Pitting Corrosion Materials Engineering Dr Lubna Ghalib Pitting

Pitting Corrosion Materials Engineering Dr. Lubna Ghalib

Pitting Corrosion: It is a form of localized corrosion of a metal surface where small. areas corrode preferentially leading to formation of cavities or pits, and bulk of the surface remains unattached. Metals which form passive films, such as aluminum and steel are more susceptible to this form of corrosion. It causes failure by penetration with only a small percent weight-loss of the entire structure. Environment: Generally the most conductive environment for pitting is the marine environment. Ions such as Cl- , Br- and I- in appreciable concentrations tend to cause pitting of steel. Thiosulfate ions also induce pitting of steels. Aluminum also pits in an same environments of steel. Present of dust or dirt particles in water may lead to pitting corrosion in copper pipes transporting seawater. With soft water pitting in copper occurs in the hottest part of system, whereas with hard waters pitting occurs in the coldest part of the system.

Pitting Corrosion: The following are the conditions for pitting to occur: . The most important condition is that the metal must be in passive state for pitting to occur. Metals which become passive by film formation have a high resistance to uniform corrosion. The process of pitting destroys this protective film at certain sits resulting in the loss of passivity and initiation of pits on metal surface. Breaks in the films or other defects. ( lack of film homogeneity) The presence of halogen ions, such as Cl-, Br-, and I- and even S 2 O 3 -. Stagnant conditions in service. (Pumps serve in seawater with longer life than kept out of service).

Pitting Corrosion: Mechanism : . In order for pitting to take place, the formation of anode is a prerequisite. With the formation of an anode, a local corrosion cell is developed. The anode may be formed as a result of: 1. Lack of homogeneity at the metal corrosive interface, which caused by presence of impurities, grain boundaries, niches, rough surface, etc. The difference in the environments can cause formation of concentration cells on the metal surface. 2. Destruction of a passive film. Resulting to formation a small anode, results in several anode sits and the surrounding surface acts as cathode. Thus, an unfavorable area ratio results. 3. Deposit of debris or solids on the metal surface. This generally leads to the formation of anodic and cathodic sites. 4. Formation of an active – passive cell with a large potential difference.

Pitting Corrosion: 5. The formation of small anode on the passive steel surface, leads to the formation of the above cell. Conditions: (1)The passive metal surrounding the anode is not subject to pitting as it forms the cathode and it is the site for reduction of oxygen. (2)The corrosion products which are formed at the anode cannot spread on to the cathode areas. Therefore, corrosion penetrates the metal rather than spread, and pitting is initiated. (3)There is a certain potential characteristic of a passive metal, below which pitting cannot initiate. This is called pitting potential (Ep ).

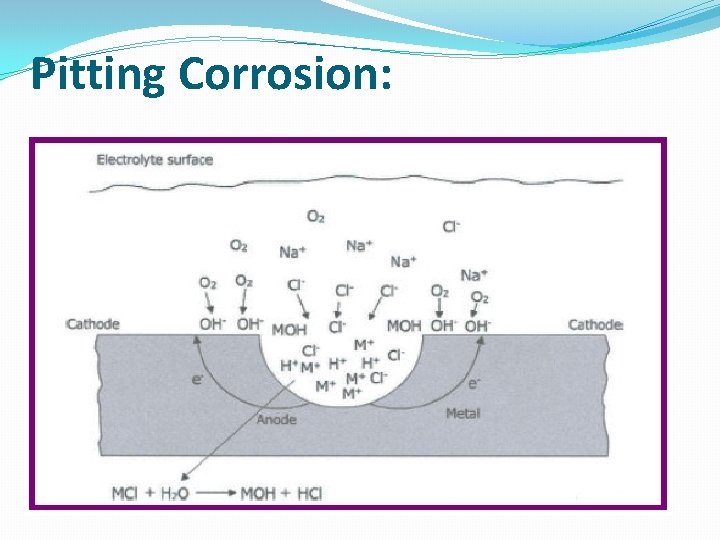
Pitting Corrosion: Pitting Processes 1) The formation of anodic sits by disruption of the protective passive film on the metal surface. M Mn+ +ne. This is balanced by the cathodic reaction of oxygen on the adjacent surface O 2 +2 H 2 O +4 e 4 OH 2) Due to the continuing metal dissolution, an excess of positive ions (M+) is accumulated in the anodic area. The process is selfstimulating and self-propagating. To maintain charge neutrality negative ions (anions), like chloride, migrate from electrolyte (for example seawater or a 5% Na. Cl solution). M+Cl- +H 2 O MOH + H+ + Cl(OH-) ions also migrate to neutralize the positive charges. This process is called hydrolysis.

Pitting Corrosion: 3) The presence of (H+) ions and chloride content prevents repassivation. The above process generates free acid and the p. H value at the bottom of pit is substantially lowered (1. 5 -1. 0). (4) The increase in the rate of dissolution at the anode increases the rate of migration of the chloride ions and the reaction becomes time dependent and continues, resulting in the formation of more and more M+Cl- , generation of more and more H+Cl- by hydrolysis. (5) The process continues until the metal is perforated. The process is autocatalytic and it increases with time resulting in more and more dissolution. (6) Finally, the metal is perforated and the reaction is terminated. As shown above, basically three processes are involved: 1 – Pitting Initiation. 2 – Pitting Propagation. 3 – Pitting Termination.
Pitting Corrosion:

Pitting Corrosion: Corrosion Pits are the primary source of leaks in water handling systems

Pitting Corrosion: Prevention: The methods suggested for combating crevice corrosion generally apply also for pitting. Or using inhibiters e. g Chromates (Cr. O 4) -2, Phosphates (PO 4)-3, Silicates (Si. O 4)-4

Pitting Corrosion: Pitting is an insidious and destructive form of corrosion: Difficult to detect (pits may be small on surface, but extensive below surface from undercutting; may be covered with deposit). Can cause equipment to fail (by perforation) with very little weight loss. Difficult to measure as pit depth and distribution vary widely under (nominally) identical conditions. “Incubation” period may be months or years.

Pitting Corrosion: Some alloys developed especially to resist pitting Effects of alloying on pitting resistance of stainless alloys Element Chromium Nickel Molybdenum Silicon Effect on pitting resistance Increases Decreases; increases when present with Molybdenum Titanium and niobium Decreases resistance in Fe. CI 3, other mediums no effect Sulfur and selenium Decreases Carbon Decreases, especially in sensitized condition
- Slides: 12