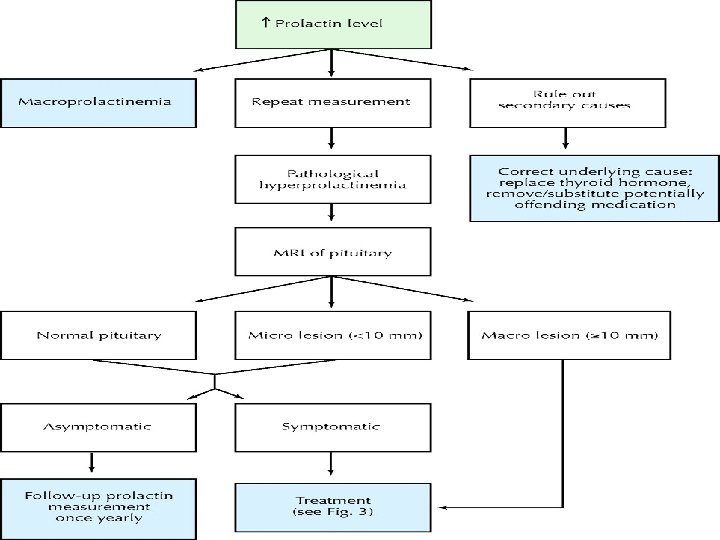
Evaluation Biological evaluation of related hormonal axes: • Careful drug history and physical examination • TFT, RFT • PCO • and exclusion of pregnancy
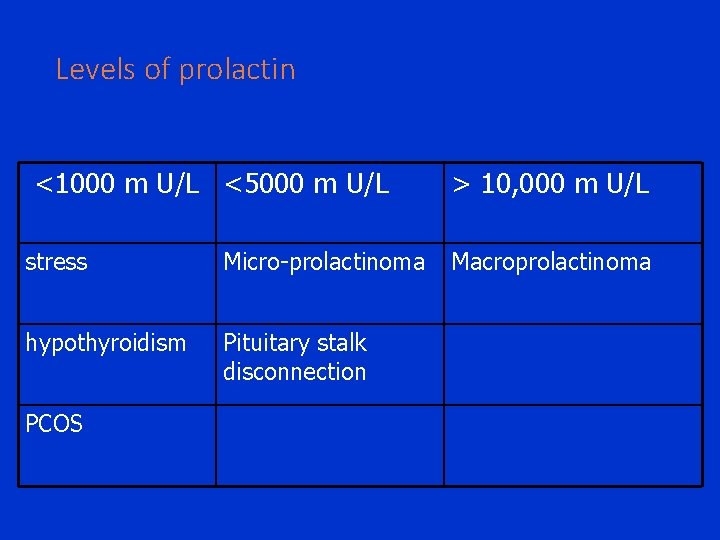
Levels of prolactin <1000 m U/L <5000 m U/L stress Micro-prolactinoma hypothyroidism Pituitary stalk disconnection PCOS > 10, 000 m U/L Macroprolactinoma

Evaluation for hypothalamicpituitary pathology • Clinical examination: assessment of visual fields • Imaging : MRI /CT • Pituitary microadenoma < 10 mm • Pituitary macroadenoma > 10 mm • Pituitary stalk lesions • Hypothalamic tumours, granulomas
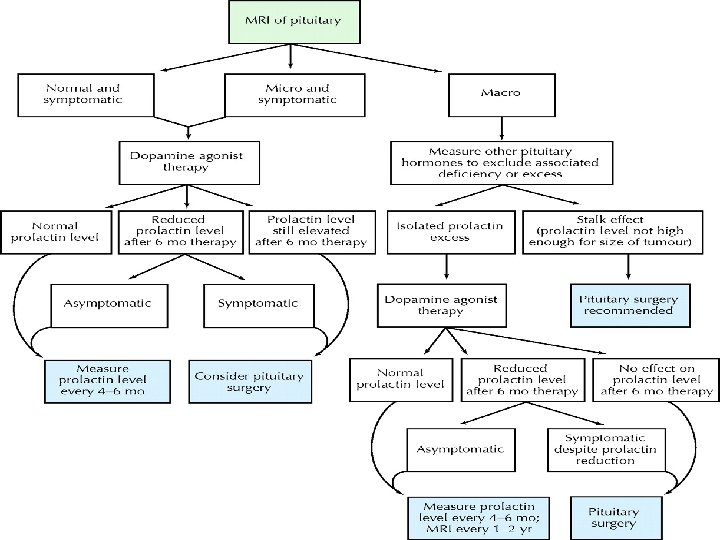

