Physics light waves Chapter 14 Chapter 14 Vocabulary

Physics: light waves Chapter 14

Chapter 14 Vocabulary Terms § § § § § reflection black fluorescence intensity color blue light ray CMYK color ultraviolet § § § § § infrared photon RBG color photoluminescence additive color white red green spherical pattern cyan § § § § § magenta yellow pigment speed of light (c) incandescence rod cell cone cell subtractive color photoreceptor

Properties and Sources of Light Key Question: What are some useful properties of light?

Properties and Sources of Light § § Light travels almost unimaginably fast and far. Light carries energy and information. Light travels in straight lines. Light bounces and bends when it comes in contact with objects. § Light has color. § Light has different intensities, it can be bright or dim.

Electric Light § The process of making light with heat is called incandescence. § Incandescent bulbs generate light when electricity passes through a thin piece of metal wire called a filament. § The filament heats up and gives off light.

Electric Light § The other common kind of electric light is the fluorescent bulb. § Fluorescent bulbs convert electricity directly to light without generating a lot of heat. § Fluorescent bulbs use high-voltage electricity to energize atoms of gas that fill the bulb.

Light carries energy and power § Light is a form of energy that travels. § The intensity of light is the amount of energy per second falling on a surface. § Most light sources distribute their light equally in all directions, making a spherical pattern. § Because light spreads out in a sphere, the intensity decreases the farther you get from the source.
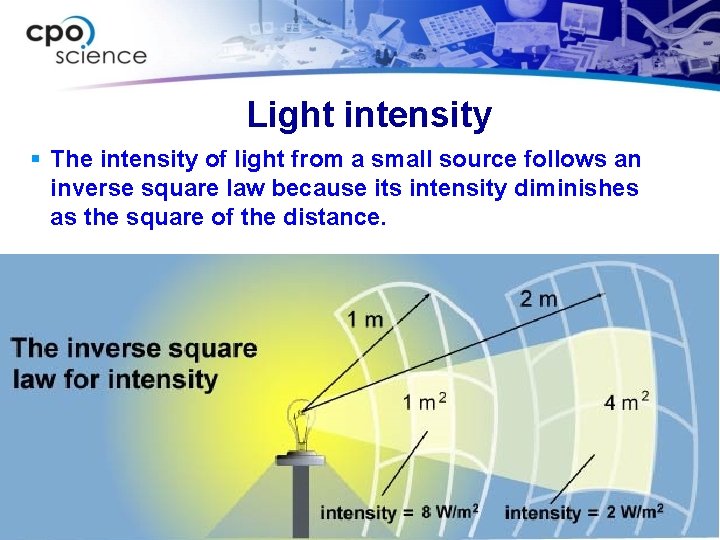
Light intensity § The intensity of light from a small source follows an inverse square law because its intensity diminishes as the square of the distance.

16. 1 Light carries information § The fiber-optic networks you read about are pipelines for information carried by light.

Light carries information § In some cities, a fiberoptic cable comes directly into homes and apartments carrying telephone, television, and Internet signals.

The speed of light § The speed at which light travels through air is approximately 300 million meters per second. § Light travels almost a million times faster than sound.

The speed of light § The speed of light is so important in physics that it is given its own symbol, a lower case c. § The best accepted experimental measurement for the speed of light in air is 299, 792, 500 m/sec. § For most purposes, we do not need to be this accurate and may use a value for c of 3 × 108 m/s.

