Physician Reimbursement Disclaimer Health economic and reimbursement information

Physician Reimbursement

Disclaimer • Health economic and reimbursement information provided by Boston Scientific Corporation is gathered from third party sources and is subject to change without notice as a result of complex and frequently changing laws, regulations, rules and policies. This information is presented for illustrative purposes only and does not constitute reimbursement or legal advice. Boston Scientific encourages providers to submit accurate and appropriate claims for services. It is always the provider’s responsibility to determine medical necessity, the proper site for delivery of any services and to submit appropriate codes, charges, and modifiers for services that are rendered. Boston Scientific recommends that you consult with your payers, reimbursement specialists and/or legal counsel regarding coding, coverage and reimbursement matters. Boston Scientific does not promote the use of its products outside their FDA approved label. It is always the provider's responsibility to understand comply with national coverage determinations (NCD), local coverage decisions (LCD) and any other coverage requirements established by relevant payers which can be updated frequently. • Payer policies will vary and should be verified prior to treatment for limitations on diagnosis, coding or site of service requirements. The coding options listed within this guide are commonly used codes and are not intended to be an all inclusive list. We recommend consulting your relevant manuals for appropriate coding options. • CPT copyright 2017 American Medical Association. All rights reserved. CPT is a registered trademark of the American Medical Association. Applicable FARS/DFARS Restrictions Apply to Government Use. Fee schedules, relative value units, conversion factors and/or related components are not assigned by the AMA, are not part of CPT, and the AMA is not recommending their use. The AMA does not directly or indirectly practice medicine or dispense medical services. The AMA assumes no liability for data contained or not contained herein. 2

Agenda 1. Medicare’s Incentive Payment System 2. Patient Visits 3. Pacemaker & Defibrillator Management 3
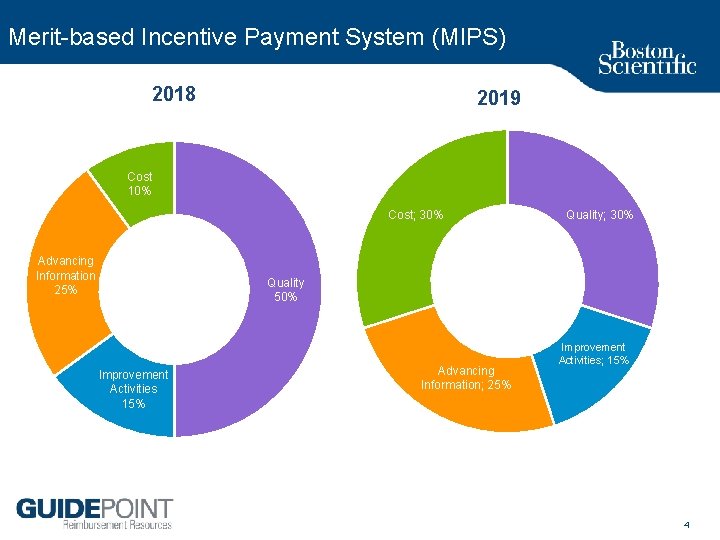
Merit based Incentive Payment System (MIPS) 2018 2019 Cost 10% Cost; 30% Advancing Information 25% Quality; 30% Quality 50% Improvement Activities 15% Advancing Information; 25% Improvement Activities; 15% 4

Incentive System Components • 271 quality measures – At least 6 must be reported – Example: 30 day all cause readmission rate for patients 65+ – https: //qpp. cms. gov/mips/quality measures • 93 activities: care coordination, beneficiary engagement, patient safety – Attest to 2 or 4 activities (15+ providers report 4) – Example: chronic & preventive care management – https: //qpp. cms. gov/mips/improvement activities • Formerly “Meaningful Use” of EMR – Security risk analysis, E prescribing, patient access, health information exchange, etc. – https: //qpp. cms. gov/mips/advancing care information • Total Capita Cost & Medicare Spending Per Beneficiary (MSPB) – Total Capita Cost includes all professional & facility fees • Heart failure = biggest expense to Medicare – MSPB includes 3 days prior to admission & 30 days after discharge • Post op infections & readmissions are costly 5

