Pi bonds Paulings really great idea to use
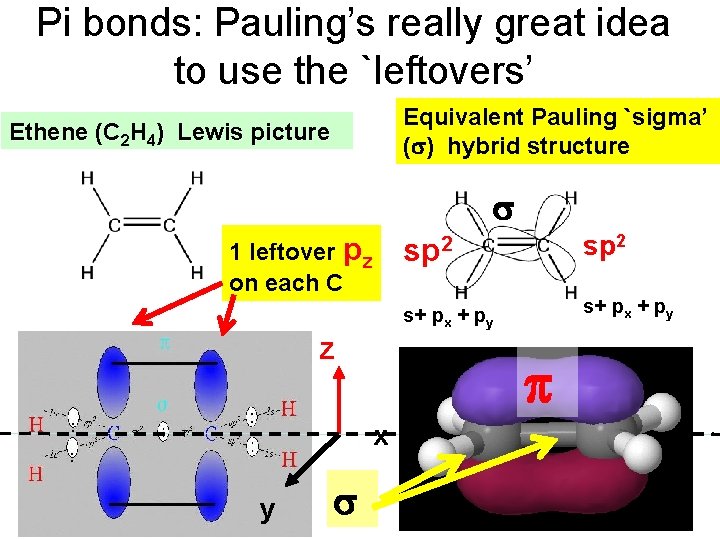
Pi bonds: Pauling’s really great idea to use the `leftovers’ Equivalent Pauling `sigma’ ( ) hybrid structure Ethene (C 2 H 4) Lewis picture 1 leftover pz on each C z sp 2 s+ px + py x y sp 2

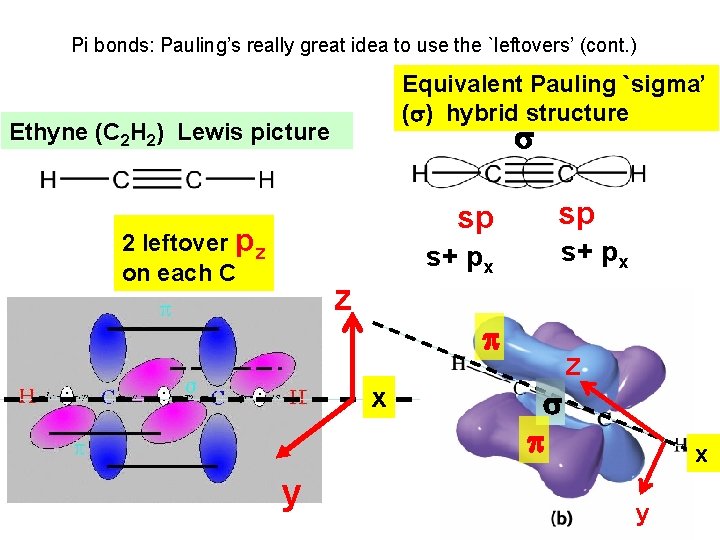
Pi bonds: Pauling’s really great idea to use the `leftovers’ (cont. ) Equivalent Pauling `sigma’ ( ) hybrid structure Ethyne (C 2 H 2) Lewis picture sp 2 leftover pz on each C s+ px z s+ px x y sp Z x y

How Pauling’s model `fixes’ the problems with Lewis model Atomic orbitals (AO) `reorganize’ (hybridize) when individual atoms approach each other such that the number of `links’ predicted by the Lewis model = the number of s, p (and d and f) orbitals combined in the reorganization. The `hybrid’ combinations are called Linear Combinations of Atomic Orbitals (LCAO). The `lobes’ in LCAO on individual atoms overlap and share two electrons between the atoms in a `sigma’ bond (often called a `valence’ or structural linkage bond. )

How Pauling’s model `fixes’ the problems with Lewis model (continued) `pi’ bonds are far less stable and far more reactive than sigma bonds. (Further out, softer, not between atoms but above and below) Ethane is held together by just `sigma bonds and is thus not very reactive. Both ethylene and acetylene have pi bonds which are easily reacted. That acetylene is more reactive thane ethylene results because it has two pi bonds while ethylene has only 1 pi bond

How Pauling’s model `fixes’ the problems with Lewis model (continued) The large and loose electronic clouds above the metals are `soft’ and easily `blended’ (overlapped’ with like electronic distributions (e. g. soft and fluid). Pi bonds are soft and fluid; sigma bonds aren’t. Moreover, the pi bonds are far away from the central core of the molecule, thus reducing nuclear-nuclear repulsions.

