Physics 101 Lecture 14 Thermal Physics Entropy Probability

Physics 101 Lecture 14

Thermal Physics Entropy, Probability Third Physics

What is Entropy? • Measure of probability of state of a system – State with high probability has high entropy – State with low probability has low entropy – On macroscopic scale time points in direction of increasing entropy • Measure of the disorder of a system – System with high disorder has high entropy – System with low disorder (high order) has low entropy • Heat divided by Kelvin temperature
Boltzman Paradox Third Physics
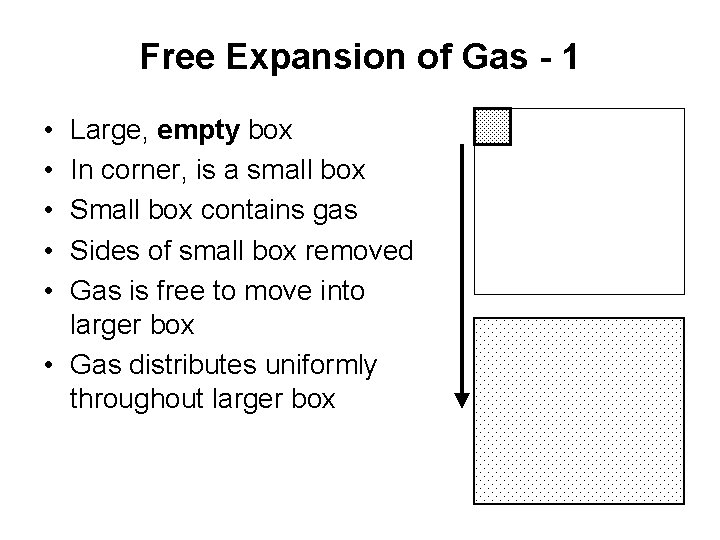
Free Expansion of Gas - 1 • • • Large, empty box In corner, is a small box Small box contains gas Sides of small box removed Gas is free to move into larger box • Gas distributes uniformly throughout larger box

Free Expansion of Gas - 2 • Small box opened at time ti • Each gas molecule had an initial position and velocity • Gas has filled the larger room at time tf • Final position and velocity of each molecule have values related to the initial values of the positions and velocities
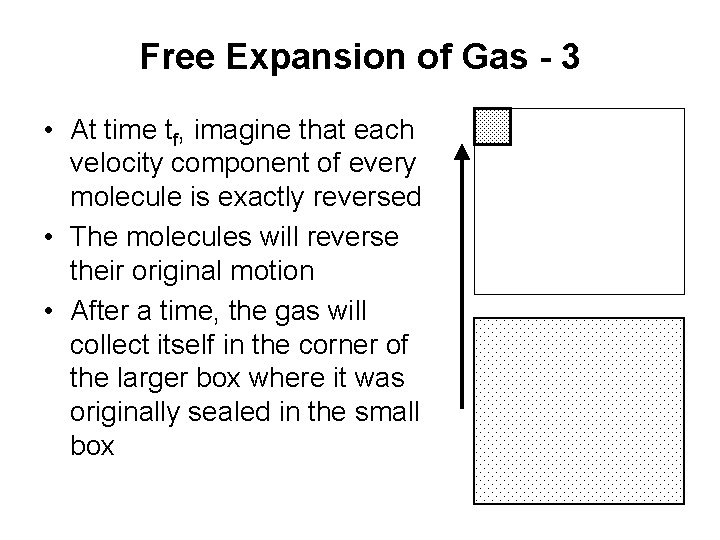
Free Expansion of Gas - 3 • At time tf, imagine that each velocity component of every molecule is exactly reversed • The molecules will reverse their original motion • After a time, the gas will collect itself in the corner of the larger box where it was originally sealed in the small box

Boltzmann Paradox • No reason the motion which led to the uniform filling of the large box is any more probable than the same motion reversed which leads to the collection of the gas in one corner of large box • Why is it that we never observe the air in a large box collecting in one particular portion of the large box? • Fact that we never observe some motions of a system which are just as probable as those we do observe is called the Boltzmann paradox

