Physical Science EOC Review Marilyn Pendley Instructor CCCMC






















![Radioactive Decay Radioactive decay results in the emission [or release] of either: • an Radioactive Decay Radioactive decay results in the emission [or release] of either: • an](https://slidetodoc.com/presentation_image_h2/9fb0e561d0cdacade92d1dd10c0d577f/image-51.jpg)


















- Slides: 89

Physical Science EOC Review Marilyn Pendley, Instructor, CCCMC Modified by R. Kittrell Jan 2015 & 5 Jan 2016 1

2

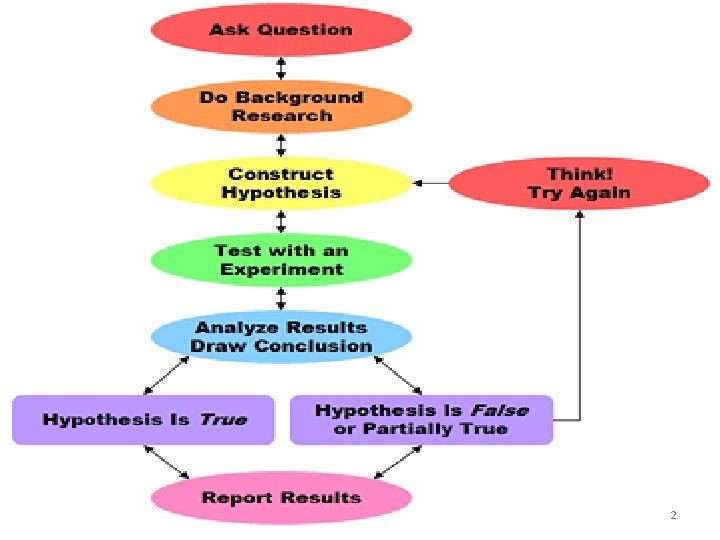
Scientific Experimentation controlled experiment -Only one variable should be changed a time. Manipulated (independent) variable – changed by the experimenter. Responding (dependent) variable – the effect that is measured. at 3

Law vs. Theory • Develop a law – Law: Summarizes the outcome of several experiments that occur repeatedly and consistently. – Example: The spoiled food served at lunch makes people sick with a stomachache. • Develop a theory – Theory: Explanation for why a law exists. – Example: It is the bacteria in the spoiled food that makes people ill. 4

Types of Measurements 1. Length a. The distance from one point to point. b. Base unit is the meter (m). c. Tool is the metric ruler. another 2. Volume a. The amount of space a substance occupies. b. Base unit is the liter (L). c. Tools: metric ruler for regular solids or graduated cylinder for liquids. 5

3. Mass a. The amount of matter in a substance. b. Base unit - kilogram (kg). c. Tool is the balance. 4. Weight a. A measure of gravitational force on an object. b. Unit is the newton (N). c. Tool is the scale. 6

5. Time a. How long an event takes to occur. b. Unit is the second (s). c. Tool is the clock (stopwatch). 6. Temperature a. The amount of kinetic energy a substance has. b. SI unit is the Kelvin (K). c. Tool is thermometer. 7

7. Density a. How compacted the matter is in a substance. b. Units can be g/m. L, g/cm 3, kg/m 3. c. Density is a derived unit (it is made up of other types of measurement). d. D = m / v e. Objects float if their density is less than the density of the fluid they are in. 8

