Physics 1161 PreLecture 33 34 Nuclear Binding Radioactivity

Physics 1161: Pre-Lecture 33 - 34 Nuclear Binding, Radioactivity

Strong Nuclear Force Hydrogen atom: Binding energy =13. 6 e. V (of electron to nucleus) Coulomb force proton electron proton neutron Simplest Nucleus: Deuteron=neutron+proton Very strong force Binding energy of deuteron = 2. 2 Mev! That’s around 200, 000 times bigger! or
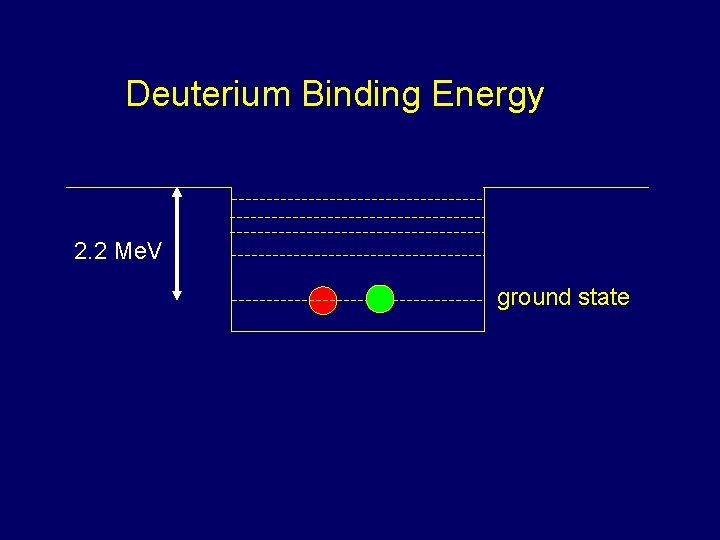
Deuterium Binding Energy 2. 2 Me. V ground state

Nuclei have energy levels — just like atoms energy needed to remove a proton from 12 C is 16. 0 Me. V energy needed to remove a neutron from 12 C is 18. 7 Me. V 12 C energy levels Note the energy scale is Me. V rather than e. V

Smaller is Bigger! Comparing Nuclear and Atomic sizes Hydrogen Atom: Bohr radius = Nucleus with nucl number A: A has radius Z Note the TREMENDOUS difference Nucleus is 104 times smaller and binding energy is 105 times larger!

Binding Energy Einstein’s famous equation E = m c 2 proton: mc 2=(1. 67 x 10 -27 kg)(3 x 108 m/s)2=1. 50 x 10 -10 J Proton: mc 2 = 938. 3 Me. V Neutron: mc 2= 939. 5 Me. V Deuteron: mc 2 =1875. 6 Me. V Adding these, get 1877. 8 Me. V Difference is Binding energy, 2. 2 Me. V MDeuteron = MProton + MNeutron – |Binding Energy|
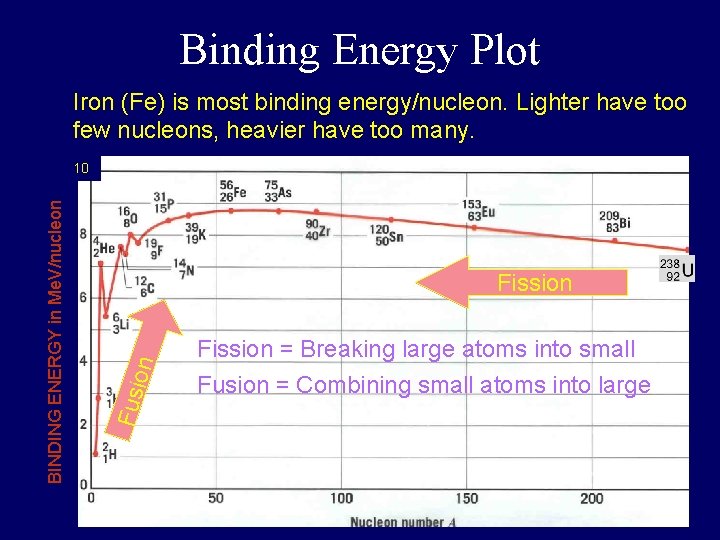
Binding Energy Plot Iron (Fe) is most binding energy/nucleon. Lighter have too few nucleons, heavier have too many. on Fission Fusi BINDING ENERGY in Me. V/nucleon 10 Fission = Breaking large atoms into small Fusion = Combining small atoms into large

3 Types of Radioactivity B field into screen detector Radioactive sources a particles: nuclei b- particles: electrons Easily Stopped by metal g : photons (more energetic than x-rays) penetrate! 26

Decay Rules 1) Nucleon Number is conserved. 2) Atomic Number (charge) is conserved. 3) Energy and momentum are conserved. : example 1) 238 = 234 + 4 2) 92 = 90 + 2 recall Nucleon number conserved Charge conserved : example g: example Needed to conserve momentum.

Decay Function time

Radioactivity Quantitatively No. of nuclei present Decays per second, or “activity” decay constant Survival: No. of nuclei present at time t Instead of base e we can use base 2: where Half life Then we can write No. we started with at t=0

You are radioactive! One in 8. 3 x 1011 carbon atoms is 14 C which - decays with a ½ life of 5730 years. Determine # of decays/gram of Carbon.

Summary • Nuclear Reactions – – – Nucleon number conserved Charge conserved Energy/Momentum conserved a particles = nucleii b- particles = electrons g particles = high-energy photons Survival: • Decays – Half-Life is time for ½ of atoms to decay
- Slides: 13