Boltzmann Paradox Solution - 1 • It is true that any exactly specified motion of the molecules has the same probability as any other exactly specified motion • It is also true that of all the possible exactly specified motions of a group of molecules, the total number of these motions which lead to the uniform filling of the available space is enormously greater than the number of these motions which leads to the occupation of only a small part of the available space

Boltzmann Paradox Solution - 2 • Although each individual motion of the system has the same probability, the probability of observing the uniform filling is overwhelmingly large compared with the probability of any other observation • It is difficult to imagine the detailed motion of even one particle, much less that of many particles • Fortunately, for the calculation we do not have to deal with the motions of the particles, but only with the number of ways of distributing the particles in a given volume.

Probabilities Dice

Definitions • Microstate – a description of a system that specifies the position and / or velocity of each individual particle • Macrostate – a more generalized description of the system – it can be in terms of macroscopic quantities such as pressure or volume – it can be in terms of the number of particles whose properties fall within a given range • Multiplicity (W) – the number microstates corresponding to the same macrostate • Every microstate is equally likely to occur
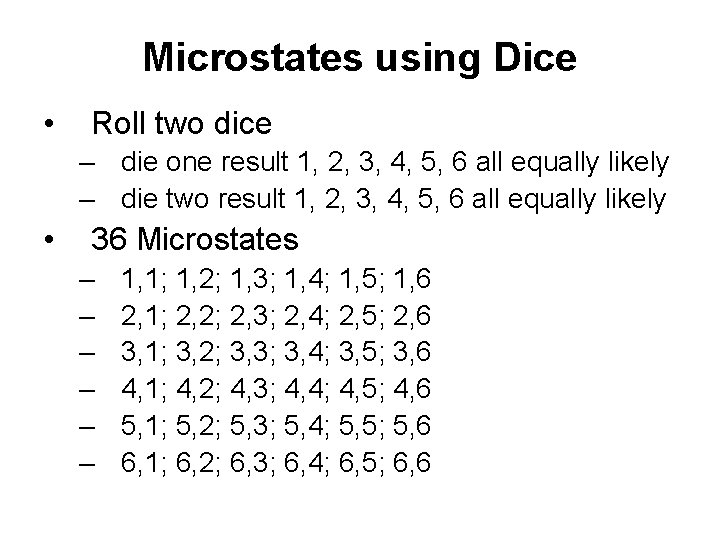
Microstates using Dice • Roll two dice – die one result 1, 2, 3, 4, 5, 6 all equally likely – die two result 1, 2, 3, 4, 5, 6 all equally likely • 36 Microstates – – – 1, 1; 1, 2; 1, 3; 1, 4; 1, 5; 1, 6 2, 1; 2, 2; 2, 3; 2, 4; 2, 5; 2, 6 3, 1; 3, 2; 3, 3; 3, 4; 3, 5; 3, 6 4, 1; 4, 2; 4, 3; 4, 4; 4, 5; 4, 6 5, 1; 5, 2; 5, 3; 5, 4; 5, 5; 5, 6 6, 1; 6, 2; 6, 3; 6, 4; 6, 5; 6, 6

Macrostates using Dice - 1 • Macrostate dice sum = 1 – Macrostate multiplicity = 0 • Macrostate dice sum = 2 – 1, 1 – Macrostate multiplicity = 1 • Macrostate dice sum = 3 – 1, 2; 2; 1 – Macrostate multiplicity = 2

Macrostates using Dice - 2 • Macrostate dice sum = 4 – 1, 3; 2, 2; 3, 1 – Macrostate multiplicity = 3 • Macrostate dice sum = 5 – 1, 4; 2, 3; 4, 1; 3, 2 – Macrostate multiplicity = 4 • Macrostate dice sum = 6 – 1, 5; 2, 4; 3, 3; 5, 1; 4, 2 – Macrostate multiplicity = 5