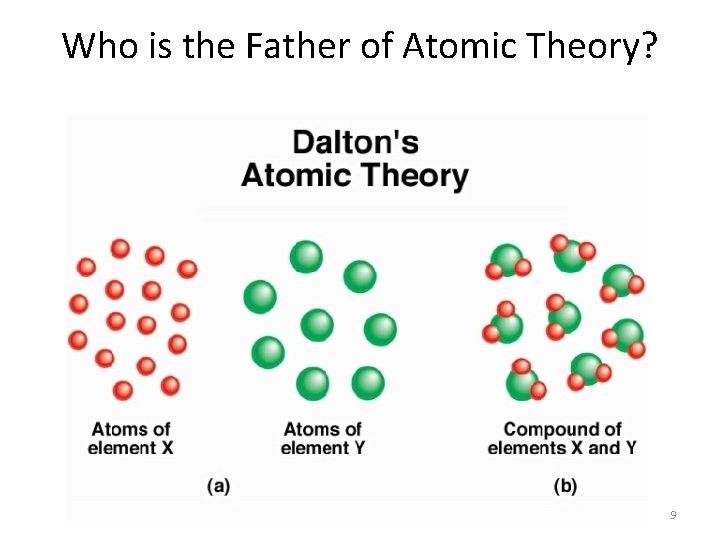
Who is the Father of Atomic Theory? 9

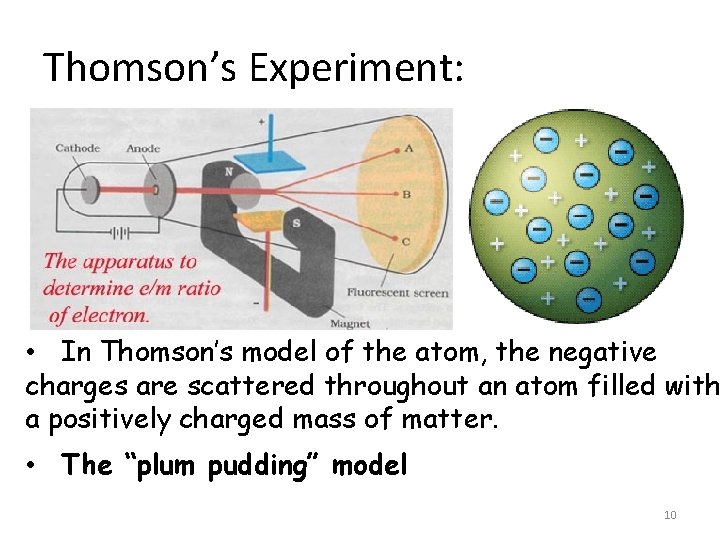
Thomson’s Experiment: • In Thomson’s model of the atom, the negative charges are scattered throughout an atom filled with a positively charged mass of matter. • The “plum pudding” model 10

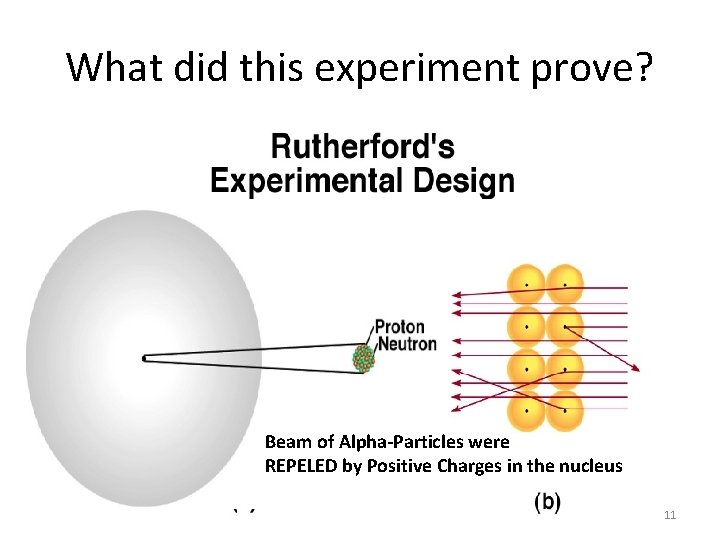
What did this experiment prove? Beam of Alpha-Particles were REPELED by Positive Charges in the nucleus 11

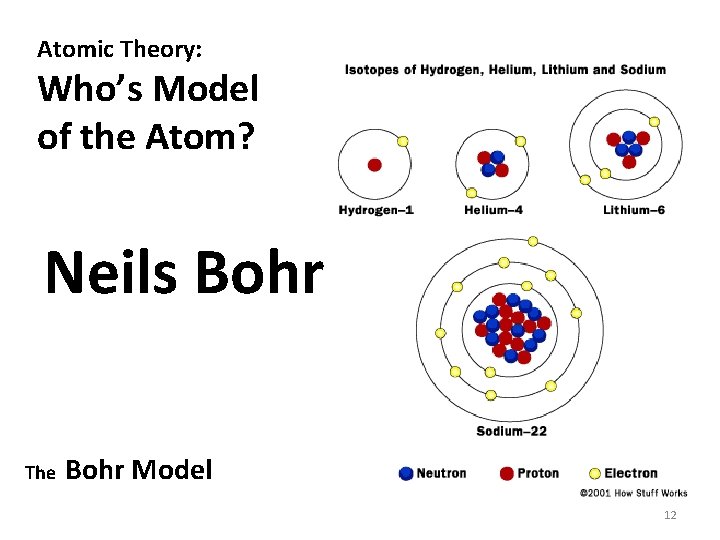
Atomic Theory: Who’s Model of the Atom? Neils Bohr The Bohr Model 12

