Physics 121 Electricity and Magnetism Introduction Syllabus rules

Physics 121: Electricity and Magnetism Introduction Syllabus, rules, assignments, exams, etc. § § Text: Young & Friedman, University Physics Homework & Tutorial System: Mastering Physics Course Content: • • • 5 Weeks: Stationary charges – – Forces, fields, Electric flux, Gauss’ Law, – Potential, potential energy, capacitance 2 Weeks: Moving charges – – Currents, resistance, circuits containing resistance and capacitance, – Kirchoff’s Laws, multi-loop circuits, RC circuits 2 Weeks: Magnetic fields (static fields due to moving charges) – Magnetic force on moving charges, – Magnetic fields caused by currents (Biot-Savart’s and Ampere’e Laws) 2 Weeks: Induction & Inductance – Changing magnetic flux (field) produces currents (Faraday’s Law) – Inductance, LR Circuits 2 – 3 Weeks: AC (LCR) circuits, – Eelectromagnetic oscillations, Resonance – Impedance, Phasors Not covered: – Maxwell’s Equations - unity of electromagnetism – Electromagnetic Waves – light, radio, gamma rays, etc Copyright R. Janow – Fall 2015 1

Physics 121 - Electricity and Magnetism Lecture 01/02 – Vectors, Charge, Coulombs Law Y&F Chapter 21, Sec. 1 - 3 Review of Vectors: • Components in 3 D. Right-hand rule • Scalar multiplication, Dot product, vector product Charge: • Basic properties of Electric Charge • Quantization of Charge • Structure of Matter • Conservation of Charge • Conductors and Insulators • Coulomb’s Law – Force • Shell Theorem • Examples Copyright R. Janow – Fall 2015 2

Definition: Right-Handed Coordinate Systems • • • We always use right-handed coordinate systems. In three-dimensions the righthand rule determines which way the positive axes point. Curl the fingers of your RIGHT HAND so they go from x to y. Your thumb will point in the positive z direction. z y x This course uses several right hand rules related to this one! Copyright R. Janow – Fall 2015

Vectors in 3 dimensions • Unit vector (Cartesian) notation: • Spherical polar coordinate representation: 1 magnitude and 2 directions Rene Descartes 1596 - 1650 z • Conversion into x, y, z components az • Conversion from x, y, z components q ax x Copyright R. Janow – Fall 2015 ay f a sin(q) y

Surface Integral Example: Show that the surface area of a sphere A= 4 p. R 2 by integrating over the sphere’s surface Find d. A – an area segment on the surface of the sphere, then integrate on angles f (azimuth) and q (co-latitude). z Where: • dl is a curved length segment of the circle around the z-axis (along a constant latitude line) • dh is a segment along the q direction (along a constant longitude line) q r f y x Angle range for a full sphere: Factors into 2 simple 1 dimensional integrations Copyright R. Janow – Fall 2015

There are 3 Kinds of Vector Multiplication of a vector by a scalar: vector times scalar vector whose length is multiplied by the scalar Dot product (or Scalar product or Inner product): f - vector times vector scalar - projection of A on B or B on A - commutative unit vectors measure perpendicularity: Copyright R. Janow – Fall 2015