Macrostates using Dice - 3 • Macrostate dice sum = 7 – 1, 6; 2, 5; 3, 4; 6, 1; 5, 2; 4, 3 – Macrostate multiplicity = 6 • Macrostate dice sum = 8 – 2, 6; 3, 5; 4, 4; 6, 2; 5, 3 – Macrostate multiplicity = 5 • Macrostate dice sum = 9 – 3, 6; 4, 5; 6, 3; 5, 4 – Macrostate multiplicity = 4

Macrostates using Dice - 4 • Macrostate dice sum = 10 – 4, 6; 5, 5; 6, 4 – Macrostate multiplicity = 3 • Macrostate dice sum = 11 – 5, 6; 6, 5 – Macrostate multiplicity = 2 • Macrostate dice sum = 12 – 6, 6 – Macrostate multiplicity = 1

Probability / Multiplicity Molecules
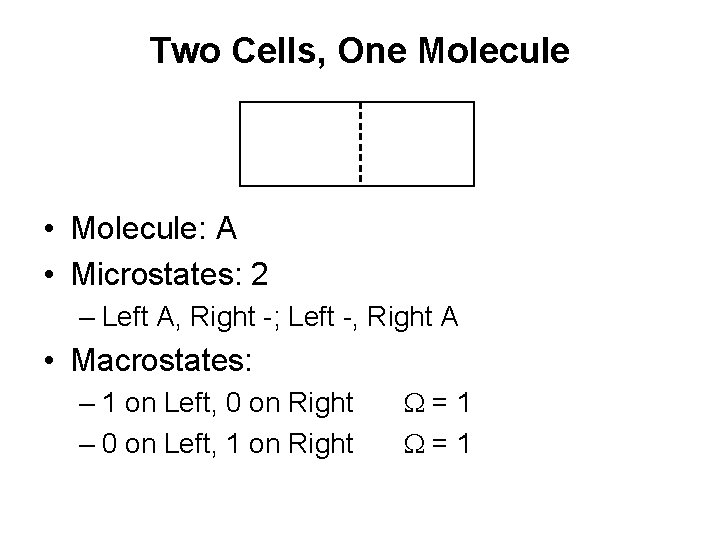
Two Cells, One Molecule • Molecule: A • Microstates: 2 – Left A, Right -; Left -, Right A • Macrostates: – 1 on Left, 0 on Right – 0 on Left, 1 on Right W=1
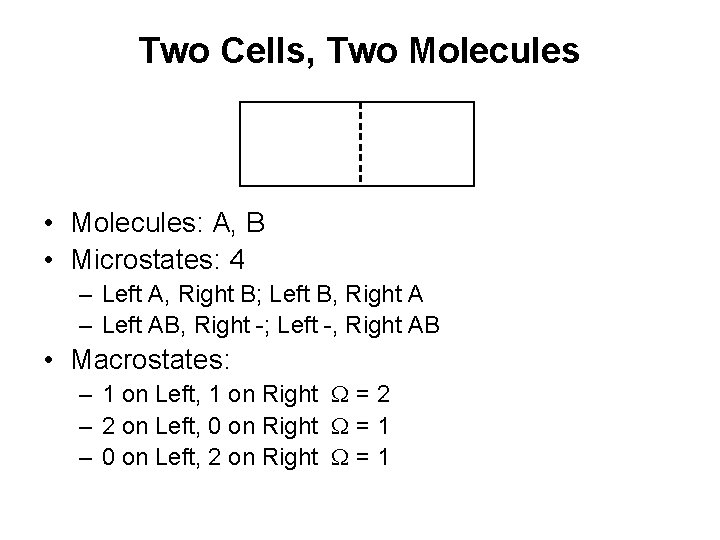
Two Cells, Two Molecules • Molecules: A, B • Microstates: 4 – Left A, Right B; Left B, Right A – Left AB, Right -; Left -, Right AB • Macrostates: – 1 on Left, 1 on Right W = 2 – 2 on Left, 0 on Right W = 1 – 0 on Left, 2 on Right W = 1

