Pier Lorenzo Puri M D Sanford Burnham Medical

. Pier Lorenzo Puri, M. D. Sanford. Burnham Medical Research Institute & Fondazione Santa Lucia Introduction to muscular dystrophy pathogenesis: muscle environment, cell types and potential targets for interventions
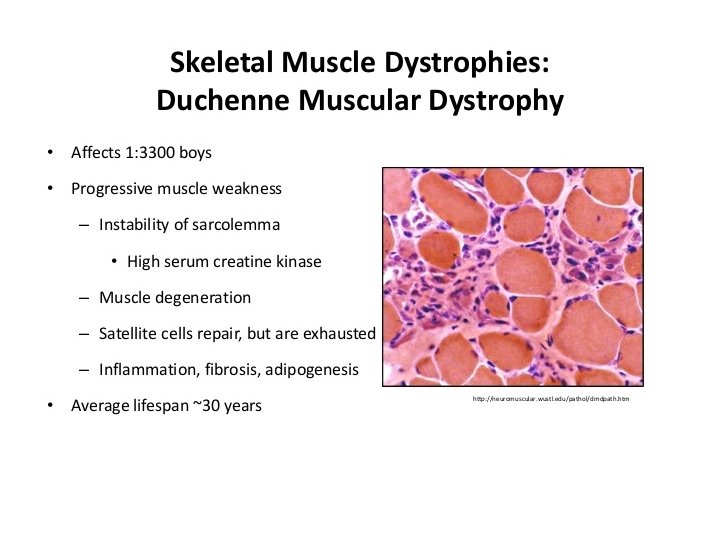
Linking dystrophin to DMD pathogenesis