Vector multiplication, continued Cross product (or Vector product or Outer product): - Vector times vector another vector perpendicular to the plane of A and B - Draw A & B tail to tail: right hand rule shows direction of C f Ø - If A and B are parallel or the same, A x B = 0 - If A and B are perpendicular, A x B = AB (max) Algebra: Unit vector representation: Applications: Copyright R. Janow – Fall 2015 i j k
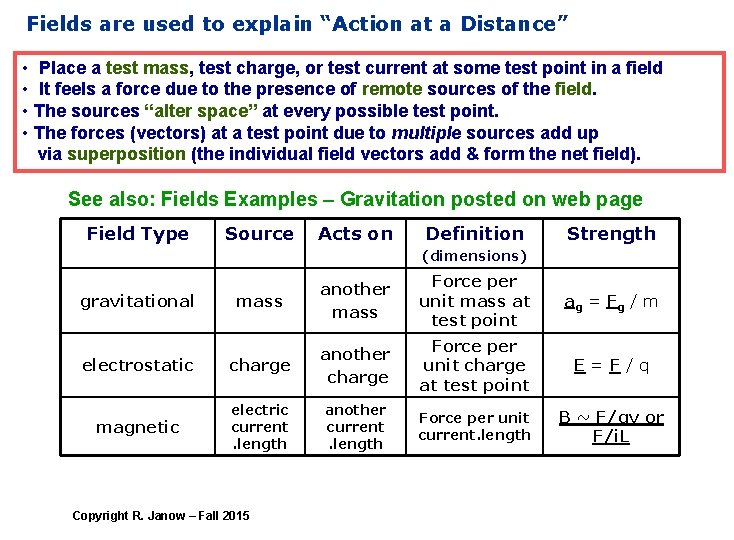
Fields are used to explain “Action at a Distance” • Place a test mass, test charge, or test current at some test point in a field • It feels a force due to the presence of remote sources of the field. • The sources “alter space” at every possible test point. • The forces (vectors) at a test point due to multiple sources add up via superposition (the individual field vectors add & form the net field). See also: Fields Examples – Gravitation posted on web page Field Type Source Acts on Definition Strength (dimensions) mass another mass Force per unit mass at test point a g = Fg / m electrostatic charge another charge Force per unit charge at test point E=F/q magnetic electric current. length another current. length Force per unit current. length B ~ F/qv or F/i. L gravitational Copyright R. Janow – Fall 2015
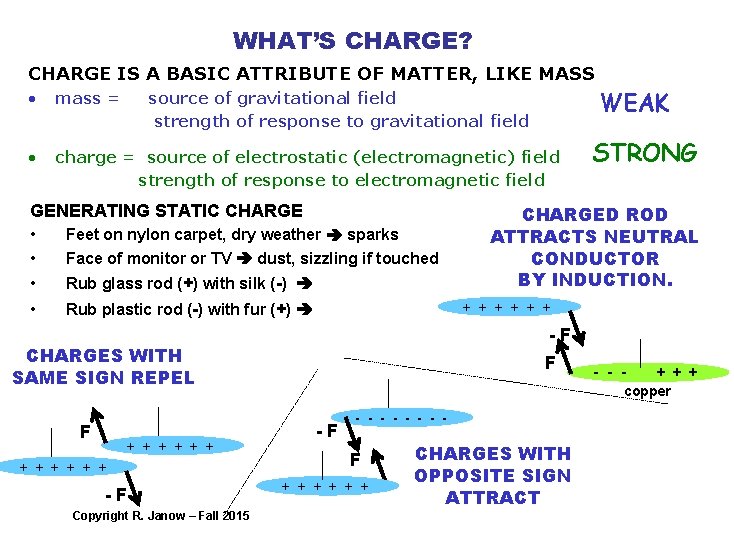
WHAT’S CHARGE? CHARGE IS A BASIC ATTRIBUTE OF MATTER, LIKE MASS WEAK • mass = • charge = source of electrostatic (electromagnetic) field strength of response to electromagnetic field source of gravitational field strength of response to gravitational field GENERATING STATIC CHARGE • • Feet on nylon carpet, dry weather sparks Face of monitor or TV dust, sizzling if touched • Rub glass rod (+) with silk (-) • Rub plastic rod (-) with fur (+) + + + -F Copyright R. Janow – Fall 2015 CHARGED ROD ATTRACTS NEUTRAL CONDUCTOR BY INDUCTION. + + + -F CHARGES WITH SAME SIGN REPEL F STRONG F -F - - - - F + + + CHARGES WITH OPPOSITE SIGN ATTRACT - - - + + + copper

CHARGE IS A “CONSERVED” QUANTITY LIKE ENERGY, MOMENTUM, ANGULAR MOMENTUM, usually MASS • • • Net charge never just disappears or appears from nowhere. Charges can be cancelled or screened by those of opposite sign All of chemistry is about exchanging & conserving charge among atoms & molecules Nuclear reactions conserve charge, e. g. radioactive decay: 92 U 238 90 Th 234 + 2 He 4 Pair Production & annihilation: g e- + e+ (electron – positron pair) ALL MATTER IS ELECTRICAL… … BUT…matter is normally electrically neutral (equal numbers of + and – charges). (Why? ) OBJECTS WITH A NET CHARGE HAVE LOST OR GAINED SOME BASIC CHARGES • Net + charge deficit of electrons (excess of “holes”) • Net – charge excess electrons (deficit of “holes”). • Why isn’t all matter a gas? Electrical forces hold matter together. • Outside neutral objects electrical forces are “screened”, but not completely • Weak “leftover” electrical force (Van der Waal’s) makes atoms stick together & defines properties of solids, liquids, and gases. Copyright R. Janow – Fall 2015