Two Cells, Three Molecules • Molecules: A, B, C • Microstates: 8 – Left ABC, Right –; Left -, Right ABC – Left AB, Right C; Left AC, Right B; Left BC, Right A – Left A, Right BC; Left B, Right AC; Left C, Right BC • Macrostates: – – 3 on Left, 0 on Right 0 on Left, 3 on Right 2 on Left, 1 on Right 1 on Left, 2 on Right W=1 W=3
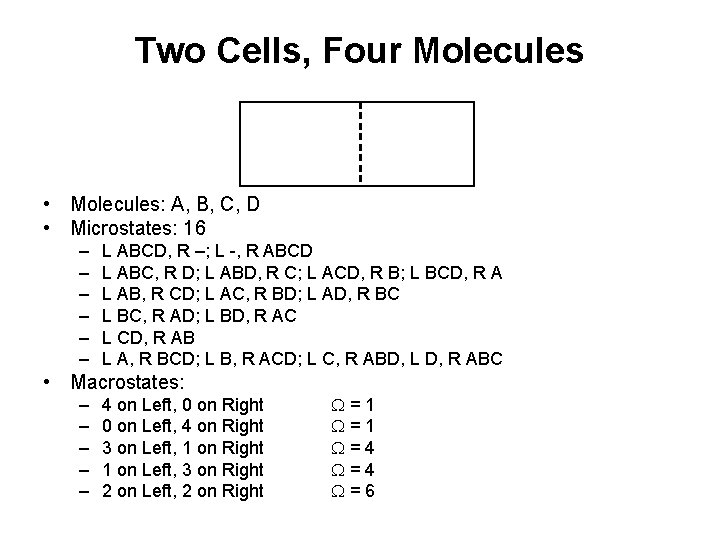
Two Cells, Four Molecules • Molecules: A, B, C, D • Microstates: 16 – – – L ABCD, R –; L -, R ABCD L ABC, R D; L ABD, R C; L ACD, R B; L BCD, R A L AB, R CD; L AC, R BD; L AD, R BC L BC, R AD; L BD, R AC L CD, R AB L A, R BCD; L B, R ACD; L C, R ABD, L D, R ABC • Macrostates: – – – 4 on Left, 0 on Right 0 on Left, 4 on Right 3 on Left, 1 on Right 1 on Left, 3 on Right 2 on Left, 2 on Right W=1 W=4 W=6
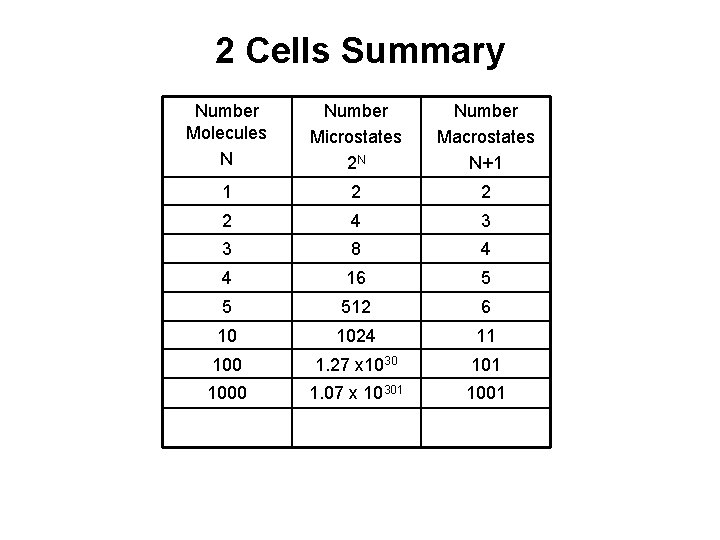
2 Cells Summary Number Molecules N Number Microstates 2 N Number Macrostates N+1 1 2 2 2 4 3 3 8 4 4 16 5 5 512 6 10 1024 11 100 1. 27 x 1030 101 1000 1. 07 x 10301 1001