Electromagnetic Waves § Radio waves § Microwaves § IR waves § Visible Light § UV waves § X rays § Gamma rays Wavelengt h Longest Frequenc y smallest Highest
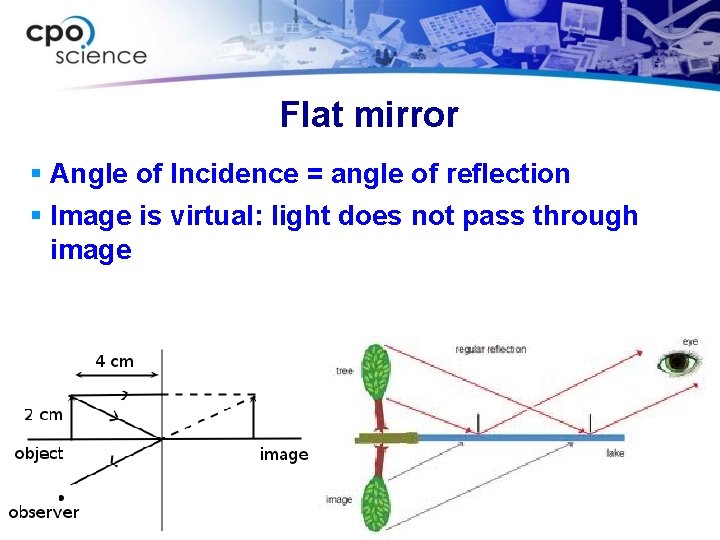
Flat mirror § Angle of Incidence = angle of reflection § Image is virtual: light does not pass through image
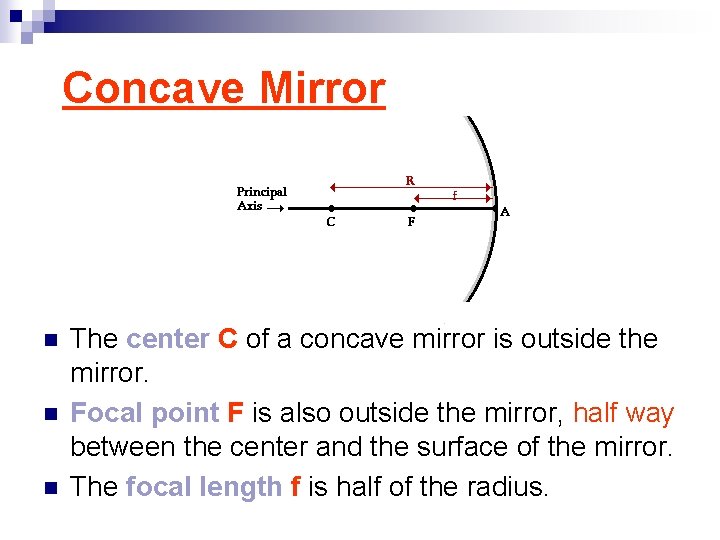
Concave Mirror n n n The center C of a concave mirror is outside the mirror. Focal point F is also outside the mirror, half way between the center and the surface of the mirror. The focal length f is half of the radius.

Significance of the focal point All light rays that are parallel to the axis will pass (after extrapolating for convex mirror) through the focal point! n For a concave mirror, the light can pass through the focal point. n

Image from a concave mirror: case 1 Step 3 Step 2 Step 4
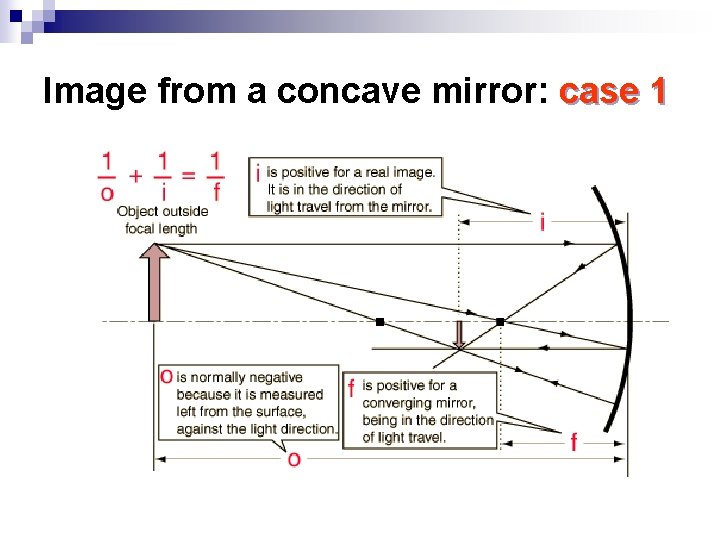
Image from a concave mirror: case 1

Properties of the Image n If we put an object outside of the center of a concave mirror, we find the image is ¨ Real, in the sense that all light rays pass through the image. ¨ Inverted, in the sense that the direction of the arrow has been changed. ¨ The image is smaller!