No effective interventions 30 years after the discovery of dystrophin as the genetic mutation responsible for DMD
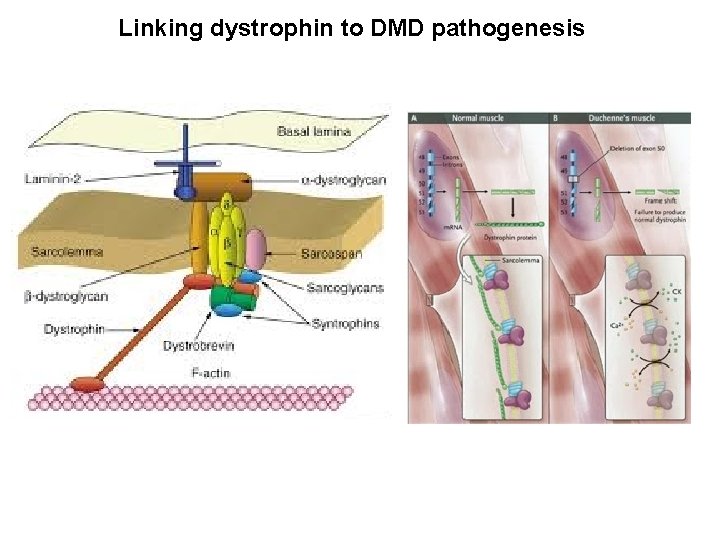
Targeting pathogenic events in DMD therapy: gene mutation, compensatory regeneration or fibrotic degeneration X X Gene therapy Exon skipping, PTC, mini-dystrophin Cell therapy X Stem cells, mesoangioblasts Loss of contractile mass LATE Disease progression Recovery of contractile mass EARLY “Honey moon” Degeneration post-contraction Contraction/Degeneration Pathogenic fibrosis Compensatory Regeneration Cycles and fat Fibrosis/Necrosis deposition Pharmacological therapy Necrosis Muscle loss Pharmacological therapy
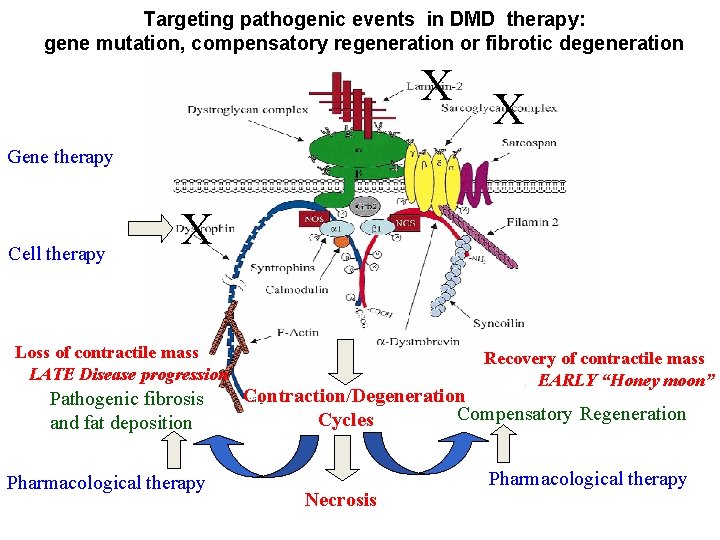
Skeletal muscle regeneration Myo. D/Myf 5+ Infiltrate Resolution: Transient Infiltrate: Apoptosis Phenotypic switch Macrophages Eosynophils Neutrophis T cells Activated resident , Interstitial cells Pax 7+
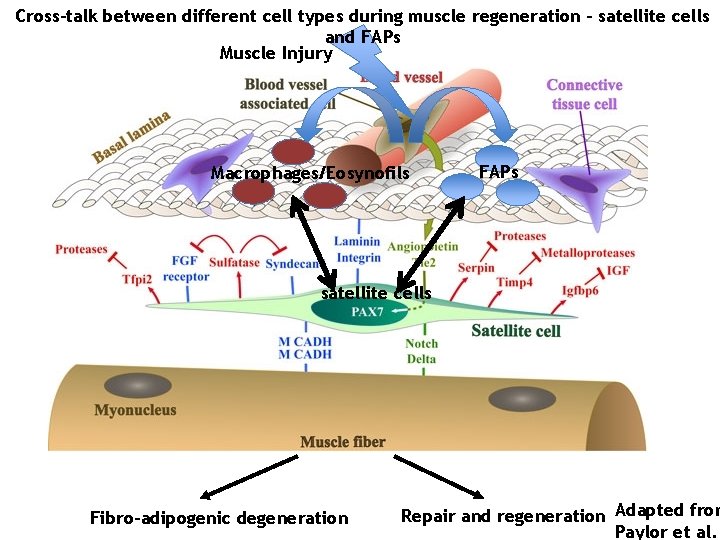
Cross-talk between different cell types during muscle regeneration – satellite cells and FAPs Muscle Injury Macrophages/Eosynofils FAPs satellite cells Fibro-adipogenic degeneration Repair and regeneration Adapted from Paylor et al.
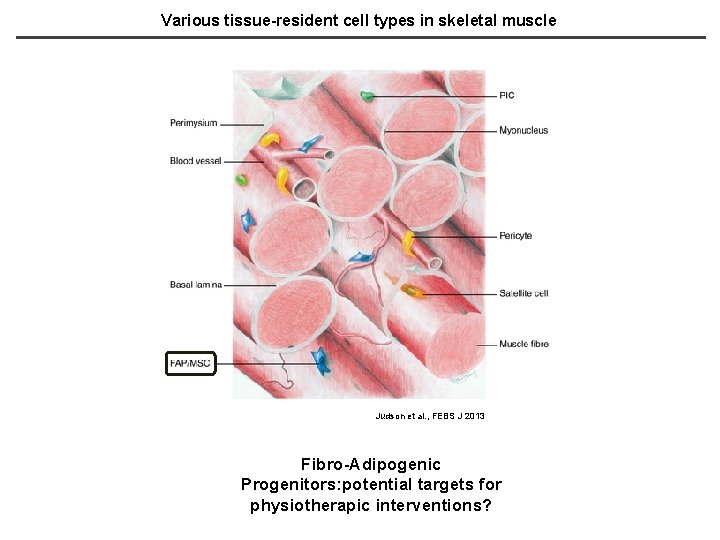
Various tissue-resident cell types in skeletal muscle Judson et al. , FEBS J 2013 Fibro-Adipogenic Progenitors: potential targets for physiotherapic interventions?

