Physicochemical Properties at Bayer Health Care Wuppertal and

Physicochemical Properties at Bayer Health. Care (Wuppertal) and Their Use in Medicinal Chemistry Jörg Keldenich Nov. 2006 2. Phys. Chem Forum 1

Contents Ü Measured physico-chemical parameters Ü Introduction of our laboratory Ü Model systems for lipophilicity Ü Solubility Ü Use in medicinal chemistry Jörg Keldenich Nov. 2006 2. Phys. Chem Forum 2

Physicochemical Properties Measured Lipophilicity Membrane Affinity MA Plasma binding human serum albumin binding rat serum albumin binding HSA RSA p. Ka Solubility p. Ka screening in various buffers equilibrium in buffer equilibrium in galenic formulations Jörg Keldenich Nov. 2006 2. Phys. Chem Forum 3 SOL

Logistics for HT Physicochemistry BLJ input for sample identification Vial collecting rack Bar-coded vial for sample registration Jörg Keldenich Nov. 2006 2. Phys. Chem Forum 4
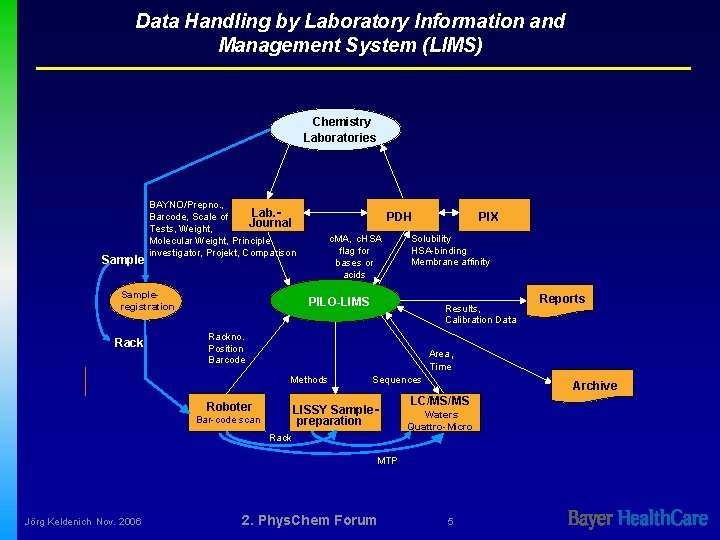
Data Handling by Laboratory Information and Management System (LIMS) Chemistry Laboratories Sample BAYNO/Prepno. , Lab. Barcode, Scale of Journal Tests, Weight, Molecular Weight, Principle investigator, Projekt, Comparison Sampleregistration Rack PDH c. MA, c. HSA flag for bases or acids PIX Solubility HSA-binding Membrane affinity PILO-LIMS Results, Calibration Data Rackno. Position Barcode Area, Time Methods Roboter Sequences LISSY Samplepreparation Bar-code scan Rack Archive LC/MS/MS Waters Quattro-Micro MTP Jörg Keldenich Nov. 2006 Reports 2. Phys. Chem Forum 5
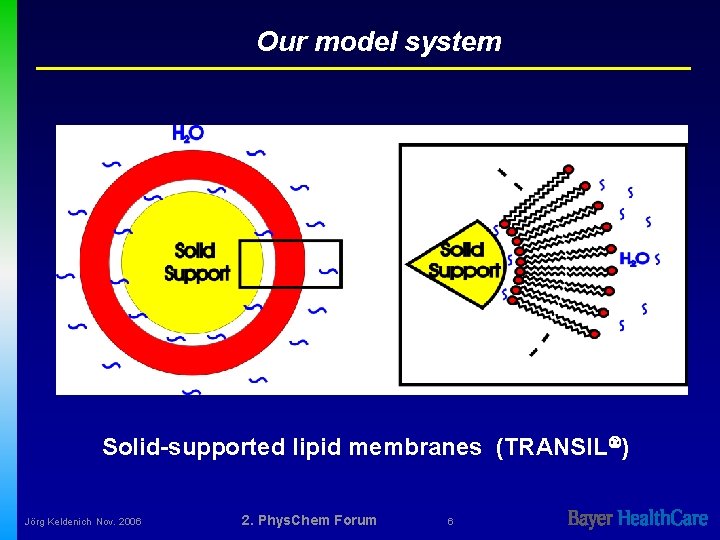
Our model system Solid-supported lipid membranes (TRANSIL ) Jörg Keldenich Nov. 2006 2. Phys. Chem Forum 6