Electron_______ Cloudis a visual model An _____ of the most likely locations for electrons in an atom. www. unitedstreaming. com – Physical Science – Elements, Compounds, and Atoms An Orbital is a region of space around the nucleus where an electron is likely to be found. * An electron cloud is a good approximation of how electrons behave in their orbitals. 13

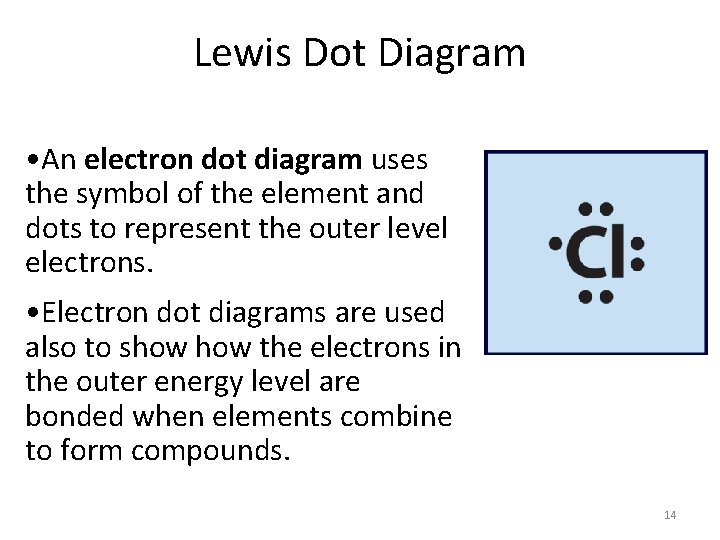
Lewis Dot Diagram • An electron dot diagram uses the symbol of the element and dots to represent the outer level electrons. • Electron dot diagrams are used also to show the electrons in the outer energy level are bonded when elements combine to form compounds. 14

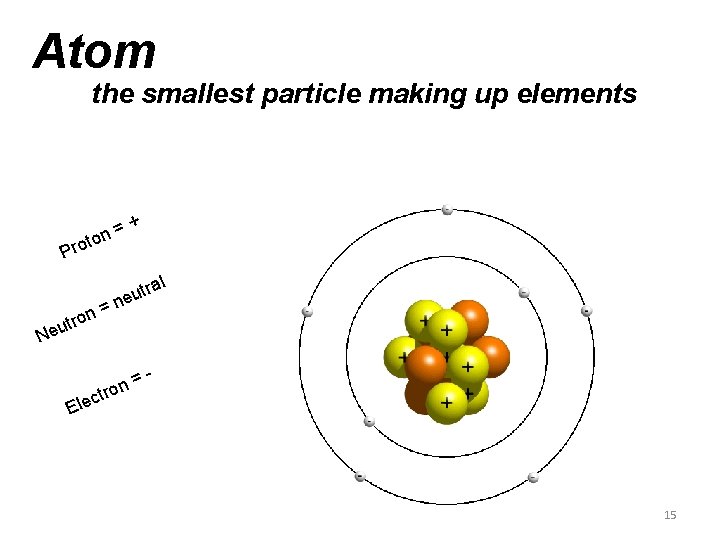
Atom the smallest particle making up elements n= o t ro + P = ron t u Ne ral t u ne = n o r t - c Ele 15