Penalty/Bonus Calculations • Budget neutral shift ($199 m) – 2020 5% bonus/penalty for 2018 performance – 2021 7% bonus/penalty for 2019 performance – 2022 9% bonus/penalty for 2020 performance • Additional $500 m/yr. incentive – Exceptional Performers: up to 3 x bonus – Top Performers: up to 10% additional bonus • 2018 Performance Year Example – Worst possible: • 5% penalty • $100 k becomes $95 k – Best possible • 15% bonus (5% x 3) • +10% top performer = 25% bonus • $100 k becomes $125 k – Variance = $30 k = 32% of $95 K Same Service Reimbursement Difference (Between Best & Worst Possible MIPS Scores) 60% 51% 50% 41% 32% 30% 20% 10% 0% 2020 2021 2022 6
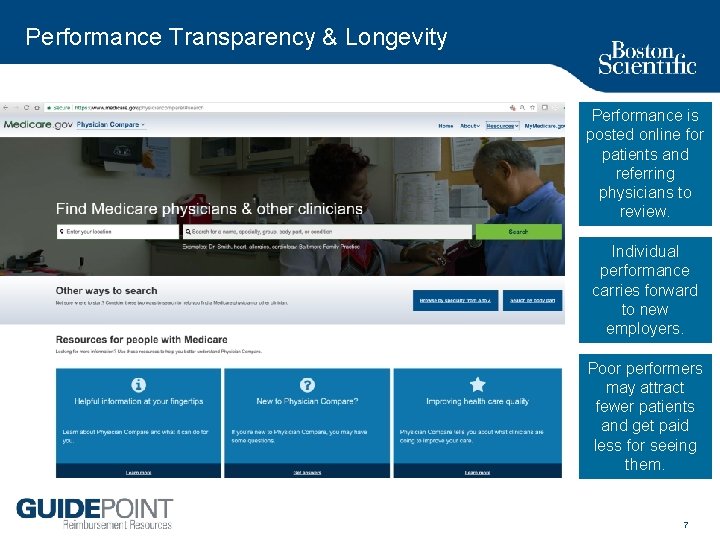
Performance Transparency & Longevity Performance is posted online for patients and referring physicians to review. Individual performance carries forward to new employers. Poor performers may attract fewer patients and get paid less for seeing them. 7
![Top 10 Expensive Conditions for Medicare Pneumonia 9% [CATEGORY NAME] [PERCENTAGE] Complication of device, Top 10 Expensive Conditions for Medicare Pneumonia 9% [CATEGORY NAME] [PERCENTAGE] Complication of device,](http://slidetodoc.com/presentation_image/3e5ed0863ed03f7c7dd329f2fabd6697/image-8.jpg)
Top 10 Expensive Conditions for Medicare Pneumonia 9% [CATEGORY NAME] [PERCENTAGE] Complication of device, implant or graft 10% Congestive heart failure 10% Acute myocardial infarction 10% Cardiology 41% Coronary atherosclerosis 7% Acute cerebrovascular disease 7% Osteoarthritis 13% Cardiac dysrhythmias 7% Septicemia 21% Source: Agency for Healthcare Research and Quality (AHRQ), Healthcare Cost and Utilization Project (HCUP), National Inpatient Sample (NIS), 2014 8
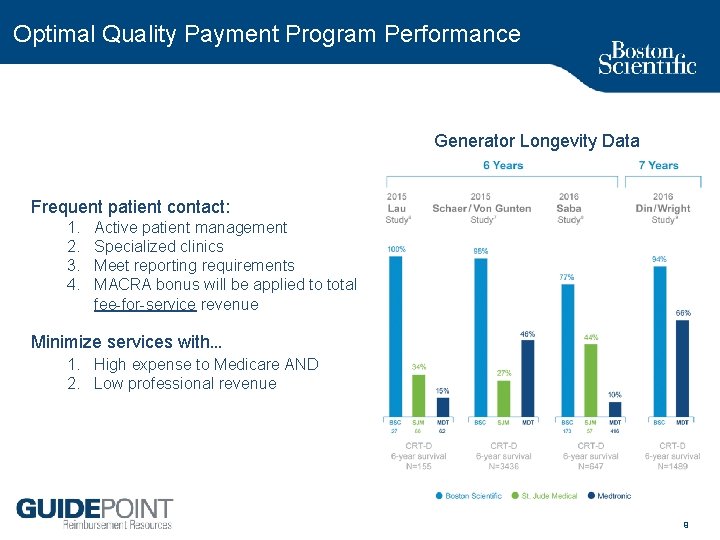
Optimal Quality Payment Program Performance Generator Longevity Data Frequent patient contact: 1. 2. 3. 4. Active patient management Specialized clinics Meet reporting requirements MACRA bonus will be applied to total fee for service revenue Minimize services with… 1. High expense to Medicare AND 2. Low professional revenue 9
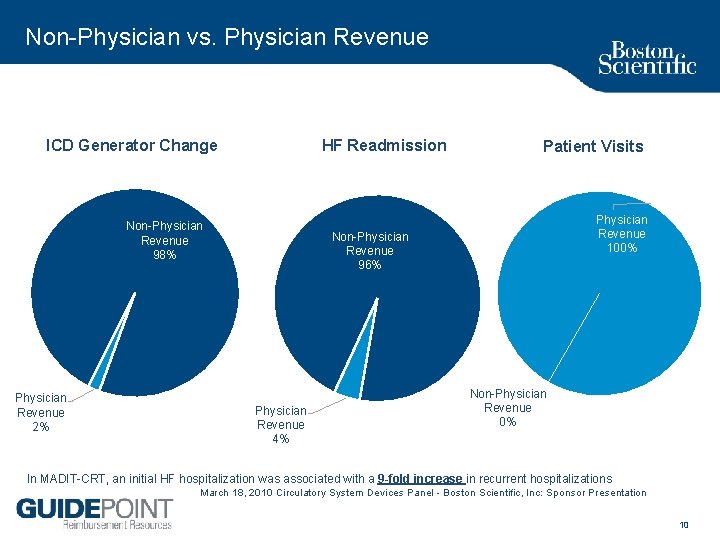
Non Physician vs. Physician Revenue ICD Generator Change HF Readmission Non Physician Revenue 98% Physician Revenue 2% Patient Visits Physician Revenue 100% Non Physician Revenue 96% Physician Revenue 4% Non Physician Revenue 0% In MADIT CRT, an initial HF hospitalization was associated with a 9 -fold increase in recurrent hospitalizations March 18, 2010 Circulatory System Devices Panel Boston Scientific, Inc: Sponsor Presentation 10

Patient Visits

Patient Visit Documentation Guidelines • The most commonly coded services for cardiology patients • The most frequently audited services by Medicare • Accuracy is important: under & over coding is bad History (all 3 required) History of Present Illness Review of Systems Medical, Family, Social Hx. • • Exam Complexity (2 of 3 required) History of Present Illness Review of Systems Medical, Family, Social Hx. New patients and admits require history, exam, & complexity Established patients & hospital follow up need just 2 of the 3 12