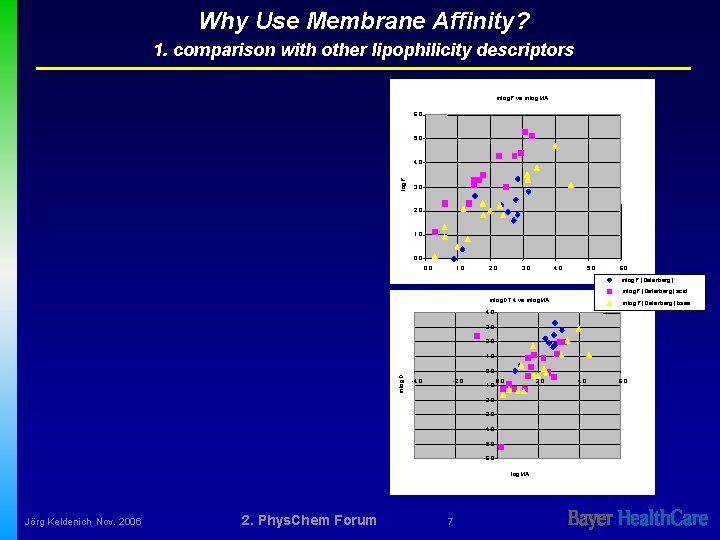
Why Use Membrane Affinity? 1. comparison with other lipophilicity descriptors mlog. P vs mlog. MA 6. 0 5. 0 log. P 4. 0 3. 0 2. 0 1. 0 0. 0 1. 0 2. 0 3. 0 4. 0 5. 0 6. 0 mlog. P (Österberg) acid mlog. D 7. 4 vs mlog. MA mlog. P (Österberg) base 4. 0 3. 0 2. 0 1. 0 mlog. D 0. 0 -4. 0 -2. 0 -1. 0 0. 0 2. 0 -3. 0 -4. 0 -5. 0 -6. 0 log. MA Jörg Keldenich Nov. 2006 2. Phys. Chem Forum 7 4. 0 6. 0
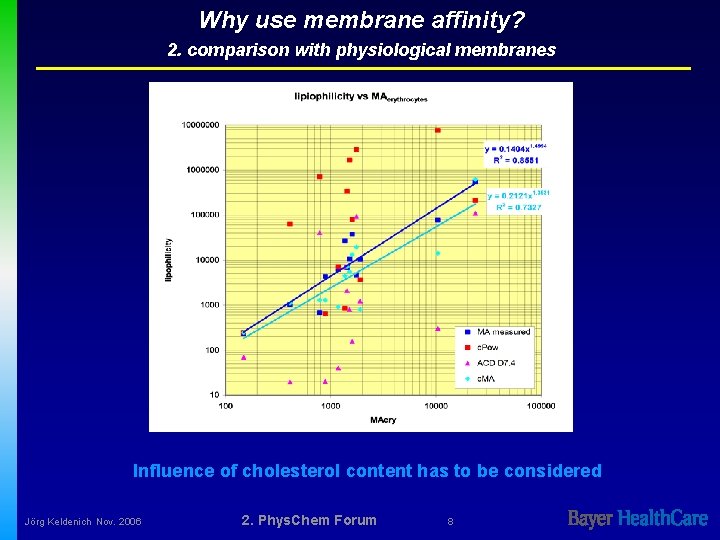
Why use membrane affinity? 2. comparison with physiological membranes Influence of cholesterol content has to be considered Jörg Keldenich Nov. 2006 2. Phys. Chem Forum 8
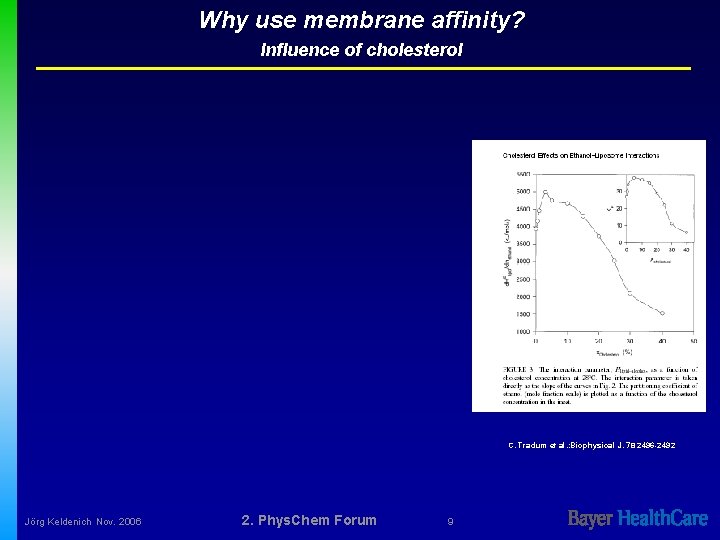
Why use membrane affinity? Influence of cholesterol C. Tradum et al. : Biophysical J. 78 2496 -2492 Jörg Keldenich Nov. 2006 2. Phys. Chem Forum 9