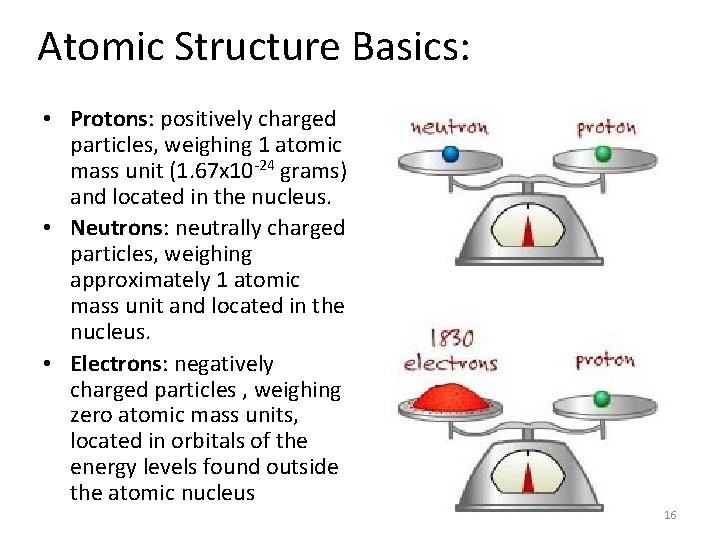
Atomic Structure Basics: • Protons: positively charged particles, weighing 1 atomic mass unit (1. 67 x 10 -24 grams) and located in the nucleus. • Neutrons: neutrally charged particles, weighing approximately 1 atomic mass unit and located in the nucleus. • Electrons: negatively charged particles , weighing zero atomic mass units, located in orbitals of the energy levels found outside the atomic nucleus 16

Atomic Number: • The number of protons • Play the Name the Atom in an atom determines Game at: what element it is. • http: //www. learner. org • Add or subtract even /interactives/periodic/b one proton from an asics_interactive. html atom of any element and you no longer have the original element in any form. Now you have a different element! 17

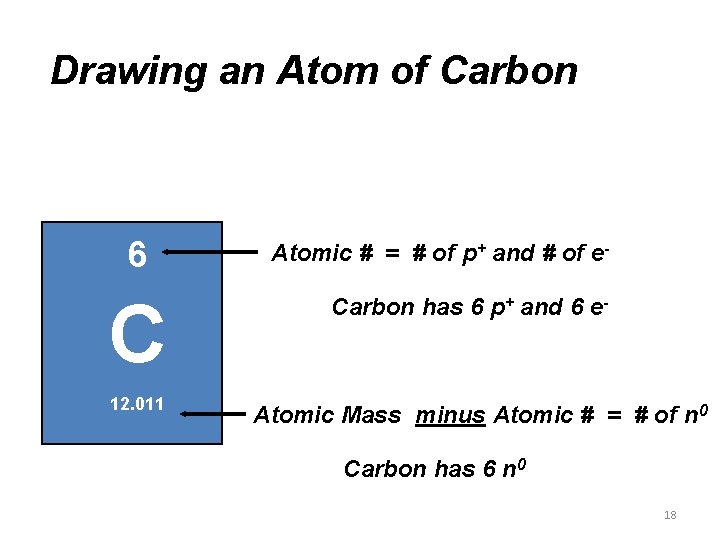
Drawing an Atom of Carbon 6 C 12. 011 Atomic # = # of p+ and # of e. Carbon has 6 p+ and 6 e- Atomic Mass minus Atomic # = # of n 0 Carbon has 6 n 0 18

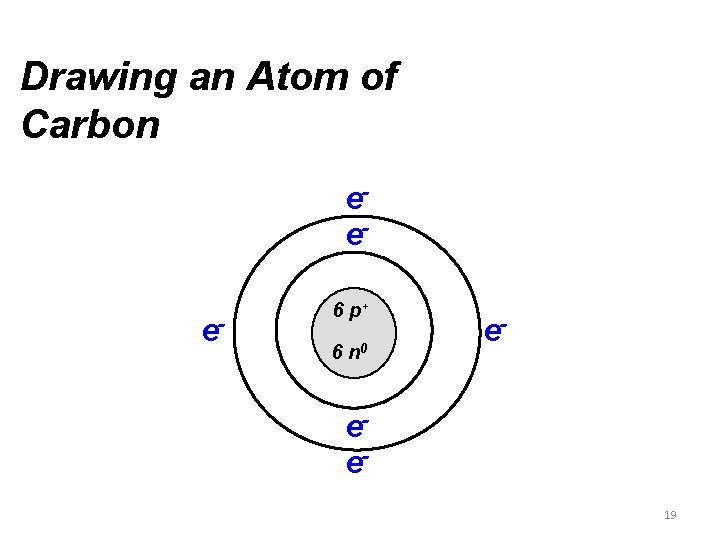
Drawing an Atom of Carbon eee- 6 p+ 6 n 0 e- ee 19