The physical world we see is electrical MODERN ATOMIC PHYSICS (1900 – 1932): Rutherford, Bohr • Charge is quantized: e = 1. 6 x 10 -19 coulombs (small). • • Electrons ( -e) are in stable orbital clouds with radius ~ 10 -10 m. , • • mass me = 9. 1 x 10 -31 kg. (small). Atoms are mostly space. Protons ( +e) are in a small nucleus, r ~10 -15 m , mp= 1. 67 x 10 -27 kg, • • • Every charge is an exact multiple of +/- e. Milliken ~1900. Ze = nuclear charge, Z = atomic number. There are neutrons too. A neutral atom has equal # of electrons and protons. An ion is charged + or – and will attract opposite charges to become neutral Electron Proton Neutron Copyright R. Janow – Fall 2015 2 -1: Which type of charge is easiest to pull out of an atom? A. Proton B. Electron The protons all repel each other Why don’t nuclei all fly apart?
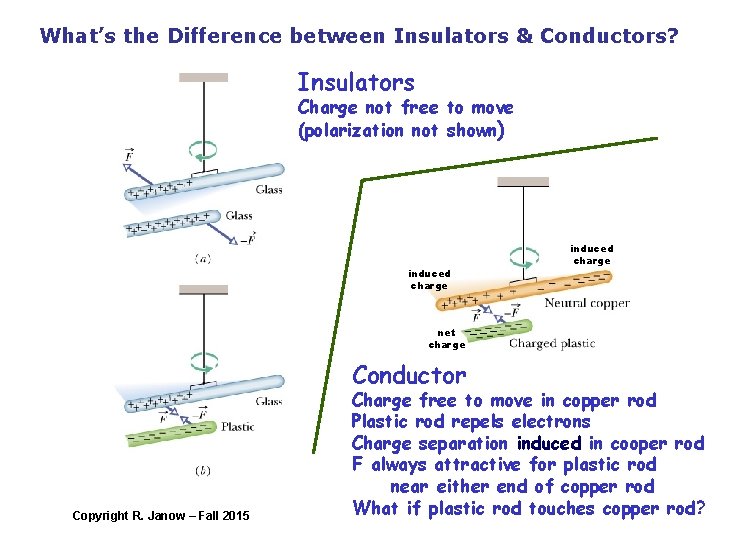
What’s the Difference between Insulators & Conductors? Insulators Charge not free to move (polarization not shown) induced charge net charge Conductor Copyright R. Janow – Fall 2015 Charge free to move in copper rod Plastic rod repels electrons Charge separation induced in cooper rod F always attractive for plastic rod near either end of copper rod What if plastic rod touches copper rod?
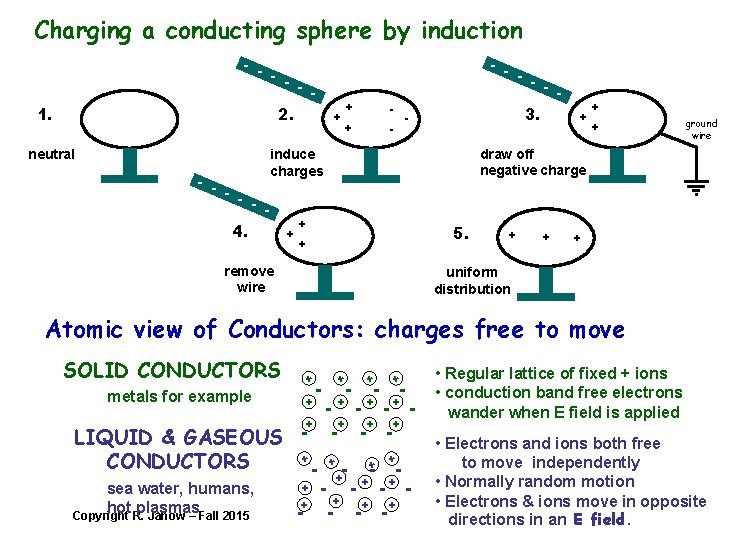
Charging a conducting sphere by induction - - - - 1. 2. neutral - - + + - 3. - + + + ground wire draw off negative charge induce charges 4. + + 5. + remove wire + + + uniform distribution Atomic view of Conductors: charges free to move SOLID CONDUCTORS + metals for example LIQUID & GASEOUS CONDUCTORS sea water, humans, hot plasmas Copyright R. Janow – Fall 2015 + - + + - - + - + + + - + - - - + + + - - - + - • Regular lattice of fixed + ions • conduction band free electrons wander when E field is applied • Electrons and ions both free to move independently • Normally random motion • Electrons & ions move in opposite directions in an E field.
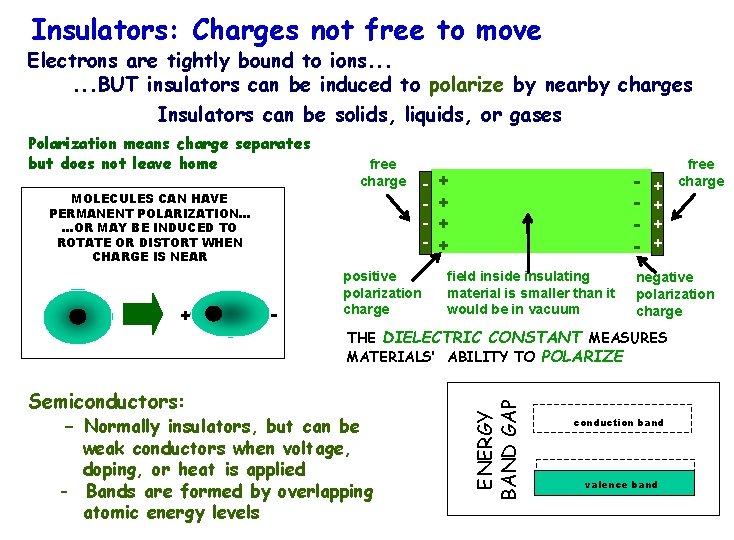
Insulators: Charges not free to move Electrons are tightly bound to ions. . . BUT insulators can be induced to polarize by nearby charges Insulators can be solids, liquids, or gases free charge MOLECULES CAN HAVE PERMANENT POLARIZATION. . . OR MAY BE INDUCED TO ROTATE OR DISTORT WHEN CHARGE IS NEAR + - - positive polarization charge - + + field inside insulating material is smaller than it would be in vacuum + + Semiconductors: - Normally insulators, but can be weak conductors when voltage, doping, or heat is applied - Bands are formed by overlapping atomic. R. energy levels Copyright Janow – Fall 2015 free charge negative polarization charge THE DIELECTRIC CONSTANT MEASURES MATERIALS’ ABILITY TO POLARIZE ENERGY BAND GAP Polarization means charge separates but does not leave home conduction band valence band