History & Exam Documentation Guidelines 13
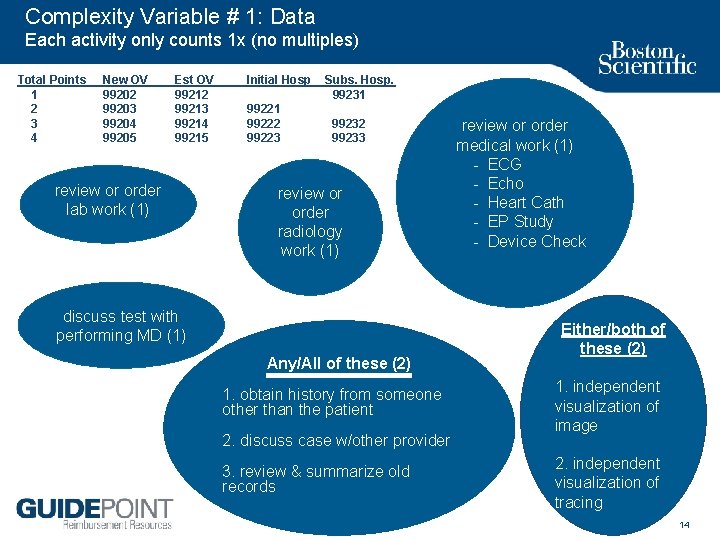
Complexity Variable # 1: Data Each activity only counts 1 x (no multiples) Total Points 1 2 3 4 New OV 99202 99203 99204 99205 Est OV 99212 99213 99214 99215 review or order lab work (1) Initial Hosp 99221 99222 99223 Subs. Hosp. 99231 99232 99233 review or order radiology work (1) discuss test with performing MD (1) Any/All of these (2) 1. obtain history from someone other than the patient 2. discuss case w/other provider 3. review & summarize old records review or order medical work (1) ECG Echo Heart Cath EP Study Device Check Either/both of these (2) 1. independent visualization of image 2. independent visualization of tracing 14

Complexity Variable # 2: Presenting Problems Total Points 1 2 3 4 New OV 99202 99203 99204 99205 Est OV 99212 99213 99214 99215 Initial Hosp 99221 99222 99223 Subs. Hosp. 99231 99232 99233 New vs. Established Problem = “to the examiner” not the patient Count x Points = Total • Stable, established problem (1) ___ x 1 = ___ • Worsening, established problem (2) ___ x 2 = ___ • New problem, no work up planned (3) ___ x 3 = ___ • New problem, work up planned (4) ___ x 4 = ___ TOTAL: _____ 15

Complexity Variable #3: Risk Level 1 2 3 4 New OV 99202 99203 99204 99205 Est OV 99212 99213 99214 99215 Initial Hosp Subs. Hosp. 99231 99222 99223 99232 99233 Credit is earned for the highest level of risk reached by any one variable. 4 Problems: Data: Chronic illness w/ severe exacerbation/progression/treatment side effects, acute MI, pulmonary embolus, TIA EP study, cardiovascular imaging with contrast + risk factors 3 Problems: 2 or more stable, chronic conditions, undiagnosed new problem, chronic condition w/ mild exacerbation Data: stress test, cardiovascular imaging with contrast w/ out identified risk factors Management: Prescription drug management, elective major surgery w/ out identified risk factors 2 Problems: 1 stable chronic illness Data: pulmonary function test, arterial puncture Management: over the counter drugs, physical therapy Management: DNR, elective major surgery + risk factors, emergency major surgery, drug therapy w/ intensive monitoring for toxicity 16

Time based Coding & Documentation must establish: 1) Face to Face time (office) or Unit/Floor time (hospital) 2) Majority of time was in Counseling or Coordination of Care New Pt. Office Visit 99201 – 10 min. 99202 – 20 min. 99203 – 30 min. 99204 – 45 min. 99205 – 60 min. Established Pt. Office Visit 99211 – 5 min. 99212 – 10 min. 99213 – 15 min. 99214 – 25 min. 99215 – 40 min. Outpatient Consultation 99241 – 15 min. 99242 – 30 min. 99243 – 40 min. 99244 – 60 min. 99245 – 80 min. Sample 99215: “I spent 40 minutes with the patient; the majority of this time was spent discussing heart failure treatment options. ” Observation Admission 99218 – 30 min. 99219 – 50 min. 99220 – 70 min. Subsequent Observation Care 99224 – 15 min. 99225 – 25 min. 99226 – 35 min. Initial Inpatient Care 99221 – 30 min. 99222 – 50 min. 99223 – 70 min. Subsequent Inpatient Care 99231 – 15 min. 99232 – 25 min. 99233 – 35 min. Inpatient Consultation 99251 – 20 min. 99252 – 40 min. 99253 – 55 min. 99254 – 80 min. 99255 – 110 min. 17

Device Management Coding & Billing

HRS/AHA Statements on Remote Monitoring • “HRS Expert Consensus Statement on Cardiovascular Implantable Electronic Devices. ” – Remote monitoring – a Class 1 A recommendation – “This consensus document reflects the wealth of recent clinical data generated by large randomized prospective trials from around the world that included patients with pacemakers, ICDs, and CRT Ds from various manufacturers. These consistently show meaningful patient benefits from the early detection capabilities of automatic RM… These data form the basis of our recommendations that RM represents the new standard of care for patients with CIEDs” • AHA “Abstract 13944: Early Initiation of Remote Monitoring in CIED Patients is Associated with Reduced Mortality” – “Our data show that survival is higher in patients who have a shorter time between device implant and remote monitoring initiation, across all CIED device types. These data suggest RM should be initiated as soon as possible following device implantation. ” – early initiation of remote monitoring (within 91 days of device implant) reduces mortality by over 16%. . . ” 19