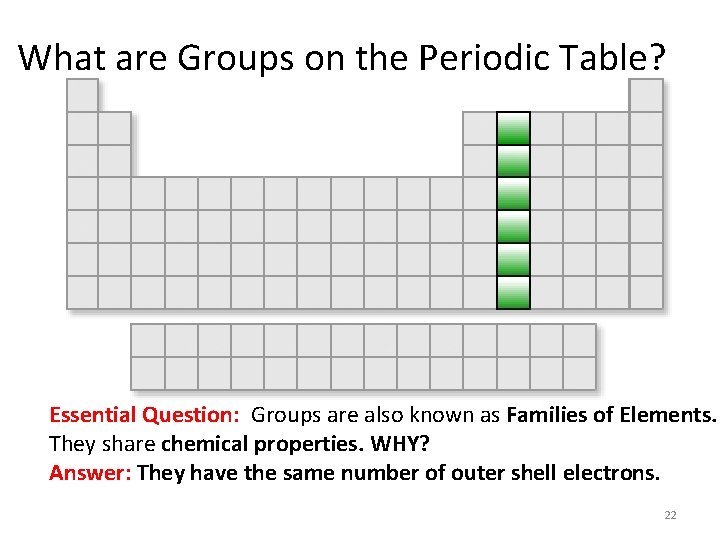
Periodic Table Basics: Essential Question: What are Horizontal Rows called? Answer: PERIODS 20

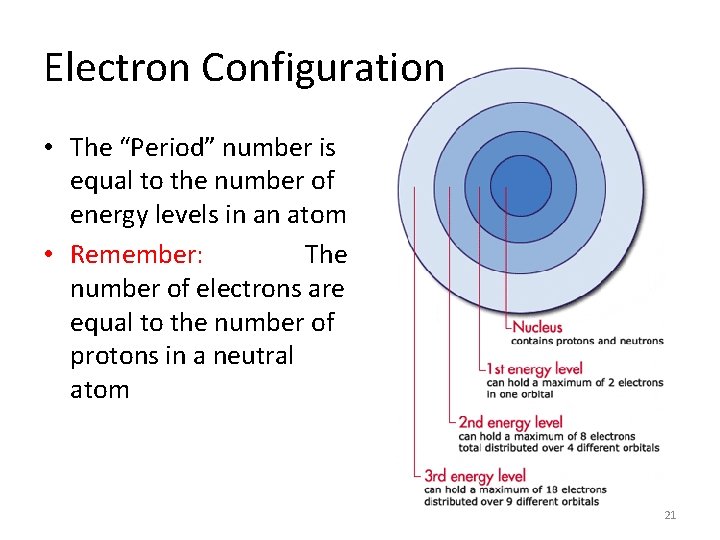
Electron Configuration • The “Period” number is equal to the number of energy levels in an atom • Remember: The number of electrons are equal to the number of protons in a neutral atom 21

What are Groups on the Periodic Table? Essential Question: Groups are also known as Families of Elements. They share chemical properties. WHY? Answer: They have the same number of outer shell electrons. 22

Electron Configuration • For Groups IA – VIII the group numbers are equal to the number of outer shell electrons or valence electrons • Group “IA” has ONE valence electron • Group “IIA” has TWO valence electrons; etc. 23

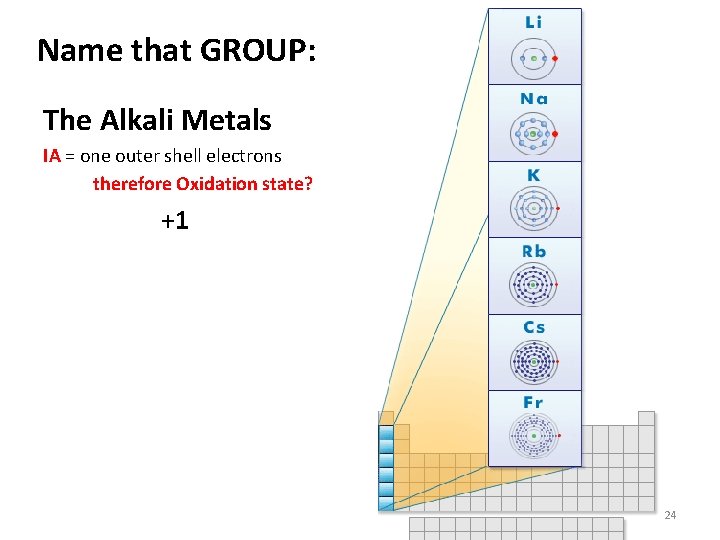
Name that GROUP: The Alkali Metals IA = one outer shell electrons therefore Oxidation state? +1 24