Why use membrane affinity? Influence of cholesterol Jörg Keldenich Nov. 2006 2. Phys. Chem Forum 10

Why use membrane affinity? Influence of cholesterol Ü Influence on passive permeation expected to be strong Ü Ü flexibility of plasma membrane is strongly influence by cholesterol, content usually about 80 mol% of phospholipids content Influence on distribution expected to be low Ü Jörg Keldenich Nov. 2006 80% of all membranes are intracellular with a cholesterol content about 4 mol% of phospholipids content 2. Phys. Chem Forum 11

Comparison of solubility methods Ü precipitation Ü from powder · all compounds · selected compounds · small amounts · large amounts (two samples) · fast analytics · specific analytics · compound dissolved in · sensitive to morphology organic solvent · sensitive to purity · oversaturated solutions possible Jörg Keldenich Nov. 2006 · sensitive to solvent impurities 2. Phys. Chem Forum 12

Comparison of solubility methods Ü precipitation Ü from powder · Dissolve compound in DMSO (2 mg/40µl) · Weight an appropriate amount of compound as solid · Add 10µl of this solution to 1000µl buffer (1% DMSO) · Shake for 24 h at room temperature · Add 1000µl buffer · Shake for 24 h at room temperature · Centrifuge to get supernatant · Establish LC/MS/MS method · Measure calibration standards and probe Jörg Keldenich Nov. 2006 2. Phys. Chem Forum 13
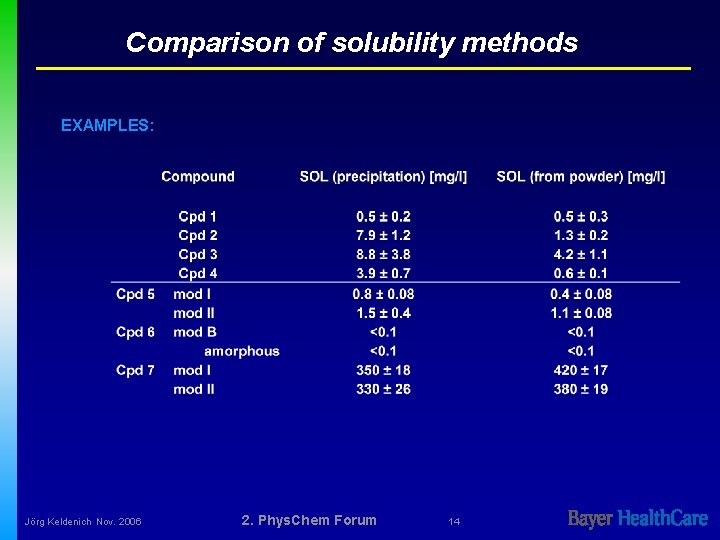
Comparison of solubility methods EXAMPLES: Jörg Keldenich Nov. 2006 2. Phys. Chem Forum 14
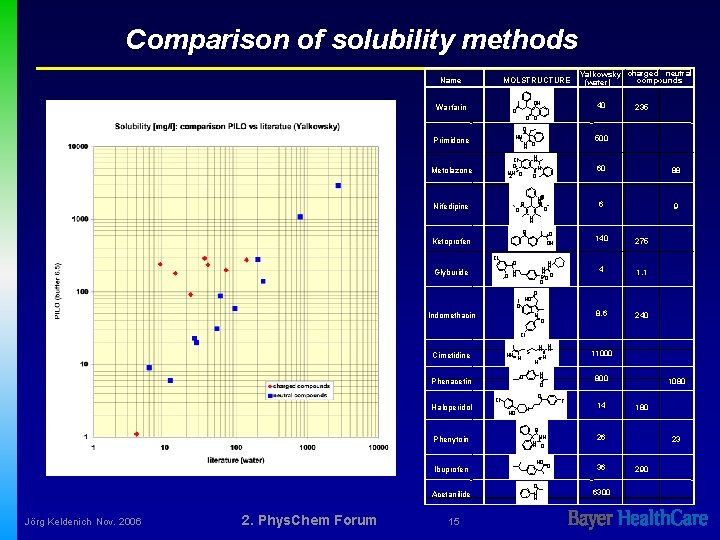
Comparison of solubility methods Name MOLSTRUCTURE OH Warfarin Yalkowsky charged neutral compounds (water) 40 O 235 O O O HN N H O Primidone H N Cl O H 2 NSO Metolazone N O N+O O O Nifedipine 500 60 88 6 9 N H O O OH Ketoprofen Cl Glyburide O N O H H N S OO O O HO Indomethacin 140 275 4 1. 1 8. 6 240 O N O Cl HN N S Cimetidine Phenytoin Ibuprofen Acetanilide Jörg Keldenich Nov. 2006 2. Phys. Chem Forum 15 800 O Cl HO 11000 H N O O Phenacetin Haloperidol H H N N F N O NH N H O HO O N H 14 1080 180 26 O 36 6300 23 290

Lessons learned from solubility comparisons Ü method differences not really critical Ü physical form very important differences between research and development result from: · morphology differences · impurities • solvent content Ü counter-ions and buffers are important when compound is charged in solution Jörg Keldenich Nov. 2006 2. Phys. Chem Forum 16