ELECTROSTATIC FORCE LAW (coulomb, 1785) 3 rd law pair of forces F 12 q 1 Constant k=8. 89 x 109 Nm 2/coul 2 r 12 repulsion shown q 2 F 21 Force on q 1 due to q 2 (magnitude) • Force is between pairs of point charges • An electric field transmits the force (no contact, action at a distance) • Symmetric in q 1 & q 2 so F 12 = - F 21 • Inverse square law • Electrical forces are strong compared to gravitation • e 0= 8. 85 x 10 -12 Why this value? Units Unit of charge = Coulomb 1 Coulomb = charge passing through a cross section of a wire carrying 1 Ampere of current in 1 second 1 Coulomb = 6. 24 x 1018 electrons [ = 1/e] Copyright R. Janow – Fall 2015 Gravitation is weak G m 1 m 2 F 12 = 2 r 12 G» 6. 67 x 10 11 N. m 2 / kg 2

Coulomb’s Law using vector notation Force on q 1 due to q 2 Displacement y to q 1 from q 2 Unit vector pointing radially away from q 2 at location of q 1 Sketch shows repulsion: q 1 q 2 For attraction (opposite charges): cubed Copyright R. Janow – Fall 2015 x squared

Superposition of forces & fields The electrostatic force between a specific pair of point charges does not depend on interaction with other charges that may be nearby – there are no 3 -body forces (same as gravitation) Example: Find NET force on q 1 y +q 2 Method: find forces for all pairs of charges involving q 1, then add the forces vectorially +q 3 +q 1 x • • • F 11 is meaningless Use coulomb’s law to calculate individual forces -q 4 Keep track of direction, usually using unit vectors Find the vector sum of individual forces at the point For continuous charge distributions, integrate instead of summing Copyright R. Janow – Fall 2015