Animation for case 1
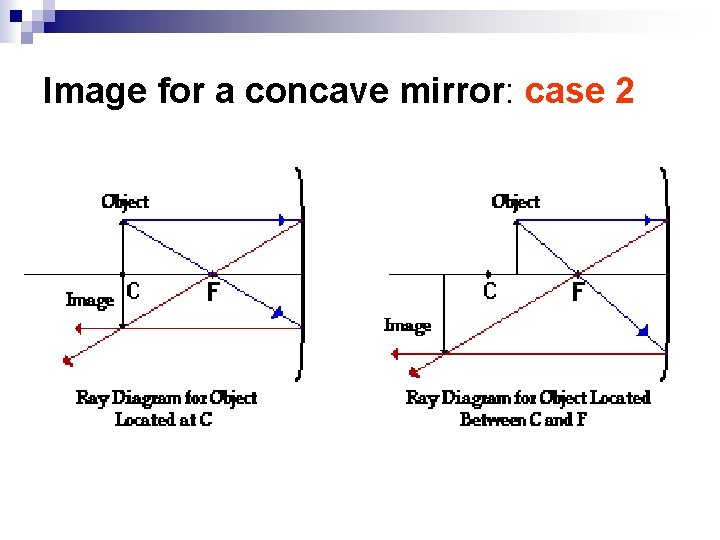
Image for a concave mirror: case 2
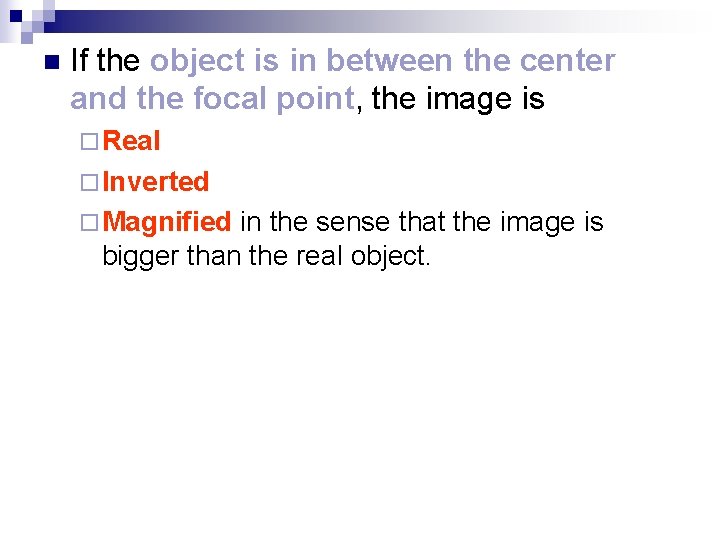
n If the object is in between the center and the focal point, the image is ¨ Real ¨ Inverted ¨ Magnified in the sense that the image is bigger than the real object.