Treatment of prolactin-secreting pituitary adenoma • Medical • Surgical • Radiotherapy

Treatment of prolactinomas Aims of therapy: • Microprolactinomas. Restoration of gonadal function. • Macroprolactinomas. • Reduction in tumor size and prevention of tumor expansion. • Restoration of gonadal function. • Although microprolactinomas may expand in size without treatment, the vast majority do not. • Macroprolactinomas, however, will continue to expand lead to pressure effects. Definitive treatment of the tumor is, therefore, necessary.

Dopamine agonist • Primary treatment of choice • Normalise prolactin levels, restoration of pituitary function and tumor shinkage in 80 -90% over several weeks JCEM 1997 82 996 -1000 • Tumour shinkage by at least 25% of volume in 80% of patients with large macroadenoma • Improvement in pressure symptoms within 48 hrs • In men, 50% may require testosterone replacement, withhold until prolactin levels are normalised
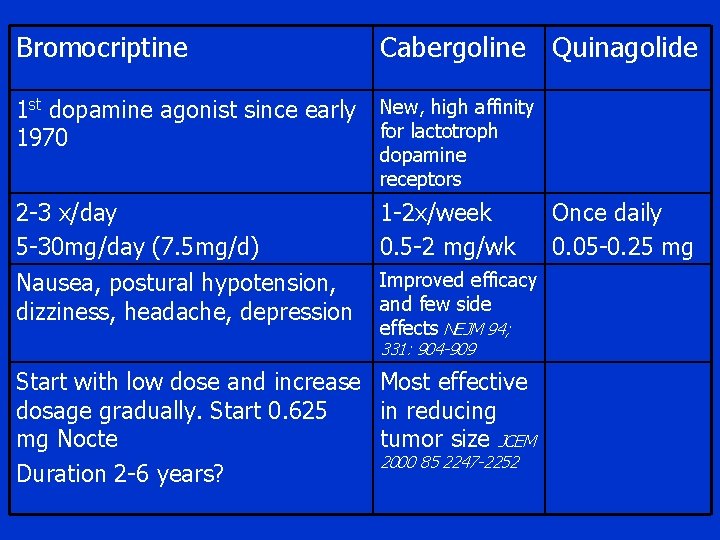
Bromocriptine Cabergoline Quinagolide 1 st dopamine agonist since early New, high affinity for lactotroph 1970 dopamine receptors 2 -3 x/day 5 -30 mg/day (7. 5 mg/d) Nausea, postural hypotension, dizziness, headache, depression 1 -2 x/week 0. 5 -2 mg/wk Improved efficacy and few side effects NEJM 94; 331: 904 -909 Start with low dose and increase Most effective dosage gradually. Start 0. 625 in reducing mg Nocte tumor size JCEM 2000 85 2247 -2252 Duration 2 -6 years? Once daily 0. 05 -0. 25 mg

Duration of treatment • Early studies showed remission is rare after interruption of therapy, life long treatment Clin Endo 1991; 34: 173 -174 • Recent studies showed increase in remission and therapeutic withdrawal is recommended J Royal College of Physicians 1997; 31: 628 -636
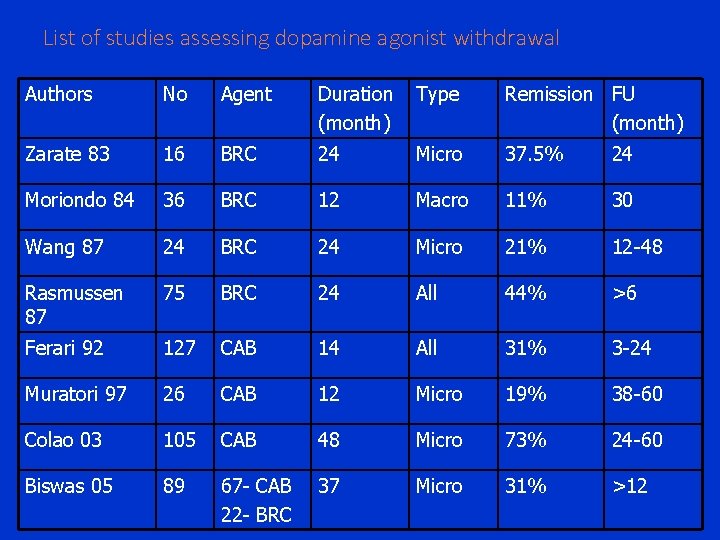
List of studies assessing dopamine agonist withdrawal Authors No Agent Duration (month) Type Remission FU (month) Zarate 83 16 BRC 24 Micro 37. 5% 24 Moriondo 84 36 BRC 12 Macro 11% 30 Wang 87 24 BRC 24 Micro 21% 12 -48 Rasmussen 87 75 BRC 24 All 44% >6 Ferari 92 127 CAB 14 All 31% 3 -24 Muratori 97 26 CAB 12 Micro 19% 38 -60 Colao 03 105 CAB 48 Micro 73% 24 -60 Biswas 05 89 67 - CAB 22 - BRC 37 Micro 31% >12

