Physics 490 Project Lab Radioactivity Counting Statistics Counting

Physics 490: Project Lab Radioactivity: Counting Statistics

Counting Statistics n Team members: Jack Young Rich Breazeale Ryan Phelan

Contents Apparatus n Experimental Procedure n Analysis n Conclusions n Presentation: Radioactive Decay n
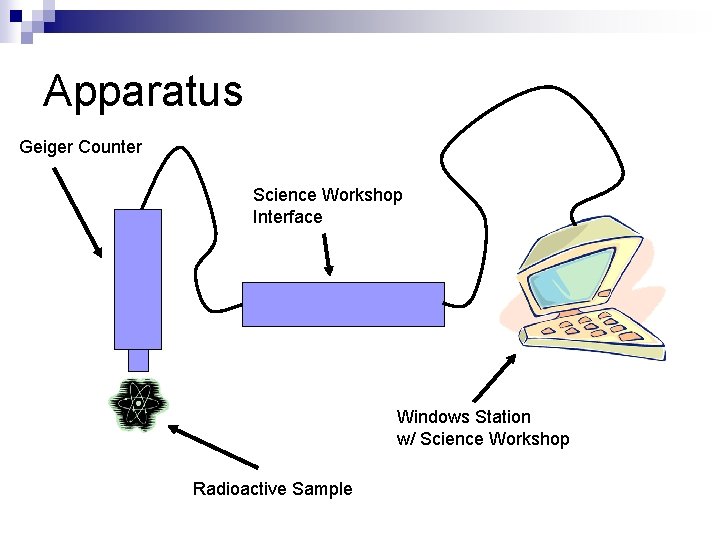
Apparatus Geiger Counter Science Workshop Interface Windows Station w/ Science Workshop Radioactive Sample
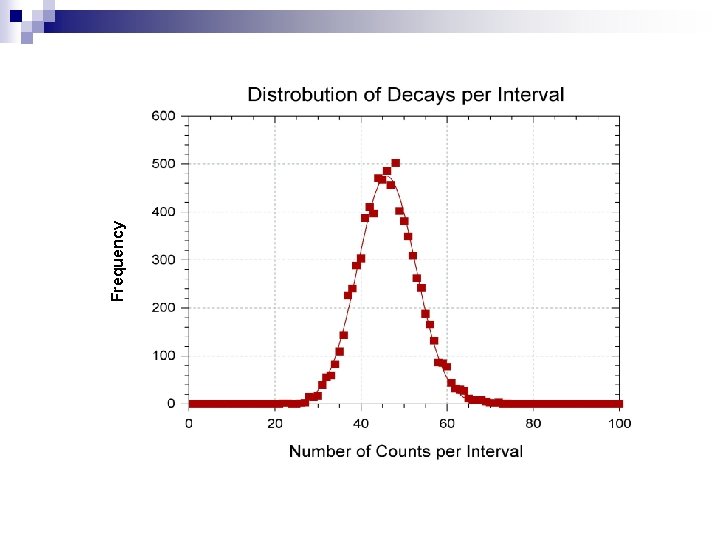
Frequency
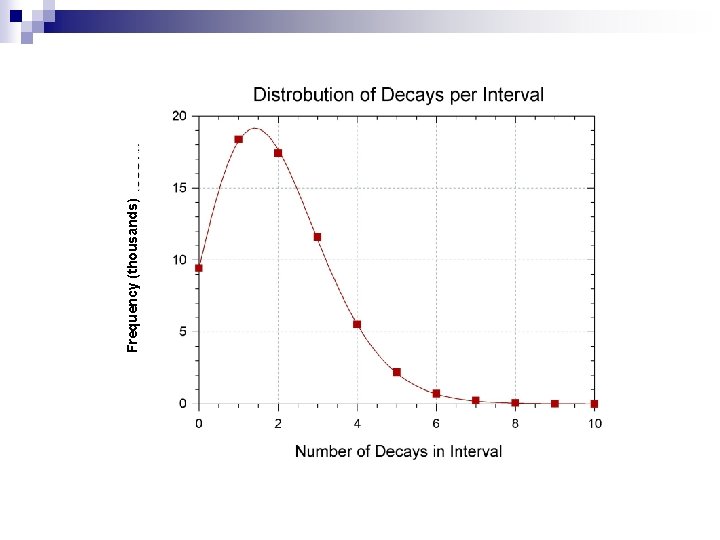
Frequency (thousands)