2 Cells Multiplicity of Macrostates - 1 • N = number of molecules • n = number of molecules on the left side • Multiplicity of macrostates is – WN, n = N! / n!(N-n)! • Probability of a macrostate is – PN, n = N! / n!(N-n)!2 N

2 Cells Multiplicity of Macrostates - 2 • N=1 – W 1, 0 = 1 – W 1, 1 = 1 • N=2 – W 2, 0 = 1 – W 2, 1 = 2 – W 2, 2 = 1 • N=3 – – W 3, 0 = 1 W 3, 1 = 3 W 3, 2 = 3 W 3, 3 = 1 • N=4 – – – W 4, 0 = 1 W 4, 1 = 4 W 4, 2 = 6 W 4, 3 = 4 W 4, 4 = 1

2 Cells Probability Macrostates • N=1 – P 1, 0 = 0. 50 – P 1, 1 = 0. 50 • N=2 – P 2, 0 = 0. 25 – P 2, 1 = 0. 50 – P 2, 2 = 0. 25 • N=3 – – P 3, 0 = 0. 125 P 3, 1 = 0. 375 P 3, 2 = 0. 375 P 3, 3 = 0. 125 • N=4 – – – P 4, 0 = 0. 0625 P 4, 1 = 0. 2500 P 4, 2 = 0. 3750 P 4, 3 = 0. 2500 P 4, 4 = 0. 0625
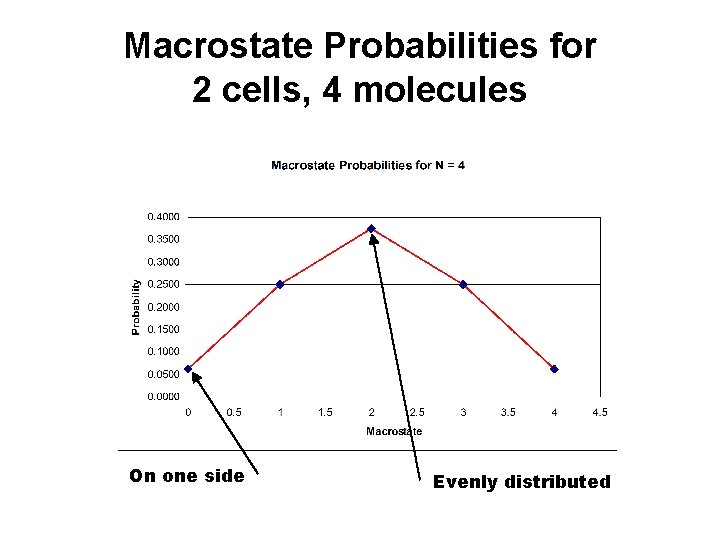
Macrostate Probabilities for 2 cells, 4 molecules On one side Evenly distributed
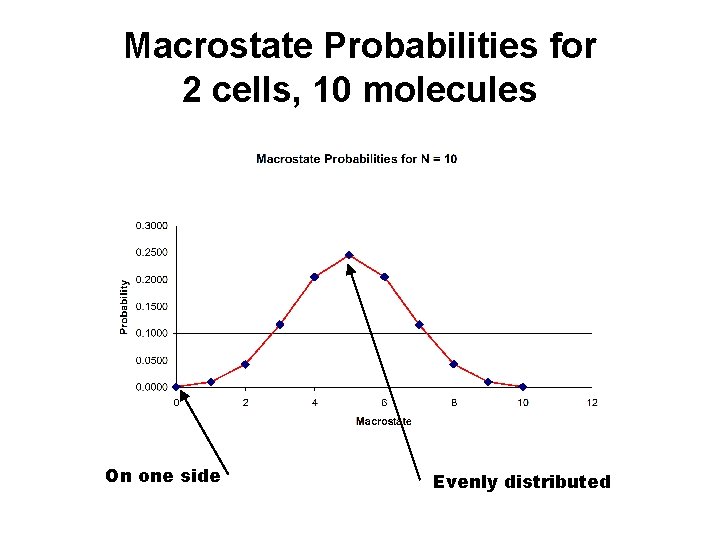
Macrostate Probabilities for 2 cells, 10 molecules On one side Evenly distributed
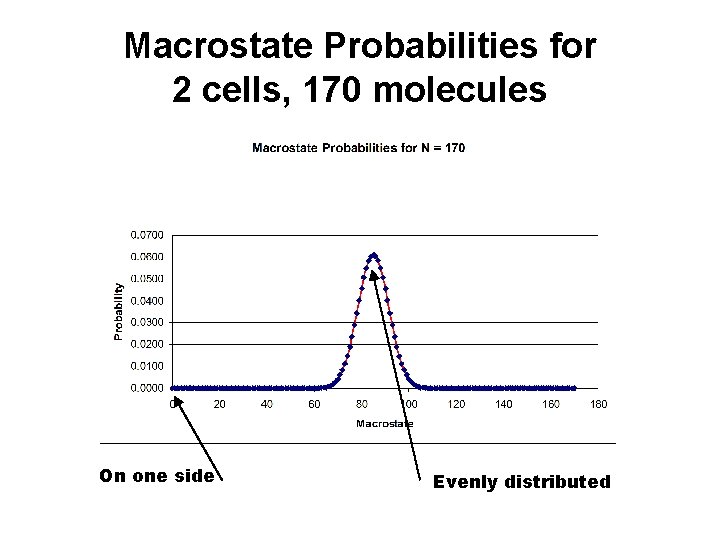
Macrostate Probabilities for 2 cells, 170 molecules On one side Evenly distributed
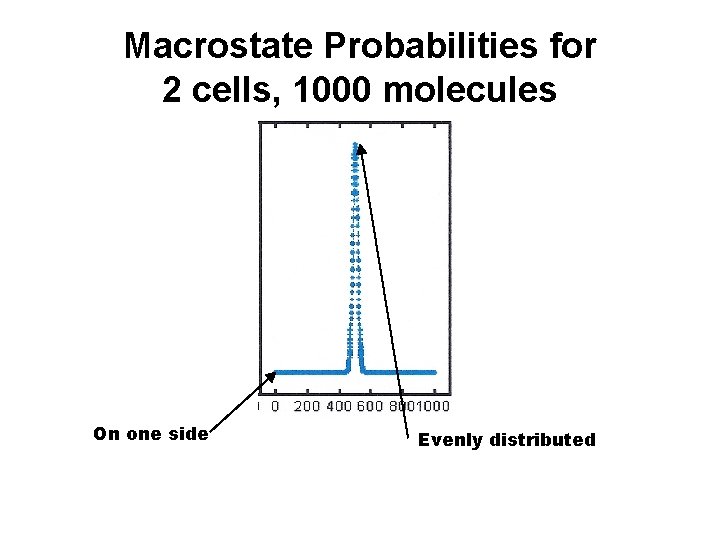
Macrostate Probabilities for 2 cells, 1000 molecules On one side Evenly distributed