CPT Codes for PM & ICD Remote Monitoring 93294 Interrogation device evaluation(s) (remote), up to 90 days; single, dual, or multiple lead pacemaker system with interim physician analysis, review(s) and report(s) ($31) 93295 Interrogation device evaluation(s) (remote), up to 90 days; single, dual, or multiple lead implantable cardioverter defibrillator system with interim physician analysis, review(s) and report(s) ($56) 93296 Interrogation device evaluation(s) (remote), up to 90 days; single, dual, or multiple lead pacemaker system or implantable cardioverter defibrillator system, remote data acquisition(s), receipt of transmissions and technician review, technical support and distribution of results ($27) 2018 Medicare (national payment) 20

Remote Monitoring Billing 93294 Interrogation device evaluation(s) (remote), up to 90 days; single, dual, or multiple lead pacemaker system with interim physician analysis, review(s) and report(s) – Remote PM/ICD monitoring codes each include: • Up to 90 days of monitoring • One or more evaluations (transmission, review, report) – CPT Instructions • “Do not report 93293, 93294, 93295, 93296 if the monitoring period is less than 30 days” – Typically applies only to initiation & termination of RM – Date of Service • “While we do not have a national policy on this, our sense is that the DOS should be the date of completion. ” – CMS Director of External Affairs 21

Remote Monitoring Billing Cycle � � � Date of Service � � � 22
Remote Website for Charge Capture ✓✓ Date of Service 23

Flaws With Quarterly Approach � � Date of Service 1) Forfeits up to 30 days of RM for 1/3 of newly enrolled patients. 2) Cash flow will be regularly irregular (4 boluses rather than steady stream) 3) If no transmission is received by 90 th day, loose revenue for missed quarterly cycle 24

Creating Three Remote Monitoring Groups • Group 1: January, February, March – Bill 3/31 • Group 2: February, March, April – Bill 4/30 • Group 3: March, April, May – Bill 5/29 • Benefits: – Cash flow is even every month – New patients get assigned to optimal group • Eliminates loss of up to 30 days RM for 1/3 of new patients – If transmission is missed, bump patient to next monthly cycle • Cut loss from 3 months to 1 month for these 25

Legacy Device Based Heart Failure Monitoring • Thoracic Impedance – Pulmonary edema is a common sign of worsening HF – TI measures impedance between RV electrode and the pulse generator – TI may be associated with pulmonary fluid level – Worsening heart failure may cause a patient’s fluid level to increase • Limitations – Thoracic impedance shifts naturally as leads maturate – Only 21 – 22% sensitivity 1, 2 Only one of these patients had a HF event after TI dropped. 1. E. Kevin Heist, et al. , Analysis of Different Device Based Intrathoracic Impedance Vectors for Detection of Heart Failure Events (from the Detect Fluid Early from Intrathoracic Impedance Monitoring Study), The American Journal of Cardiology, Volume 114, Issue 8, 2014, Pages 1249 1256, ISSN 0002 9149, http: //dx. doi. org/10. 1016/j. amjcard. 2014. 07. 048. (http: //www. sciencedirect. com/science/article/pii/S 0002914914015343) 2. Viviane M. Conraads, Luigi Tavazzi, Massimo Santini, Fabrizio Oliva, Bart Gerritse, Cheuk Man Yu, Martin R. Cowie; Sensitivity and positive predictive value of implantable intrathoracic impedance monitoring as a predictor of heart failure hospitalizations: the SENSE HF trial. Eur Heart J 2011; 32 (18): 2266 2273. doi: 10. 1093/eurheartj/ehr 050 26