Case Histories: The Use of Physicochemical Properties Two different projects as examples: 1. Reducing lipophilicity and HSA binding to increase fraction unbound: erectile disfunction 2. Influence of solubility on in vivo efficacy: the HSV project Jörg Keldenich Nov. 2006 2. Phys. Chem Forum 17
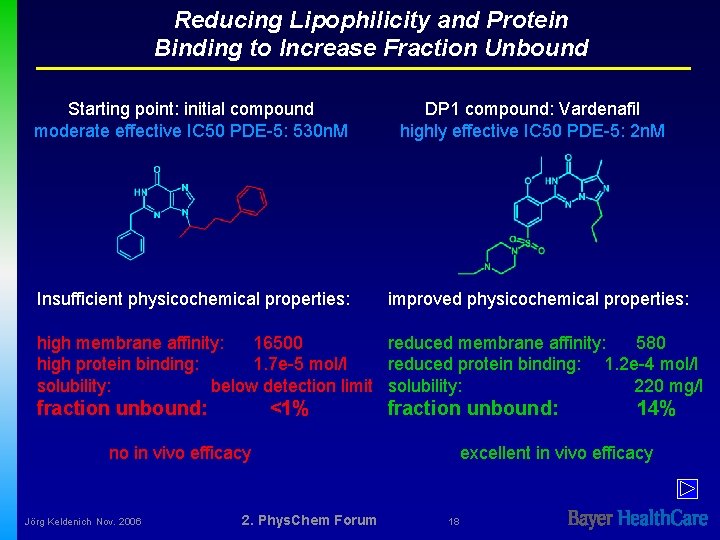
Reducing Lipophilicity and Protein Binding to Increase Fraction Unbound Starting point: initial compound moderate effective IC 50 PDE-5: 530 n. M Insufficient physicochemical properties: DP 1 compound: Vardenafil highly effective IC 50 PDE-5: 2 n. M improved physicochemical properties: high membrane affinity: 16500 reduced membrane affinity: 580 high protein binding: 1. 7 e-5 mol/l reduced protein binding: 1. 2 e-4 mol/l solubility: below detection limit solubility: 220 mg/l fraction unbound: <1% fraction unbound: no in vivo efficacy Jörg Keldenich Nov. 2006 2. Phys. Chem Forum 14% excellent in vivo efficacy 18
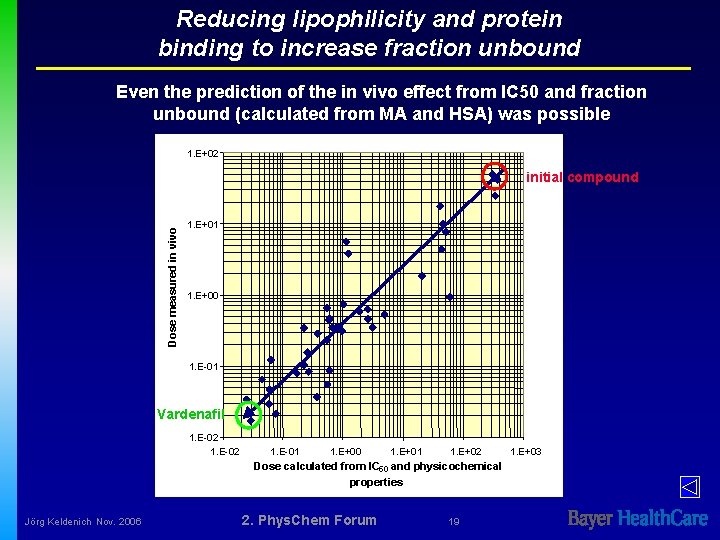
Reducing lipophilicity and protein binding to increase fraction unbound Even the prediction of the in vivo effect from IC 50 and fraction unbound (calculated from MA and HSA) was possible 1. E+02 Dose measured in vivo initial compound 1. E+01 1. E+00 1. E-01 Vardenafil 1. E-02 1. E-01 1. E+00 1. E+01 1. E+02 Dose calculated from IC 50 and physicochemical properties Jörg Keldenich Nov. 2006 2. Phys. Chem Forum 19 1. E+03
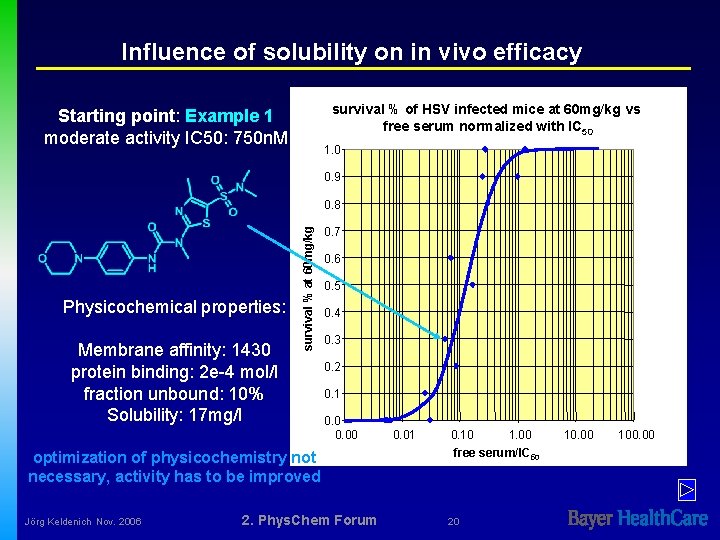