2 Cells Ratio Uniform Distribution P(N/2) to Corner Probability P(1) Number Molecules N P(N/2) / P(0) Uniform / Corner 4 6 10 252 170 9. 1 x 1049 400 1. 0 x 10119 1000 2. 7 X 10299

Free Expansion of Gas

Microstates and Space • Divide available space into c small cells • Put N particles inside the space • Number of microstates is c. N
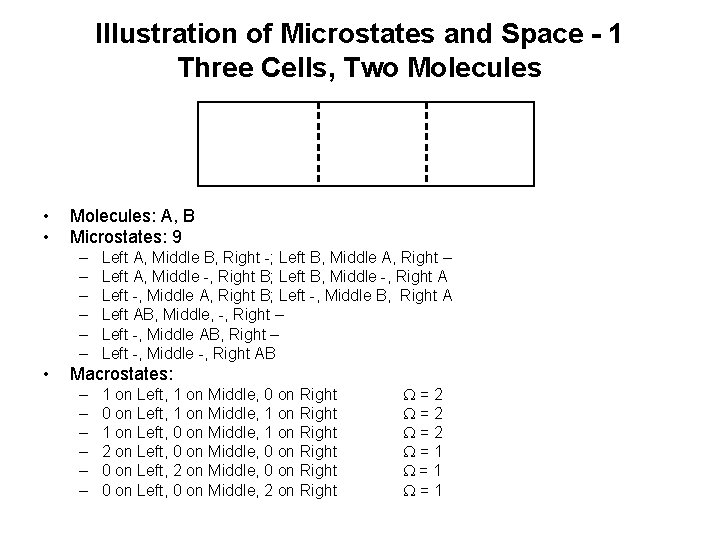
Illustration of Microstates and Space - 1 Three Cells, Two Molecules • • Molecules: A, B Microstates: 9 – – – • Left A, Middle B, Right -; Left B, Middle A, Right – Left A, Middle -, Right B; Left B, Middle -, Right A Left -, Middle A, Right B; Left -, Middle B, Right A Left AB, Middle, -, Right – Left -, Middle AB, Right – Left -, Middle -, Right AB Macrostates: – – – 1 on Left, 1 on Middle, 0 on Right 0 on Left, 1 on Middle, 1 on Right 1 on Left, 0 on Middle, 1 on Right 2 on Left, 0 on Middle, 0 on Right 0 on Left, 2 on Middle, 0 on Right 0 on Left, 0 on Middle, 2 on Right W=2 W=2 W=1 W=1

Illustration of Microstates and Space - 2 • Cells c=3 • Molecules N = 2 • Microstates c. N = 9

• Increasing the available space is equivalent to increasing the number of cells c • The volume is proportional to the number of cells c – W VN

Slow and Fast Processes • Slow Processes – reversible – very close to equilibrium – can run backwards • Fast Process – irreversible – system gets out of equilibrium – cannot run things backwards