Heart. Logic Heart Failure Diagnostic TM ™ Heart. Logic shifts heart failure patient management from reactive treatment to proactive care, and was validated in the Multi. SENSE Study to have: • High sensitivity of 70% for detecting heart failure events • Weeks of advance notice of a potential heart failure event • Low burden of less than 2 alerts per patient per year Heart Sounds Impedance S 1 & S 3 Thoracic Respiration Activity Rate & Volume Time Spent Active Heart Rate Available on LATITUDE™ NXT for patients with Resonate™ family of ICDs & CRT Ds Night multiple sensor measurements combined into a single, simple index with alert Boehmer, J et al. , JACC HF, 2017; 5(3), 2 1 6 – 2 5 27
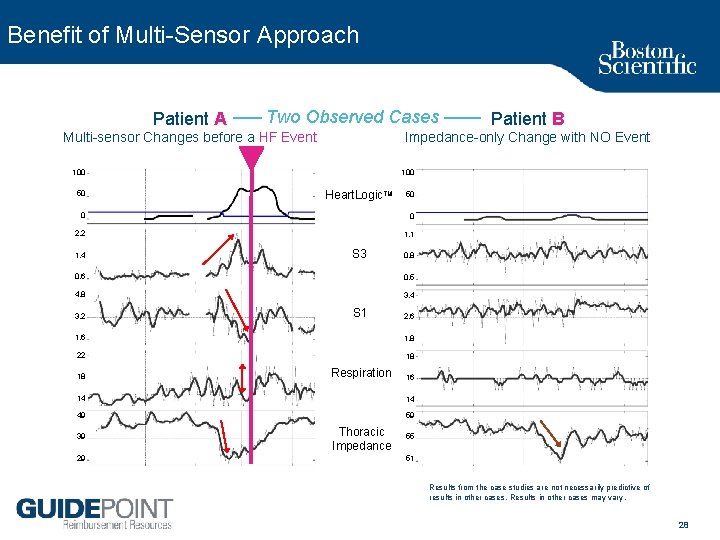
Benefit of Multi Sensor Approach Patient A Two Observed Cases Multi sensor Changes before a HF Event Impedance only Change with NO Event 100 50 100 Heart. Logic. TM 50 0 0 2. 2 1. 1 1. 4 S 3 0. 8 0. 6 0. 5 4. 8 3. 4 3. 2 S 1 2. 6 1. 8 22 18 18 Respiration 16 14 14 49 59 39 29 Patient B Thoracic Impedance 55 51 Results from the case studies are not necessarily predictive of results in other cases. Results in other cases may vary. 28
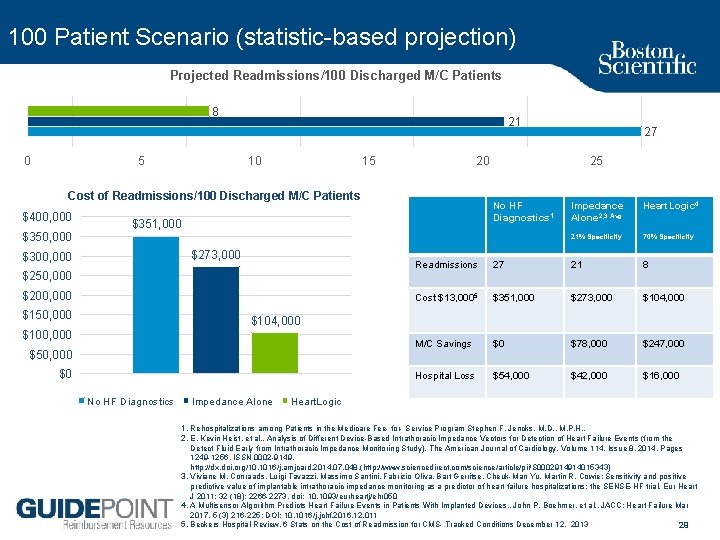
100 Patient Scenario (statistic based projection) Projected Readmissions/100 Discharged M/C Patients 8 0 5 21 10 15 20 Cost of Readmissions/100 Discharged M/C Patients $400, 000 $350, 000 $273, 000 $250, 000 $200, 000 $150, 000 25 No HF Diagnostics 1 $351, 000 $300, 000 27 Impedance Alone 2, 3 Avg. Heart. Logic 4 21% Specificity 70% Specificity Readmissions 27 21 8 Cost $13, 0005 $351, 000 $273, 000 $104, 000 M/C Savings $0 $78, 000 $247, 000 Hospital Loss $54, 000 $42, 000 $16, 000 $104, 000 $100, 000 $50, 000 $0 No HF Diagnostics Impedance Alone Heart. Logic 1. Rehospitalizations among Patients in the Medicare Fee for Service Program Stephen F. Jencks, M. D. , M. P. H. , 2. E. Kevin Heist, et al. , Analysis of Different Device Based Intrathoracic Impedance Vectors for Detection of Heart Failure Events (from the Detect Fluid Early from Intrathoracic Impedance Monitoring Study), The American Journal of Cardiology, Volume 114, Issue 8, 2014, Pages 1249 1256, ISSN 0002 9149, http: //dx. doi. org/10. 1016/j. amjcard. 2014. 07. 048. (http: //www. sciencedirect. com/science/article/pii/S 0002914914015343) 3. Viviane M. Conraads, Luigi Tavazzi, Massimo Santini, Fabrizio Oliva, Bart Gerritse, Cheuk Man Yu, Martin R. Cowie; Sensitivity and positive predictive value of implantable intrathoracic impedance monitoring as a predictor of heart failure hospitalizations: the SENSE HF trial. Eur Heart J 2011; 32 (18): 2266 2273. doi: 10. 1093/eurheartj/ehr 050 4. A Multisensor Algorithm Predicts Heart Failure Events in Patients With Implanted Devices. , John P. Boehmer, et al. , JACC: Heart Failure Mar 2017, 5 (3) 216 225; DOI: 10. 1016/j. jchf. 2016. 12. 011 5. Beckers Hospital Review, 6 Stats on the Cost of Readmission for CMS Tracked Conditions December 12, 2013 29

CPT Codes for Heart Failure Remote Monitoring 93297 Interrogation device evaluation(s), (remote) up to 30 days; implantable cardiovascular monitor system, including analysis of 1 or more recorded physiologic cardiovascular data elements from all internal and external sensors, physician analysis, review(s) and report(s) ($27) 93299 Interrogation device evaluation(s), (remote) up to 30 days; implantable cardiovascular monitor system or implantable loop recorder system, remote data acquisition(s), receipt of transmissions and technician review, technical support and distribution of results (contractor priced) � 2018 Medicare (national payment) Date of Service 30

2018 Contractor Pricing for 93299 • • Palmetto GBA Wisconsin Physician Services National Government Services CGS Administrators Noridian Healthcare Solutions Novitas Cahaba GBA First Coast Service Options • Average Payment: $23 $29 $118 $136 $26 $275 $29 $13 (2017) $38 $53 $29 $34 $38 $65/month 31

JACC 10/2/12 “Minimum Frequency” Guidelines • In Person Device Checks: – Within 72 hours of device implantation – 2– 12 weeks after device implantation – Annually until battery depletion • Additional In Person or Remote Checks: – Pacemakers: every 3 – 12 months – Defibrillators: every 3 – 6 months – Signs of battery depletion (PM & ICD): 1 – 3 months More frequent checks are expected 32