Living without dystrophin: the case of “healthy” dystrophin-deficient dogs (communication from Dr Engvall before publication
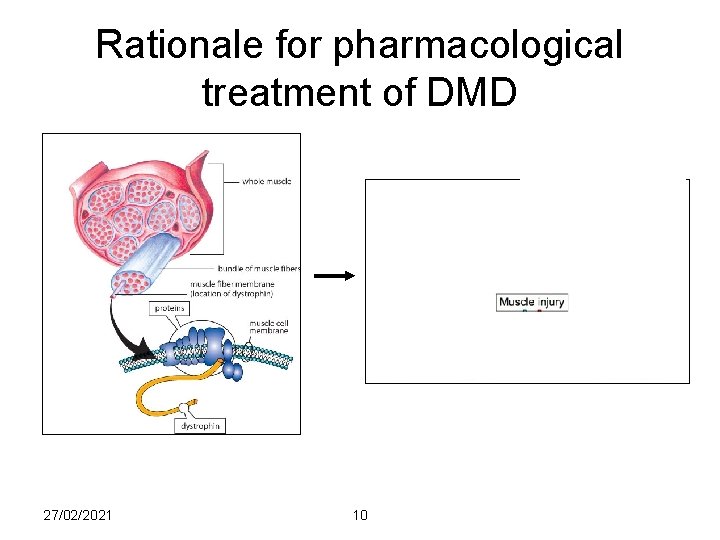
Rationale for pharmacological treatment of DMD 27/02/2021 10
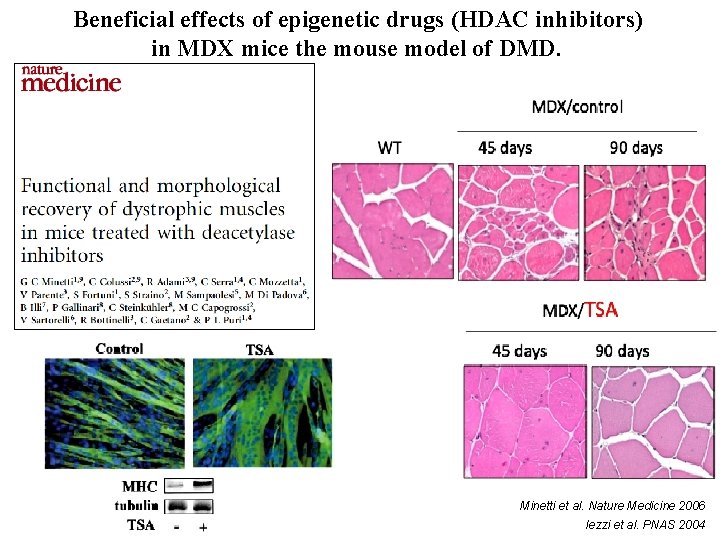
Beneficial effects of epigenetic drugs (HDAC inhibitors) in MDX mice the mouse model of DMD. Minetti et al. Nature Medicine 2006 Iezzi et al. PNAS 2004

A Phase 2 Study to Assess the Safety and Effects of Givinostat in Children With DMD SPONSOR ITALFARMACO CONDITION DUCHENNE MUSCULAR DYSTROPHY (DMD) INTERVENTION DRUG: GIVINOSTAT LOCATION ITALY STUDY PHASE 2 RECRUITMENT 20 MALE CHILDREN WITH DMD AGE: 7 YEARS TO 11 YEARS STUDY ARM GIVINOSTAT WILL BE ADMINISTERED FOR 12 MONTHS AS 2 ORAL DOSES DAILY WHILE THE CHILD IS IN FED STATE. ESTIMATED COMPLETION DATE MAY 2014 OUTCOME MEASURES CHANGE IN HISTOLOGICAL ENDPOINTS AND IN MUSCULAR FUNCTION AFTER 12 MONTHS OF TREATMENT WITH GIVINOSTAT
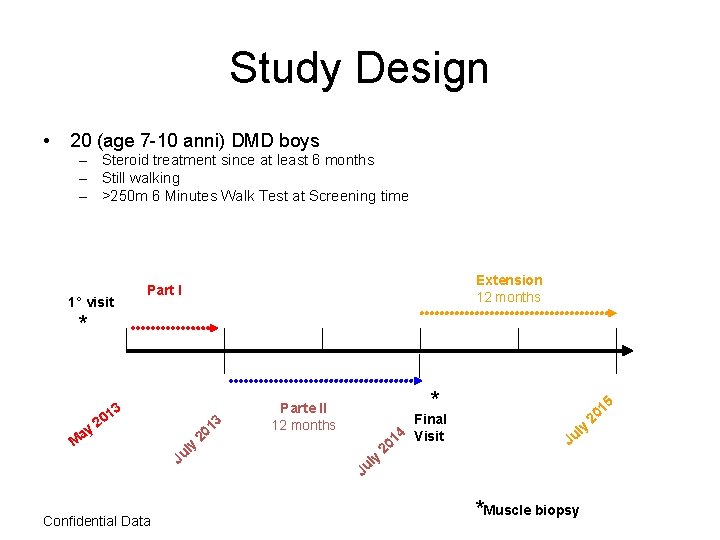
Study Design • 20 (age 7 -10 anni) DMD boys – Steroid treatment since at least 6 months – Still walking – >250 m 6 Minutes Walk Test at Screening time 1° visit Extension 12 months Part I * ay M 3 01 3 2 Confidential Data ly u J 1 20 * Parte II 12 months y ul Final 4 1 Visit 20 5 y ul J J *Muscle biopsy 1 20