Example: Charges in a Line 2 -4: Where do I have to place the +Q charge in order for the forces on it to balance, in the figure at right? A. Cannot tell, because + charge value is not given. B. Exactly in the middle between the two negative charges. C. On the line between the two negative charges, but closer to the -2 q charge. D. On the line between the two negative charges, but closer to the -q charge. E. There is no location that will balance the forces. Copyright R. Janow – Fall 2015 -2 q +Q -q

Calculate the Exact Location for the forces to balance • • • Force on test charge Q is attractive toward both negative charges, hence they could cancel. By symmetry, position for +Q in equilibrium is along y-axis Coordinate system: call the total distance L and call y the position of charge +Q from charge -q. Net force is sum of the two force vectors, and has to be zero, so k, q, and Q all cancel due to zero on the right, so our answer does not depend on knowing the charge values. We end up with Solving for y, , y is less than half-way to the top negative charge Copyright R. Janow – Fall 2015 -2 q +Q L y -q

How do we know that electrostatic force - not gravitational force - holds atoms & molecules together? Example: Find the ratio of forces in Hydrogen • 1 proton in nucleus, neutral Hydrogen atom has 1 electron • Both particles have charge e = 1. 6 x 10 -19 C. • me = electron mass = 9. 1 x 10 -31 kg. • mp = proton mass = 1833 me -8 -10 m. • a 0 = radius of electron orbit = 10 cm = 10 • Electrostatics dominates atomic structure by a factor of ~ 1039 • So why is gravitation important at all? Matter is normally Neutral! • Why don’t atomic nuclei with several protons break apart? Copyright R. Janow – Fall 2015

Example: Forces on an electron and two protons along a line (1 D) 1 2 r 12 r 23 p e 3 p By symmetry, all forces lie along the horizontal axis Show forces on each of the objects in free body diagrams for # 1, 2, & 3 F 12 FBD for 1 F 13 F 21 FBD for 2 FBD for 3 • • F 23 F 31 F 32 There are 3 action-reaction pairs of forces Which forces are attractive/repulsive? Could net force on any individual charge be zero above? What if r 12 = r 23? Copyright R. Janow – Fall 2015

EXAMPLE: Vector-based solution… … Start with two point charges on x-axis Find F 12 - the Force on q 1 due to q 2 F 21 has the same magnitude, opposite direction F 12 F 21 R q 1 Let: q 2 q 1 = +e = +1. 6 x 10 -19 C q 2 = +2 e = +3. 2 x 10 -19 C. R = 0. 02 m Magnitude of force: small! In vector notation: Copyright R. Janow – Fall 2015 x
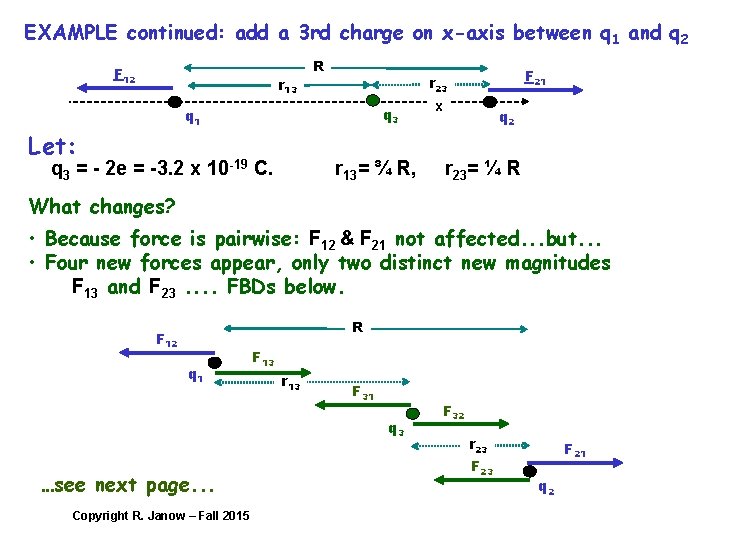
EXAMPLE continued: add a 3 rd charge on x-axis between q 1 and q 2 F 12 r 13 R q 3 q 1 Let: q 3 = - 2 e = -3. 2 x 10 -19 C. F 21 r 23 r 13= ¾ R, x q 2 r 23= ¼ R What changes? • Because force is pairwise: F 12 & F 21 not affected. . . but. . . • Four new forces appear, only two distinct new magnitudes F 13 and F 23. . FBDs below. R F 12 q 1 F 13 r 13 F 31 q 3 …see next page. . . Copyright R. Janow – Fall 2015 F 32 r 23 F 21 q 2