In Clinic PM/ICD Device Checks Iterative adjustments to assess programming? No: (Interrogation Evaluation) Pacemaker: 93288 Defibrillator: 93289 HF Monitor: 93290 Sub Q ICD: 93261 Yes: (Programming Evaluation) Pacemaker: Sgl (93279), Dual (93280), Multi (93281) Defibrillator: Sgl (93282), Dual (93283), Multi (93284) Sub Q ICD: 93260 “The final program parameters may or may not change after evaluation” CPT Instruction Medicare requires direct supervision of device checks done by physician practice employees. Direct supervision means the physician is immediately available in the office suite to furnish assistance but is not required to be in the room where the device check is being performed. https: //www. cms. gov/Regulations and Guidance/Transmittals/downloads/R 169 BP. pdf 33

In Clinic Interrogations During Remote Monitoring Heart Failure Data 93297 – Remote HF Professional 93299 – Remote HF Technical Pacemaker/Defibrillator Data 93295 – Remote PM Professional 93296 – Remote ICD Technical AAPC Coder/Correct Coding Initiative Edits 34

Device Clinic & Same Day Patient Visit • Visits are separately billable in many cases: – The visit must be medically necessary – Documented to support the level of service billed – In excess of the basic device check interaction • Billing pointers: – Modifier 25 (significant & separately identifiable service) is required on the visit code by some payers. – The diagnosis code(s) for the visit need to reflect the medical necessity for the visit (arrhythmia, CHF, etc. ) – The diagnosis codes for the check should reflect the indication for the device and the programming evaluation codes: • Pacemaker: Z 45. 010 (generator) AND Z 45. 018 (electrodes) • Defibrillator: Z 45. 02 (system evaluation) 35

Device Clinic Staffing • CMS Transmittal 135 – “The supervision requirement for physician billing is not met when the test is administered by supplier personnel” • American College of Cardiology – “If the physician doesn’t own the equipment or pay the personnel, he or she should bill only the professional component. ” • Heart Rhythm Society – “individual performing the evaluation must be employed by the MD in order to bill globally for the professional and technical services” – “Medicare guidelines prohibit a physician from reporting the technical component of services performed by IEAP who is not employed by the physician. ” • Modifier 26 (Professional Component) – 93284 26 (Bi V ICD programming evaluation – professional component) – 26 modifier reduces compensation from $92 to $64 ($28) 36

Annualized Revenue Defibrillator + Heart Failure • In person services: – 93283 X 2 ($80 x 2) – 99214 X 2 ($109 x 2) • Remote device monitoring: – 93295 X 4 ($56 x 4) – 93296 X 4 ($27 x 4) Pacemaker Without Heart Failure • In person: $160 $218 – 93280 X 1 – 99214 X 1 $39 $109 • Remote device monitoring: $224 $108 • Remote heart failure monitoring: – 93297 X 12 ($27 x 12) $324 – 93299 X 12 ($65 x 12) $780 – 93294 X 4 ($31 x 4) – 93296 X 4 ($27 x 4) $125 $108 Annual Revenue: $382 Annual Revenue: $1, 834 2018 CMS National fees used for all but 93299 ($65 used for it) 37

Here’s what we covered… 1. Medicare’s Incentive Payment System 2. Patient Visits 3. Device Management 38

Presenter Information • Jim Collins, CPC, CCC – President, Cardiology. Coder. Com, Inc. – Cardiology coding guru for over 20 years – Wrote the study guide and certification exam for the American Academy of Professional Coders (AAPC) called “Certified Cardiology Coder” – Co developed the certification exam for the Board of Advanced Medical Coding called "Advanced Coding Specialist in Cardiology" – Consulting Editor of "Cardiology Coding Alert” for over a decade. – Several articles published in ”EP Lab Digest” & ”Cath Lab Digest” • Please contact Jim if you have follow up questions or additional needs. @Cardiology. Coder 518. 320. 4376 Jim@Cardiology. Coder. Com 39
Guide. Point Is Simplifying Reimbursement We’re dedicated to providing physicians and allied health professionals with world class programs and services to help advance the standard of patient care. Reimbursement Customer Support Line Get your CRM and EP reimbursement questions answered. • Call 1. 800. CARDIAC (227. 3422) and ask for the Reimbursement Customer Support Line. Field Reimbursement Team Specialized on site support for documentation and coding education related to IC, PI, and CRM EP procedures. Webcast Programs Hear from nationally acclaimed experts addressing basic and advanced CRM and EP reimbursement topics. CRM Billing and Coding Guide Quickly find coding and billing information, including scenarios specific to CRM and EP procedures. We are proud to continue this spirit of partnership with Guide. Point Website Keep current with the latest CRM and EP reimbursement news and find other reimbursement education resources. • Make the website your first stop for all your Boston Scientific CRM and EP reimbursement needs, access http: //www. bostonscientific. com/crm/reimbursement 40

Heart. Logic™ Training Resource Heart. Logic Heart Failure Diagnostic Learning Center TM www. Heart. Logictraining. com Learn about Heart. Logic. TM, which was validated in the Multi. SENSE Study to detect the early warning signs of worsening heart failure by combining data from 5 sensors into a single composite index. Learnings include: • A 1 hour CEU interactive training program covering • Key takeaways from the Multi. SENSE study TM • Heart. Logic overview TM • Configuring Heart. Logic within LATITUDE™ NXT TM • Managing Heart. Logic Alerts • Videos • Links to available resources • FAQs • Submit your own case study 41