Influence of solubility on in vivo efficacy survival % of HSV infected mice at 60 mg/kg vs free serum normalized with IC 50 Starting point: Example 1 moderate activity IC 50: 750 n. M 1. 0 0. 9 Physicochemical properties: Membrane affinity: 1430 protein binding: 2 e-4 mol/l fraction unbound: 10% Solubility: 17 mg/l survival % at 60 mg/kg 0. 8 0. 7 0. 6 0. 5 0. 4 0. 3 0. 2 0. 1 0. 00 optimization of physicochemistry not necessary, activity has to be improved Jörg Keldenich Nov. 2006 2. Phys. Chem Forum 0. 01 0. 10 1. 00 free serum/IC 50 20 10. 00 100. 00
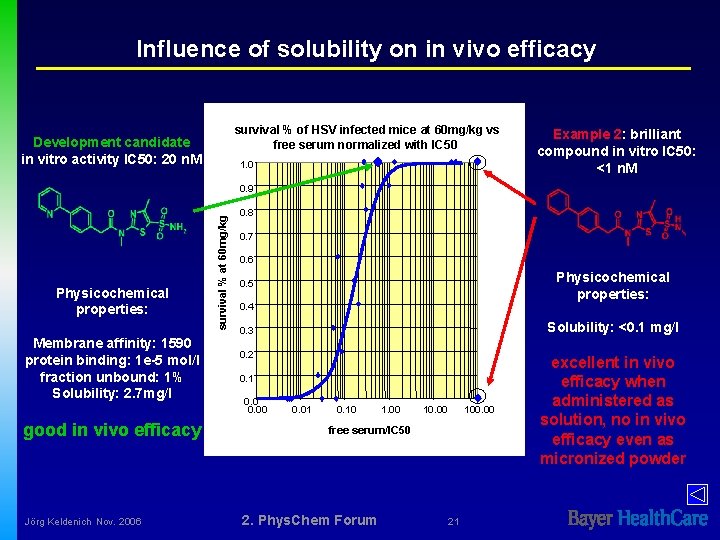
Influence of solubility on in vivo efficacy survival % of HSV infected mice at 60 mg/kg vs free serum normalized with IC 50 Development candidate in vitro activity IC 50: 20 n. M 1. 0 Example 2: brilliant compound in vitro IC 50: <1 n. M Physicochemical properties: Membrane affinity: 1590 protein binding: 1 e-5 mol/l fraction unbound: 1% Solubility: 2. 7 mg/l good in vivo efficacy Jörg Keldenich Nov. 2006 survival % at 60 mg/kg 0. 9 0. 8 0. 7 0. 6 Physicochemical properties: 0. 5 0. 4 Solubility: <0. 1 mg/l 0. 3 0. 2 0. 1 0. 00 0. 01 0. 10 1. 00 100. 00 free serum/IC 50 2. Phys. Chem Forum 21 excellent in vivo efficacy when administered as solution, no in vivo efficacy even as micronized powder

Conclusion Ü Impact of Physicochemistry Proven Ü Physicochemistry/ADME Implemented in Medicinal Chemistry Ü Properties Routinely Measured for Every Strategic Project Ü Use in Lead Optimization and Exploratory Research Established Jörg Keldenich Nov. 2006 2. Phys. Chem Forum 22
- Slides: 22