Radioactive Decay

What is radioactive decay? Radioactive decay is the process by which atomic nuclei change. n There are four different ways this can happen. n Each ejects some particle from the nucleus – all but one create another element. n

Alpha decay n n n Nucleus emits alpha particle – two protons and two neutrons. Atomic number reduced by two. Number of nucleons reduced by four.

Alpha decay n n Our example: U-238 234 + alpha U Th 92 90
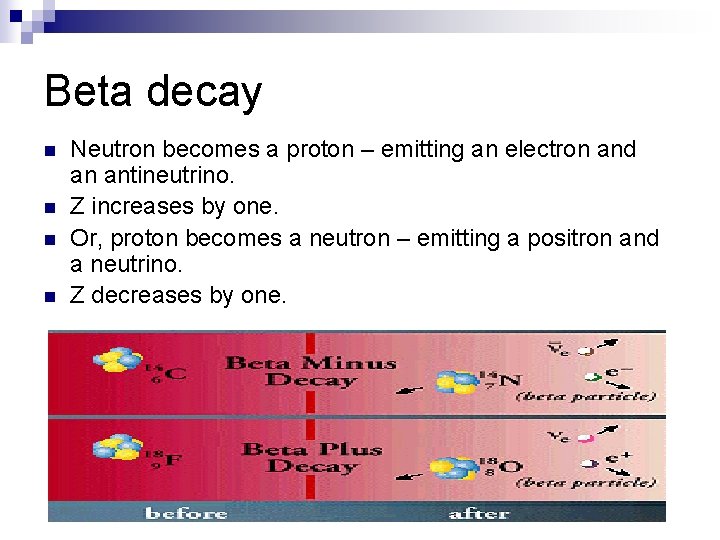
Beta decay n n Neutron becomes a proton – emitting an electron and an antineutrino. Z increases by one. Or, proton becomes a neutron – emitting a positron and a neutrino. Z decreases by one.

Beta decay n n n Our examples: Co-60 and Tl-204 60 + e- + antineutrino Co Ni 27 28 204 + e- + antineutrino Tl Pb 81 82

Gamma decay n n n Energetic nucleus deexcites, emitting a photon (gamma ray). All nuclear numbers remain unchanged. Nucleus in lower energy state after decay.
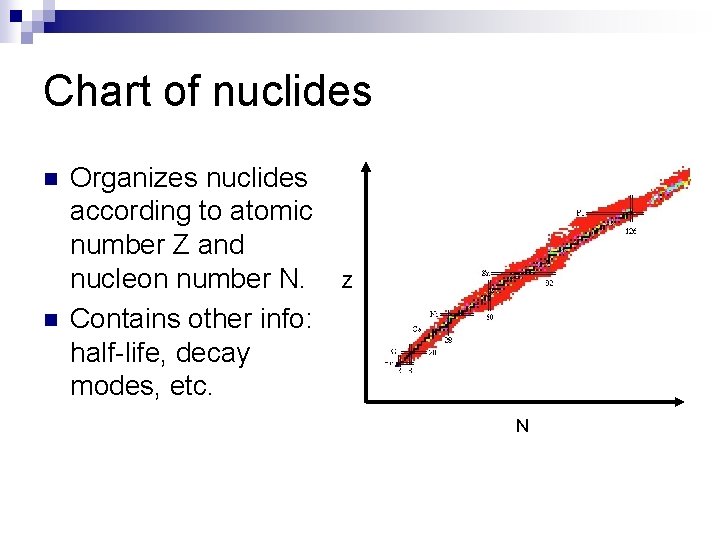
Chart of nuclides n n Organizes nuclides according to atomic number Z and nucleon number N. Contains other info: half-life, decay modes, etc. Z N

Chart of nuclides

Fin
- Slides: 16