Brief Summary • CRT-D Systems –RESONATE™HF, RESONATE™X 4, VIGILANT™X 4, MOMENTUM™ X 4 • INDICATIONS AND USAGE • These Boston Scientific Cardiac Resynchronization Therapy Defibrillators (CRT-Ds) are indicated for patients with heart failure who receive stable optimal pharmacologic therapy (OPT) for heart failure and who meet any one of the following classifications: Moderate to severe heart failure (NYHA Class III-IV) with EF ≤ 35% and QRS duration ≥ 120 ms; left bundle branch block (LBBB) with QRS duration ≥ 130 ms, EF ≤ 30%, and mild (NYHA Class II) ischemic or nonischemic heart failure or asymptomatic (NYHA Class I) ischemic heart failure • CONTRAINDICATIONS • There are no contraindications for this device. • WARNINGS • Read this manual thoroughly before implantation to avoid damage to the pulse generator and/or lead. For single patient use only. Do not reuse, reprocess, or resterilize. Always have external defibrillation equipment available during implant and electrophysiologic testing. Ensure that an external defibrillator and medical personnel skilled in CPR are present during post-implant device testing should the patient require external rescue. Do not use defibrillation patch leads with the pulse generator system. Do not use this pulse generator with another pulse generator. Program the pulse generator Tachy Mode(s) to Off during implant, explant, or postmortem procedures. Do not kink, twist, or braid the lead with other leads. For leads that require the use of a Connector Tool, use caution handling the lead terminal when the Connector Tool is not present on the lead. Do not directly contact the lead terminal with any surgical instruments or electrical connections such as PSA (alligator) clips, ECG connections, forceps, hemostats, and clamps. Do not contact any other portion of the DF 4–LLHH or DF 4–LLHO lead terminal, other than the terminal pin, even when the lead cap is in place. Do not contact any other portion of the IS 4–LLLL lead terminal, other than the terminal pin, even when the lead cap is in place. When implant a system that uses both a DF 4– LLHH or DF 4–LLHO and IS 4–LLLL lead, ensure that the leads are inserted and secured in the appropriate ports. Do not use atrial tracking modes in patients with chronic refractory atrial tachyarrhythmias. Do not use atrial-only modes in patients with heart failure. Left ventricular lead dislodgement to a position near the atria can result in atrial oversensing and left ventricular pacing inhibition. Physicians should use medical discretion when implanting this device in patients who present with slow VT. Advise patients to seek medical guidance before entering environments that could adversely affect the operation of the active implantable medical device, including areas protected by a warning notice that prevents entry by patients who have a pulse generator. RESONATE HF, RESONATE, and VIGILANT devices with an IS-1/DF 4/IS 4 lead connection are considered MR Conditional. For these devices, unless all of the MRI Conditions of Use are met, MRI scanning of the patient does not meet MR Conditional requirements for the implanted system, and significant harm to or death of the patient and/or damage to the implanted system may result. For potential adverse events applicable when the Conditions of Use are met or not met, refer to the MRI Technical Guide. . Do not subject a patient with an implanted pulse generator and/or lead to diathermy. If desired, ensure that Patient Triggered Monitor (PTM) is enabled prior to sending the patient home by confirming the magnet response is programmed to Store EGM. Once the PTM feature has been triggered and the magnet response set to Inhibit therapy the patient should not reapply the magnet. • PRECAUTIONS • For specific information on precautions, refer to the following sections of the product labeling: clinical considerations, sterilization and storage, implantation, device programming, environmental and medical therapy hazards, hospital and medical environments, home and occupational environments, follow-up testing, explant and disposal, supplemental precautionary information. Advise patients to avoid sources of EMI because EMI may cause the pulse generator to deliver inappropriate therapy or inhibit appropriate therapy. • POTENTIAL ADVERSE EVENTS • Potential adverse events include, but are not limited to, the following: allergic/physical/physiologic reaction, death, erosion/migration, fibrillation or other arrhythmias, lead or accessory breakage (fracture/insulation/lead tip), hematoma/seroma, inappropriate or inability to provide therapy (shocks /pacing/sensing), infection, procedure related, and component failure. Patients may develop psychological intolerance to a pulse generator system and may experience fear of shocking, fear of device failure, or imagined shocking. In rare cases severe complications or device failures can occur. • Refer to the product labeling for specific indications, contraindications, warnings/precautions and adverse events. Rx only. (Rev B) 42