EXAMPLE continued: calculate magnitudes Find net forces on each of the three charges by applying superposition On 1: On 2: On 3: Copyright R. Janow – Fall 2015
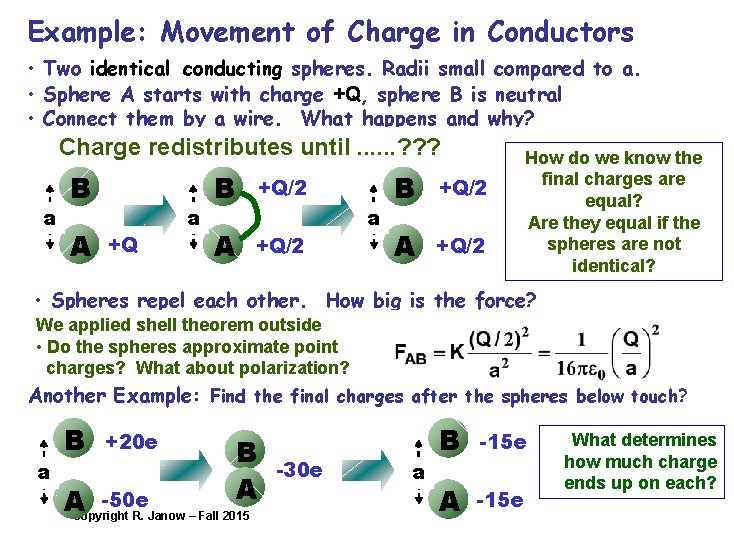
Example: Movement of Charge in Conductors • Two identical conducting spheres. Radii small compared to a. • Sphere A starts with charge +Q, sphere B is neutral • Connect them by a wire. What happens and why? Charge redistributes until. . . ? ? ? a B A +Q a B +Q/2 A +Q/2 How do we know the final charges are equal? Are they equal if the spheres are not identical? • Spheres repel each other. How big is the force? We applied shell theorem outside • Do the spheres approximate point charges? What about polarization? Another Example: Find the final charges after the spheres below touch? a B +20 e B A -50 e ACopyright R. Janow – Fall 2015 -30 e a B -15 e A -15 e What determines how much charge ends up on each?

What force does a fixed spherical charge distribution exert on a point test charge nearby? + + + + + R + + + r + q P Proof by lengthy integration or symmetry + Gauss Law + Shell Theorem (Similar to gravitational Shell Theorem) • Holds for spherical symmetry only: shell or solid sphere • Uniform surface charge density s - not free to move (insulator) • Shell has radius R, total charge Q • Outside a shell, r > R: Uniform shell responds to charged particle q as if all the shell’s charge is a point charge at the center of the shell. Inside a shell, r < R: Charged particle q inside uniform hollow shell of charge feels zero net electrostatic force from the shell. Why Copyright does the shell theorem work? spherical symmetry cancellations R. Janow – Fall 2015

Charge on a spherical conductor – charge free to move • • Net charge Q on a spherical shell spreads out everywhere over the surface. Tiny free charges inside the conductor push on the others and move as far apart as they can go. When do they stop moving? If no other charges are nearby (“isolated” system) • • Charges would spread themselves out uniformly because of symmetry Sphere would act as if all of it’s charge is concentrated at the center (for points outside the sphere) as in the Shell Theorem. Bring test charge q<<Q near the sphere q Q 2 R d Q is a point charge here q Q d The test charge induces polarization charge on the sphere • Charge distribution will NOT be uniform any more • Extra surface charge is induced on the near and far surfaces of the sphere • The net force will be slightly more attractive than for point charges The Shell Theorem works approximately for conductors if the test charge induces very little polarization; e. g. , if q << Q and/or d >> R. Copyright R. Janow – Fall 2015

Example: three charges along x-axis y x L q 1 Region III qprot q 2 Region I q 1 = + 8 q q not stated q 2 = -2 q qproton = +e Region II For q 1 & q 2 as in the sketch where should a proton (test charge) be placed to be in equilibrium (Fnet = 0, use “free body diagrams” & symmetry) Both forces to the right No equilibrium possible Proton in Region I: qprot Proton in Region III: Forces opposed but Proton in Region II: Forces opposed and qprot Forces can never balance So balance point exists here Equilibrium x: Did the. R. test charge magnitude Copyright Janow – Fall 2015 make any difference?
- Slides: 28