Animation for case 2
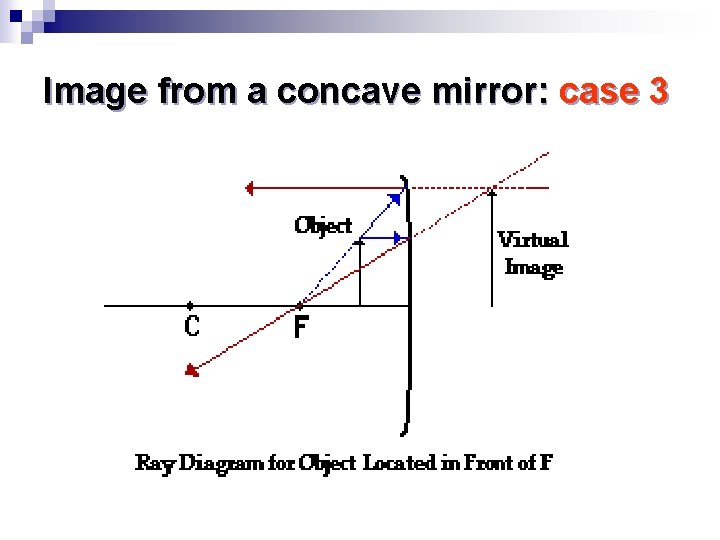
Image from a concave mirror: case 3
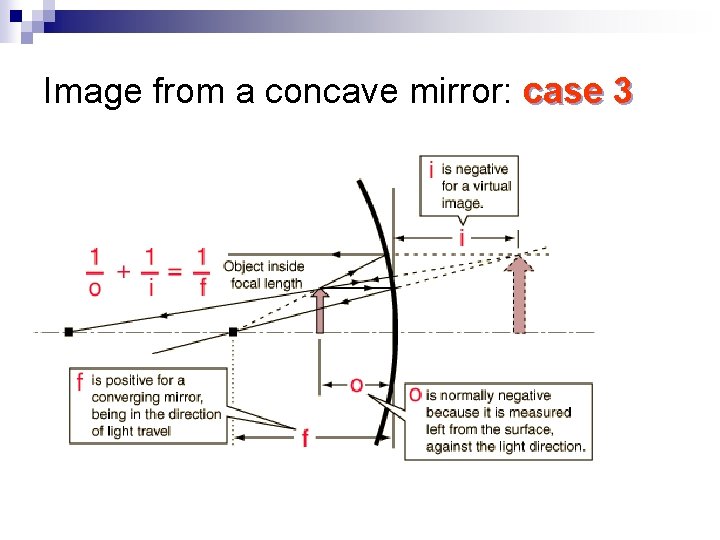
Image from a concave mirror: case 3
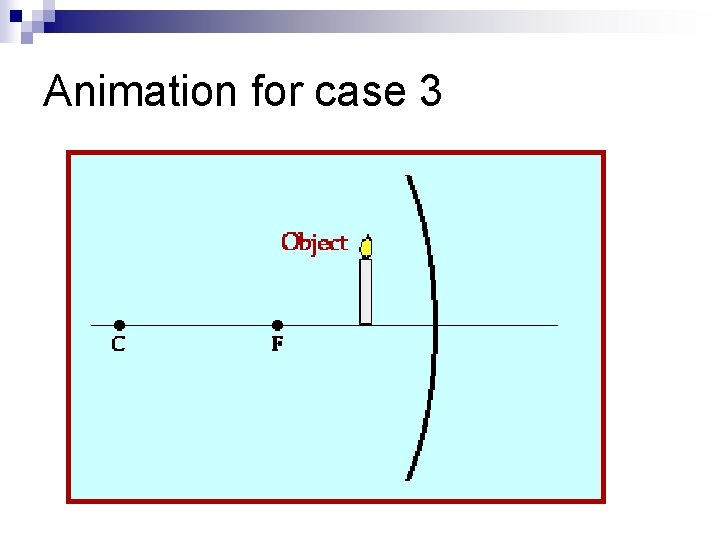
Animation for case 3

Properties of the image n If the object is closer to the mirror than the focal point F, the image is ¨ Virtual, it is behind the mirror ¨ Upright, not inverted ¨ Magnified Can be used for shaving!
Summary

Java Applet n http: //www. microscopy. fsu. edu/primer/java /mirrors/concave. html