Brief Summary • ICD Systems – RESONATE™ HF, RESONATE™ EL, PERCIVA™ HF, PERCIVA™, VIGILANT™ EL, MOMENTUM™ EL • INDICATIONS AND USAGE • Boston Scientific implantable cardioverter defibrillators (ICDs) are intended to provide ventricular antitachycardia pacing (ATP) and ventricular defibrillation for automated treatment of life-threatening ventricular arrhythmias. • CONTRAINDICATIONS • Use of these Boston Scientific pulse generators are contraindicated for the following: patients whose ventricular tachyarrhythmias may have reversible cause, such as: digitalis intoxication, electrolyte imbalance, hypoxia, sepsis; or patients whose ventricular tachyarrhythmias have a transient cause, such as: acute myocardial infarction (MI), electrocution, drowning; or patients who have a unipolar pacemaker. • WARNINGS • Read this manual thoroughly before implantation to avoid damage to the pulse generator and/or lead. For single patient use only. Do not reuse, reprocess, or resterilize. Always have external defibrillation equipment available during implant and electrophysiologic testing. Ensure that an external defibrillator and medical personnel skilled in CPR are present during post-implant device testing should the patient require external rescue. Do not use this pulse generator with another pulse generator. Program the pulse generator Tachy Mode(s) to Off during implant, explant, or postmortem procedures to avoid inadvertent high voltage shocks. Do not kink, twist, or braid the lead with other leads as doing so could cause lead insulation abrasion damage or conductor damage. For leads that require the use of a Connector Tool, use caution handling the lead terminal when the Connector Tool is not present on the lead. Do not directly contact the lead terminal with any surgical instruments or electrical connections such as PSA (alligator) clips, ECG connections, forceps, hemostats, and clamps. Do not contact any other portion of the DF 4–LLHH or DF 4– LLHO lead terminal, other than the terminal pin, even when the lead cap is in place. Do not use atrial tracking modes in patients with chronic refractory atrial tachyarrhythmias. Tracking of atrial arrhythmias could result in ventricular tachyarrhythmias. Advise patients to seek medical guidance before entering environments that could adversely affect the operation of the active implantable medical device, including areas protected by a warning notice that prevents entry by patients who have a pulse generator. RESONATE HF, RESONATE, PERCIVA HF, PERCIVA, and VIGILANT devices with a DF 4 right ventricular lead connection are considered MR Conditional. For these devices, unless all of the MRI Conditions of Use are met, MRI scanning of the patient does not meet MR Conditional requirements for the implanted system, and significant harm to or death of the patient and/or damage to the implanted system may result. For potential adverse events applicable when the Conditions of Use are met or not met, refer to the MRI Technical Guide. Do not subject a patient with an implanted pulse generator and/or lead to diathermy. If desired, ensure that Patient Triggered Monitor is enabled prior to sending the patient home. Once the Patient Triggered Monitor feature has been triggered by the magnet and an EGM has been stored, or after 60 days have elapsed from the day that Store EGM was enabled, the patient should not apply the magnet. • PRECAUTIONS • For specific information on precautions, refer to the following sections of the product labeling: clinical considerations, sterilization and storage, implantation, device programming, environmental and medical therapy hazards, hospital and medical environments, home and occupational environments, follow-up testing, explant and disposal, and supplemental precautionary information. • POTENTIAL ADVERSE EVENTS • Potential adverse events include, but are not limited to, the following: allergic/physical/physiologic reaction, death, erosion/migration, fibrillation or other arrhythmias, lead or accessory breakage (fracture/insulation/lead tip), hematoma/seroma, inappropriate or inability to provide therapy (shocks /pacing/sensing), infection, procedure related, and component failure. Patients may develop psychological intolerance to a pulse generator system and may experience fear of shocking, fear of device failure, or imagined shocking. In rare cases severe complications or device failures can occur. • Refer to the product labeling for specific indications, contraindications, warnings/precautions and adverse events. Rx only. (Rev B) 43

Brief Summary – LATITUDE™ NXT • • • • LATITUDE™ NXT Patient Management System Intended Use The LATITUDE™ NXT Patient Management System is intended for use to remotely communicate with a compatible pulse generator from Boston Scientific CRM and transfer data to a central database. The LATITUDE NXT System provides patient data that can be used as part of the clinical evaluation of the patient. Contraindications The LATITUDE NXT Patient Management System is contraindicated for use with any implanted device other than a compatible Boston Scientific implanted device. Not all Boston Scientific implanted devices are compatible with the LATITUDE NXT System. For contraindications for use related to the implanted device, refer to the System Guide for the Boston Scientific implanted device being interrogated. Precautions Alerts may appear on the LATITUDE NXT website on a daily basis. Primary notification of alert conditions is through the View Patient List page on the LATITUDE NXT website. The clinician needs to log onto the LATITUDE NXT website in order to receive alerts. Although secondary notification through email and SMS text messages is available, these reminders are dependent on external systems and may be delayed or not occur. The secondary notification feature does not eliminate or reduce the need to check the website. Implanted device data and alerts are typically available for review on the LATITUDE NXT website within 15 minutes of a successful interrogation. However, data uploads may take significantly longer (up to 14 days). If the Communicator is unable to interrogate the implanted device or if the Communicator is unable to contact the LATITUDE NXT server to upload data, up to 14 days may elapse before the LATITUDE NXT server detects these conditions and informs the clinic user that monitoring is not occurring. If both of these conditions occur at the same time, this notification could take up to 28 days. Implanted device data and alert notification may be delayed or not occur at all under various conditions, which include but are not limited to the following: System limitations; the Communicator is unplugged; the Communicator is not able to connect to the LATITUDE NXT server through the configured phone system; the implanted device and the Communicator cannot establish and complete a telemetry session; the Communicator is damaged or malfunctions; the patient is not compliant with prescribed use or is not using the LATITUDE NXT System as described in the patient manual; if subscribed to the LATITUDE Cellular Data Plan, missing two or more payments discontinues the subscription; the clinic user can identify any patients that are not being monitored as described above by using the Not Monitored filter on the View Patient List. Adverse Effects: None known. System Limitations: The LATITUDE NXT System does not provide continuous real time monitoring. As a remote monitoring system, the LATITUDE NXT System provides periodic patient monitoring based on clinician configured settings. There are many internal and external factors that can hinder, delay, or prevent acquisition and delivery of implanted device, sensor, and patient information as intended by the clinician. These factors include: implanted device clock; patient environment; cellular data service; telephone system; communicator memory capacity; clinic environment; schedule/configuration changes; or data processing. Refer to the product labeling for specific instructions for use. Rx only. (Rev. D) 44
- Slides: 44