How Pauling’s model `fixes’ the problems with Lewis model (continued) The pi bonds occupy space above and below the sigma bond and thus do not crowd them. The two pi bonds are also on different and perpendicularly aligned planes to minimize pi-pi crowding.

Linus Pauling An American Chemical Genius “I am awesome ( and kinda cute in a geeky way…)”

Problems with Pauling Valence Bond (LCAO) Model: 1940 üFails to provide procedure to determine the numeric details of molecular energy and spectra translation…can’t compute a stinking thing üDoesn’t allow adjustments for changes of atoms bonded to the central atom Example: can’t distinguish electronic shape or density of CH 4 vs CF 4 except with `electronegativity’ and vague sketches Linus Pauling Middle School Corvallis Ore.
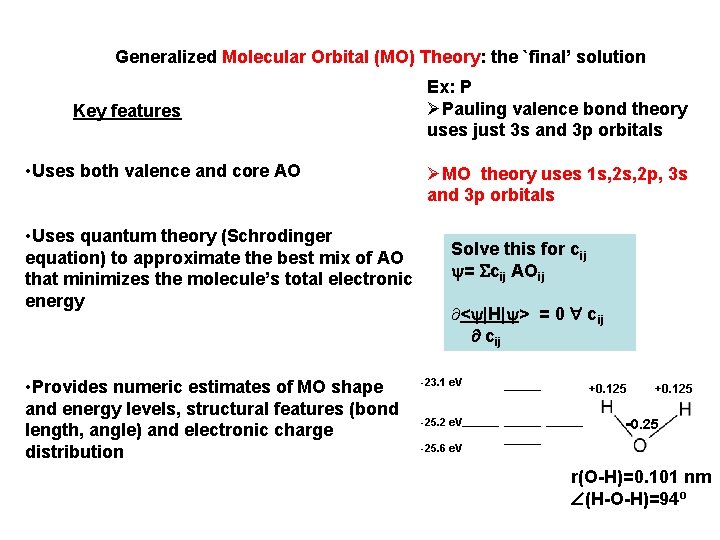
Generalized Molecular Orbital (MO) Theory: the `final’ solution Key features • Uses both valence and core AO • Uses quantum theory (Schrodinger equation) to approximate the best mix of AO that minimizes the molecule’s total electronic energy • Provides numeric estimates of MO shape and energy levels, structural features (bond length, angle) and electronic charge distribution Ex: P ØPauling valence bond theory uses just 3 s and 3 p orbitals ØMO theory uses 1 s, 2 p, 3 s and 3 p orbitals Solve this for cij = cij AOij ¶< |H| > = 0 cij -23. 1 e. V -25. 2 e. V +0. 125 -0. 25 -25. 6 e. V r(O-H)=0. 101 nm (H-O-H)=94 o