Givinostat promotes compensatory regeneration and reduces fibro-adipogenic degeneration and necrosis in DMD boys 80. 0 % Relative Change (Pre Treatment vs Post treatment-V 10) 60. 0 40. 0 29. 1 % Change 20. 0 -20. 0 MFAF -40. 0 TOTAL Fibrosis Necrosis Fatty Replacement -27. 4 -43. 5 -60. 0 -80. 0 -100. 0 Confidential Data 14 -37. 5

Givinostat increases muscle fiber size in DMD boys – cross sectional area (CSA) No. hypercontracted fibers x field (20 X) PRE MEAN SD p-value 1. 98 0. 71 POST relative change (%) 0. 77 -1. 20 -60. 4 0. 54 0. 66 <. 0001 p-value= <. 0001 Confidential Data The P-value is from a pairwise t-test to test the difference between Baseline and End of Study

Current Lab Members Barbora Malecova Sonia Albini Paula Coutinho Alessandra Dall’Agnese Sole Gatto Tom Roberts Usue Etxaniz Irigoien Valentina Saccone Silvia Consalvi Martina Sandona Luca Madaro Lucia Latella Carles Sanchez Elisabetta Fiacco Magda Passafaro Past Lab members Carlo Serra Lorenzo Giordani Sonia Forcales Daniela Palacios Cristiano Simone Chiara Mozzetta Giulia Minetti Collaborators Italfarmaco (Italy) Sacco lab (Sanford. Burnham) Minucci lab (IFOM) Mercola lab. (Sanford Burnham) Battistini L & Borsellino G (F. Santa Lucia) Sassoon D. (Université Pierre et Marie Curie. Sorbonne Universités Simon Melov & James Flynn Buck Institute for Aging, Novato CA Supports: CIRM Training grant to BM Sonal Naik Stem Cell Genomics core, SCRM-UCSD Amy Cortez Flow cytometry core, SBMRI Clinical study Bertini E. , D’Amico A. (Ospedale Pediatrico Bambin Gesù. Roma). Mercuri E. , Pane M. Sivo S. (Policlinico Gemelli, Roma), Comi G. , Magri F. (Policlinico, Milano), Vita G. , Messina S. , Vita G. (Policlinico, Messina). NIH/NIAMS SDMRC-P 30 pilot grant to BM

Sanford. Burnham Medical Research Instit Fondazione Santa Lucia
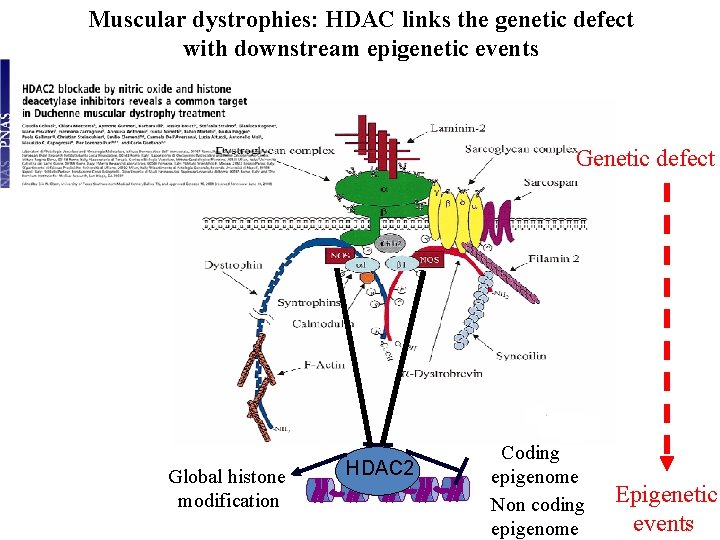
Muscular dystrophies: HDAC links the genetic defect with downstream epigenetic events Genetic defect Global histone modification HDAC 2 Coding epigenome Non coding epigenome Epigenetic events

Functional improvement of mdx mice treated with HDAC inhibitors MDX/TSA MDX/control Beginning of test time to exaustion (sec) 1500 MDX/control Treadmill test Wild type mouse 1000 500 0 WT contr. TSA MDX End of test MDX/TSA MDX/control Wild type mouse
- Slides: 19