Slow Isothermal Expansion - 1 • Slow isothermal expansion of ideal gas • Small volume change • No temperature change
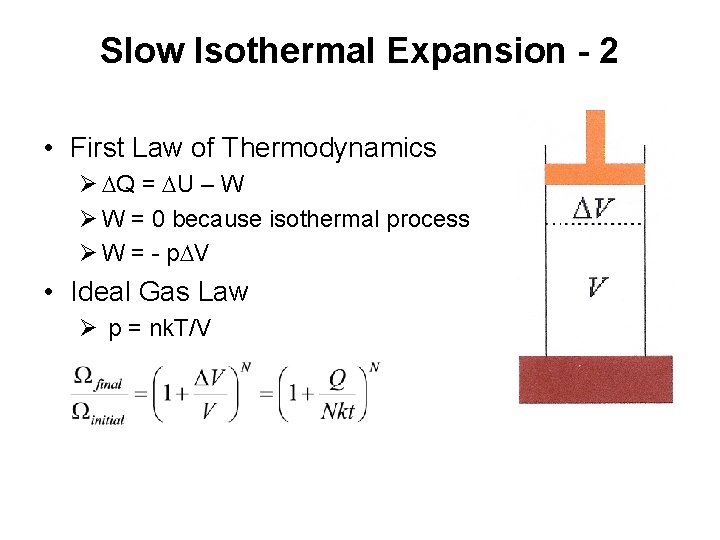
Slow Isothermal Expansion - 2 • First Law of Thermodynamics Ø DQ = DU – W Ø W = 0 because isothermal process Ø W = - p. DV • Ideal Gas Law Ø p = nk. T/V
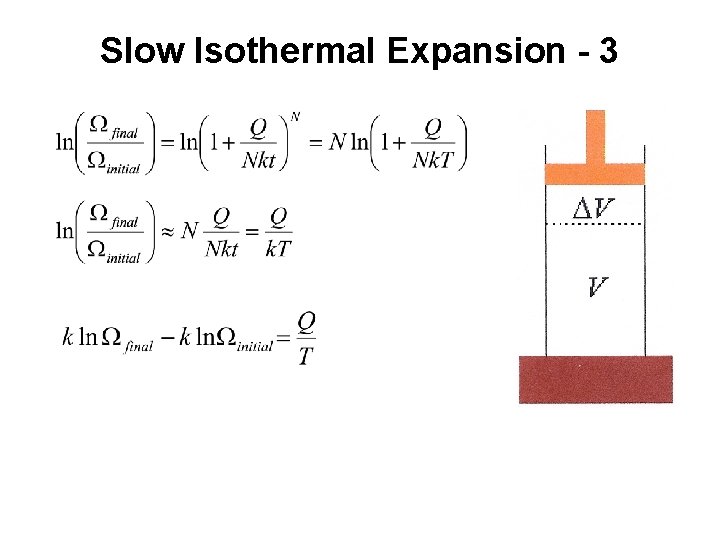
Slow Isothermal Expansion - 3
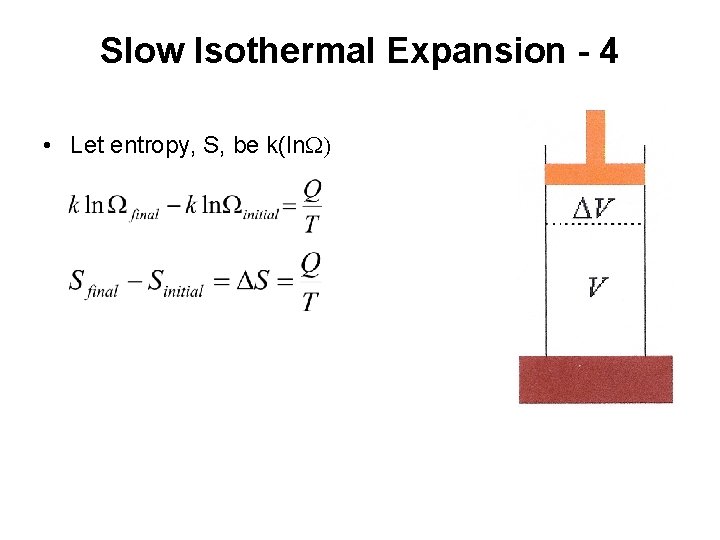
Slow Isothermal Expansion - 4 • Let entropy, S, be k(ln. W)
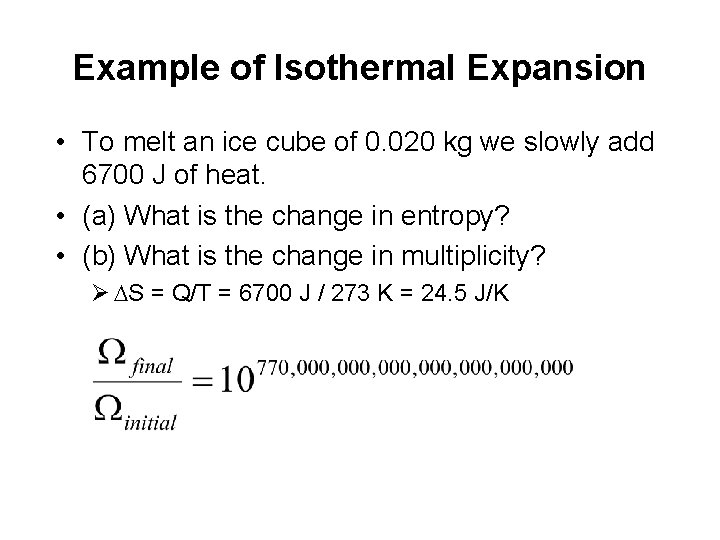
Example of Isothermal Expansion • To melt an ice cube of 0. 020 kg we slowly add 6700 J of heat. • (a) What is the change in entropy? • (b) What is the change in multiplicity? Ø DS = Q/T = 6700 J / 273 K = 24. 5 J/K
- Slides: 42