Solve this for cij = cij AOij The Big Kahuna of the general MO approach ¶< |H| > = 0 cij His un-named, but critical co-conspirator…. • Professor John Pople Northwestern University: (tenure: 1965 -2005) • Aka: the God of Quantum Computation John A. Pople wins the Nobel Prize in Chemistry, 1998 "for his development of computational methods in quantum chemistry".
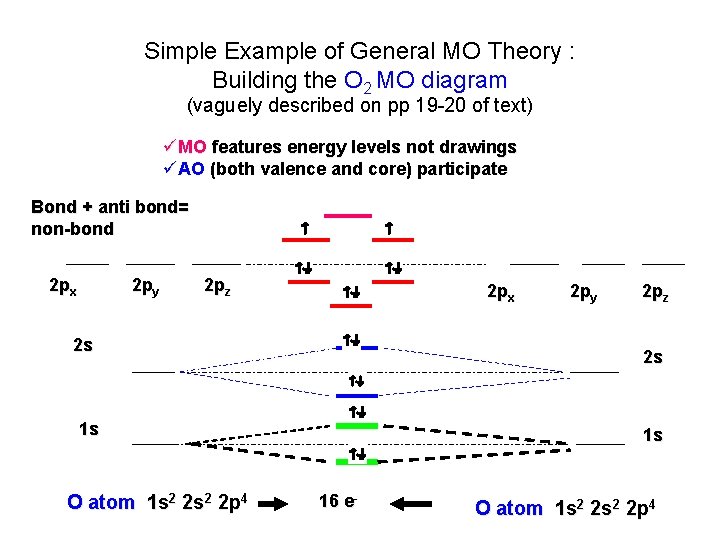
Simple Example of General MO Theory : Building the O 2 MO diagram (vaguely described on pp 19 -20 of text) üMO features energy levels not drawings üAO (both valence and core) participate Bond + anti bond= non-bond 2 px 2 py 2 pz 2 px 2 s 2 pz 2 s 1 s O atom 1 s 2 2 p 4 2 py 1 s 16 e- O atom 1 s 2 2 p 4
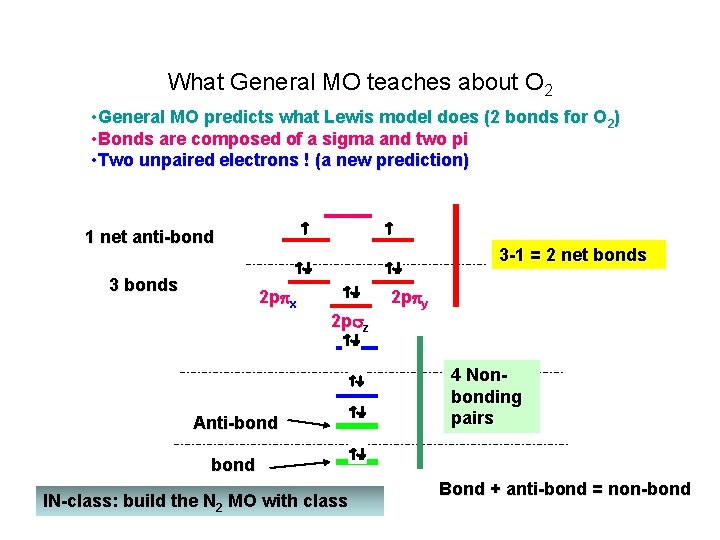
What General MO teaches about O 2 • General MO predicts what Lewis model does (2 bonds for O 2) • Bonds are composed of a sigma and two pi • Two unpaired electrons ! (a new prediction) 1 net anti-bond 3 bonds 3 -1 = 2 net bonds 2 p x 2 p z Anti-bond 2 p y 4 Nonbonding pairs bond IN-class: build the N 2 MO with class Bond + anti-bond = non-bond

Hierarchy of electronic modeling methods Most rigorous §Ab initio methods (no assumptions about `field’. . mostly for small, reactive molecules) §HF-SCF(Hartree-Fock Self-Consistent field model)*** Moeller-Plesset*** §Molecular Mechanical models*** §Extended Huckel (EHMO)* §Simple Huckel (MO)* Computer-based , numerical methods: Molecular orbital methods ***done by Spartan Qchem package in-house. . can be a quantum moron and still use to good effect ($700) *Doc used these in the days of yore as grad student…obsolete üPauling LCAO Valence bond method üLewis Model üHONC least rigorous Visual / intuitive methods
- Slides: 14