Some of the really-cool applications n Suppose you put a point source of light at F. All rays will be reflected back parallel to the axis of mirror: a neat way to construct a parallel beam! ¨ Applications: n flashlight! Headlight in the car. Conversely, if one has parallel rays, all are reflected to pass through F. So all light energy is focused to one point!
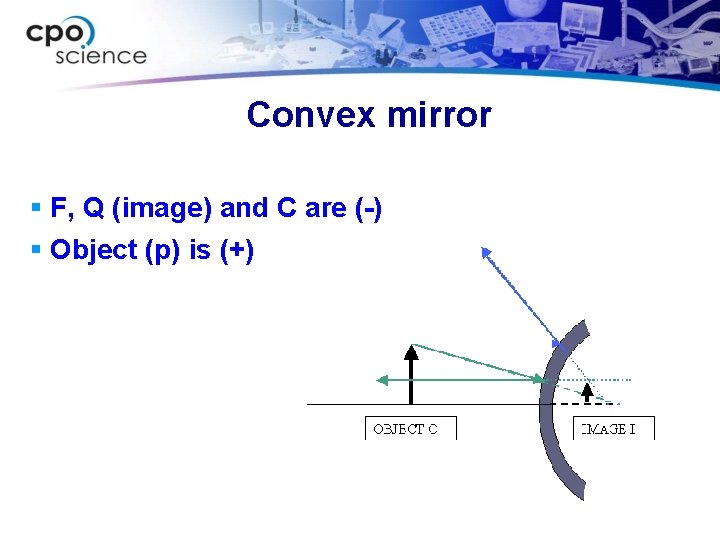
Convex mirror § F, Q (image) and C are (-) § Object (p) is (+)

Magnification § M = h'/h (image height / object height) § M = -Q/P (-image distance / object distance) § Virtual (upright) = “+” § Real (inverted) = “-” § Bigger image = >1 § Smaller image = <1

Ch. 15 Reflection and refraction § When light moves through a material it travels in straight lines. § When light rays travel from one material to another, the rays may reflect. § The light that appears to bounce off the surface of an object is shown by a reflected ray.

Ch. 15 Reflection and refraction § Objects that are in front of a mirror appear as if they are behind the mirror. § This is because light rays are reflected by the mirror. § Your brain perceives the light as if it always traveled in a straight line.

Ch. 15 Reflection and refraction § Another example of refraction of light is the twinkling of a star in the night sky § As starlight travels from space into the Earth’s atmosphere, the rays are refracted. § Since the atmosphere is constantly changing, the amount of refraction also changes.

Ch. 15 Reflection and refraction § The light that bends as it crosses a surface into a material refracts and is shown as a refracted ray.

14 -4 Color and Vision § When all the colors of the rainbow are combined, we do not see any particular color. § We see light without any color. § We call this combination of all the colors of light "white light".