Remission • Long term follow up studies of untreated patients have shown that prolactinomas are very indolent • Short term therapy appears to induce cytostatic effects including reduction in organelle size and reduction in the volume of prolactin cells JCEM 55, 11798 -1183 • Long-term therapy induces cytocidal effects such as necrosis, fibrosis and inflammatory cell infiltration JCEM 58, 1179 -1183

Dopamine resistance of prolactinoma Definition: • Failure to normalize prolactin on dopamine agonist therapy. • Failure to decrease tumor size by 50% or more. Frequency: • Bromocriptine 24%. • Pergolide 13%. • Cabergoline 11%. Cause: • D 2 receptors on tumor decreased in number: normal affinity. Treatment: • Switch dopamine agonist. • Increase dose of dopamine agonist. • Trans sphenoidal surgery. • Clomiphene gonadotropins for fertility. • Estrogen replacement therapy + - DXR for macro adenoma.

Prolactinoma in pregnancy Effect of pregnancy on tumor size • Risk of significant tumor enlargement (i. e. resulting in visual field disturbances or headaches): • Micro adenoma 1– 2%. • Macro adenoma 15– 35%. • Macro adenoma treated with surgery and/or radiotherapy before pregnancy 4– 7%.

Effect of dopamine agonists on the fetus Bromocriptine • Over 6, 000 pregnancies have occurred in women receiving bromocriptine in early pregnancy, and the incidence of complications in these pregnancies with regard to fetal outcome is similar to that of the normal population.

Continued Cabergoline: • Also probably safe in early pregnancy and has been used in >380 pregnancies, with no increased risk of fetal loss or congenital abnormalities, but fewer data are available. Quinagolide: • It has been used in >176 pregnancies, with a slightly higher rate of congenital abnormalities compared to the background rate.

Management of prolactinoma in pregnancy Microprolactinoma: • After recent MRI, initiate dopamine agonist therapy to induce normal ovulatory cycles and fertility. • Stop bromocriptine as soon as pregnancy is confirmed. • Assess for visual symptoms and headache at each trimester, although the risk of complications is low (<5%). Serum PRL levels are difficult to interpret during pregnancy, as they are normally elevated; therefore, they are not measured. • MRI is indicated in the occasional patient who becomes symptomatic. • In the post-partum period, recheck serum PRL level 2 months after cessation of breastfeeding. Reassess size of microprolactinoma by MRI only if serum PRL level is higher than pre-pregnancy concentrations. • 40– 60% chance of remission of microprolactinoma following pregnancy.

Macroprolactinoma Management is controversial and must, therefore, be individualized. Three possible approaches: • Bromocriptine may be used throughout pregnancy to reduce the risk of tumor growth. • May use bromocriptine or cabergoline to induce ovulation, and then stop it after conception. • If symptoms of tumor enlargement develop or there is deterioration in visual fields, then MRI should be performed to assess tumor growth. • If significant tumor enlargement develops, then bromocriptine therapy should be initiated. • Alternatively, the patient may undergo surgical debulking of the tumor and/or radiotherapy before seeking fertility.

Breastfeeding • There is no contraindication to breastfeeding. • If the mother would like to breastfeed, dopamine receptor agonists will have to be discontinued prior to birth, as they inhibit lactation. • The decision to discontinue medication should be assessed on a case-by-case basis, dependent upon the potential risk of optic nerve/ chiasm compression.

Thanks you
- Slides: 38