14 -4 Color and Vision § We can think of different colors of light like balls with different kinetic energies. § Blue light has a higher energy than green light, like the balls that make it into the top window. § Red light has the lowest energy, like the balls that can only make it to the lowest window.
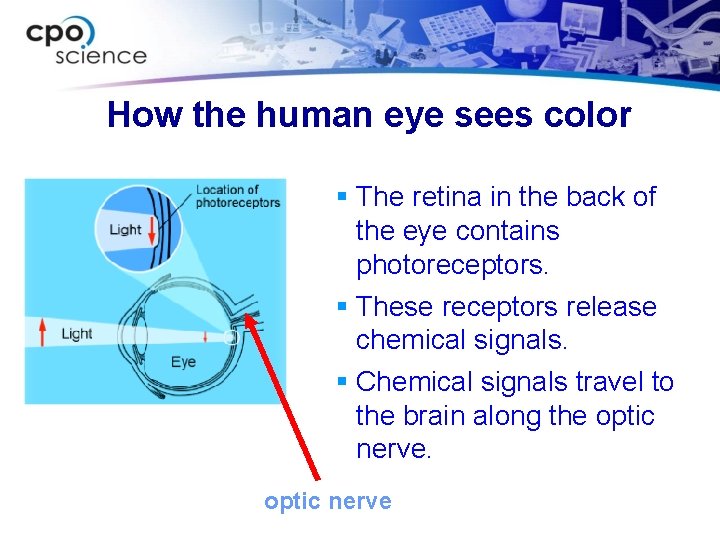
How the human eye sees color § The retina in the back of the eye contains photoreceptors. § These receptors release chemical signals. § Chemical signals travel to the brain along the optic nerve
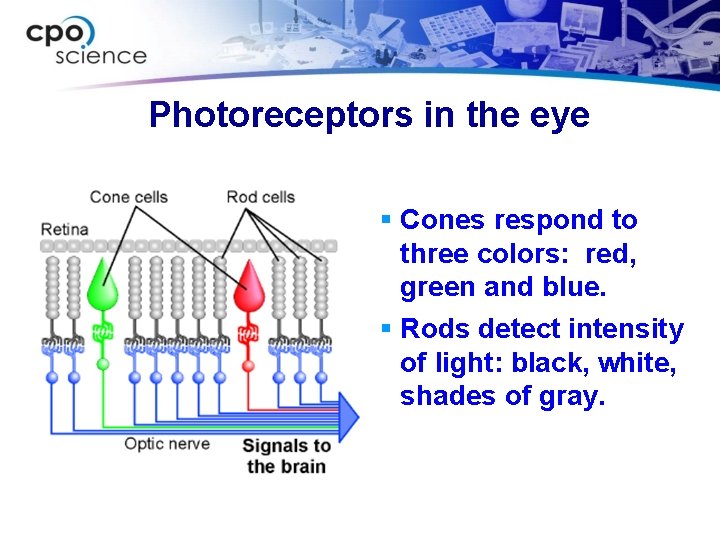
Photoreceptors in the eye § Cones respond to three colors: red, green and blue. § Rods detect intensity of light: black, white, shades of gray.
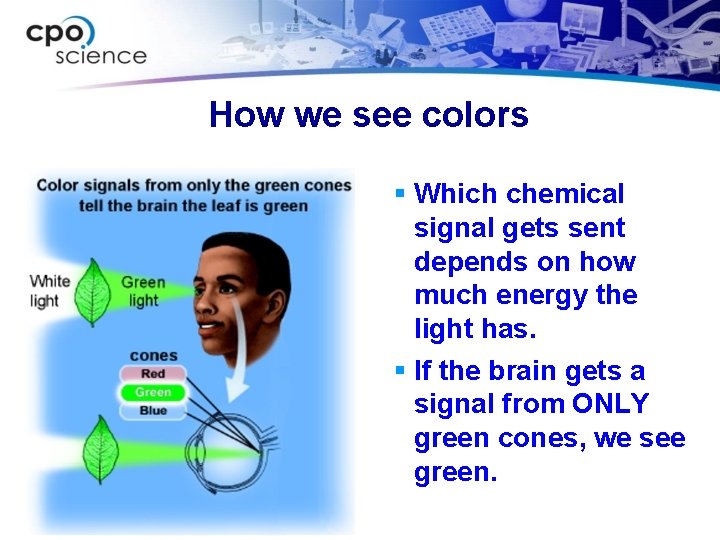
How we see colors § Which chemical signal gets sent depends on how much energy the light has. § If the brain gets a signal from ONLY green cones, we see green.

14 -4 How we see other colors § The three color receptors in the eye allow us to see millions of different colors. § The additive primary colors are red, green, and blue. § We don’t see everything white because the strength of the signal matters. § All the different shades of color we can see are made by changing the proportions of red, green, and blue.

14 -4 How we see the color of things When we see an object, the light that reaches our eyes can come from two different processes: 1. The light can be emitted directly from the object, like a light bulb or glow stick. 2. The light can come from somewhere else, like the sun, and we see the objects by reflected light.

14 -4 How we see the color of things § Colored fabrics and paints get color from a subtractive process. § Chemicals, known as pigments, in the dyes and paints absorb some colors and allow the color you actually see to be reflected. § Magenta, yellow, and cyan are three subtractive primary colors.
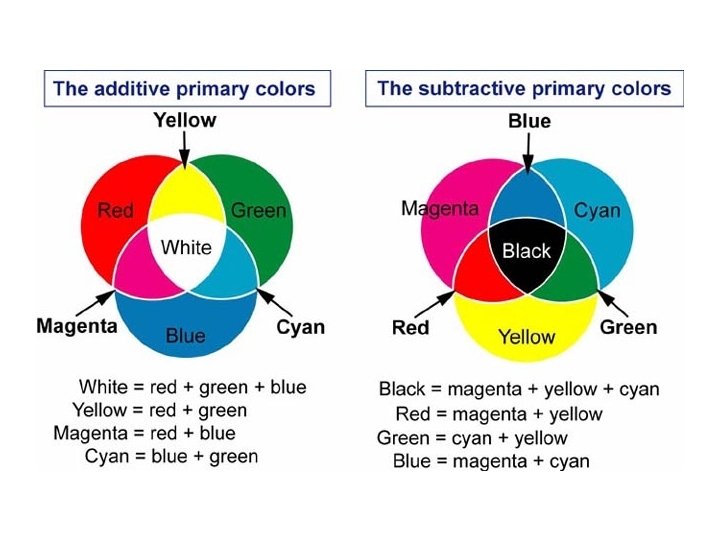

Complementary colors § Add up to make white § Red and cyan § Blue and yellow § Green and magenta § Black and white

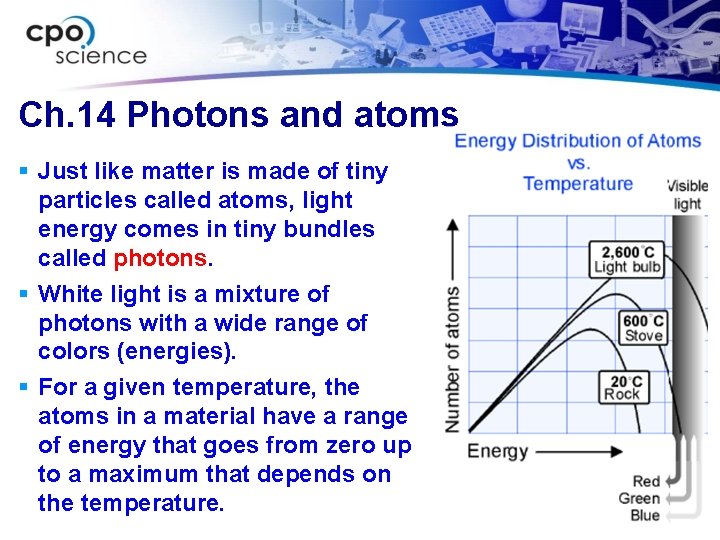
Ch. 14 Photons and atoms § Just like matter is made of tiny particles called atoms, light energy comes in tiny bundles called photons. § White light is a mixture of photons with a wide range of colors (energies). § For a given temperature, the atoms in a material have a range of energy that goes from zero up to a maximum that depends on the temperature.

Ch. 14 Photons and intensity § Intensity measures power per unit area. § There are two ways to make light of high intensity. — One way is to have high- energy photons. — A second way is to have a lot of photons even if they are low-energy. The number and energy of photons determine the intensity of the light.

Ch. 14 Photons and intensity § The light from the flashlight cannot energize phosphorus atoms that your hand blocks. § These atoms will not glow because they did not receive any energy from photons from the flashlight. § The explanation is that each phosphorus atom absorbs (or emits) only one photon at a time.

- Slides: 51