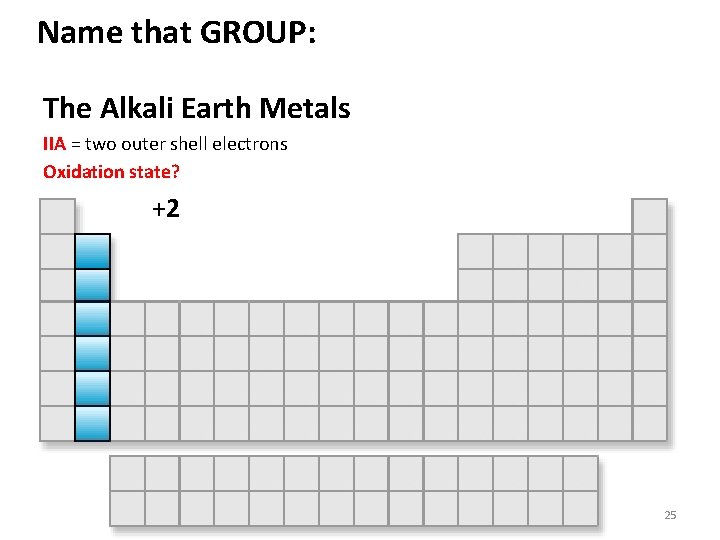
Name that GROUP: The Alkali Earth Metals IIA = two outer shell electrons Oxidation state? +2 25

Name that GROUP: The Noble Gases VIIIA = eight outer shell electrons (except Helium which has 2 but is FULL) therefore Oxidation state? NOT! 26

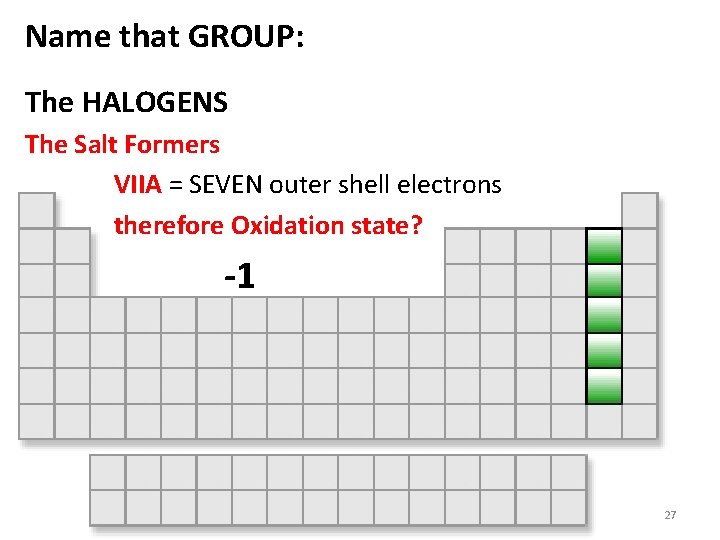
Name that GROUP: The HALOGENS The Salt Formers VIIA = SEVEN outer shell electrons therefore Oxidation state? -1 27

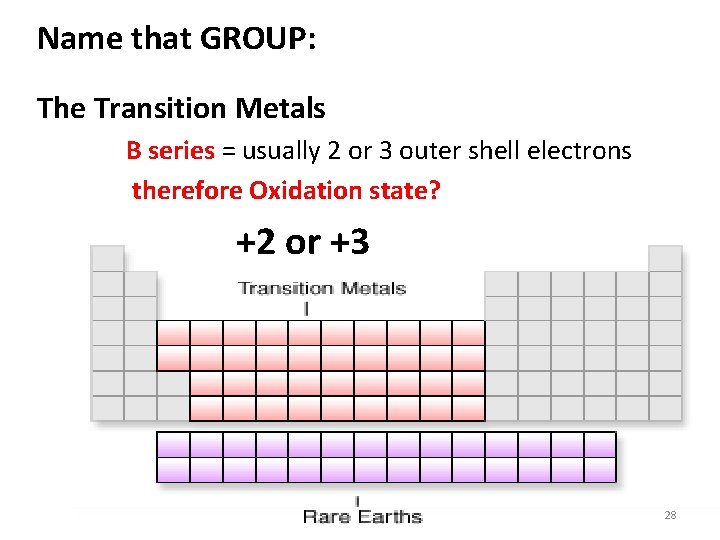
Name that GROUP: The Transition Metals B series = usually 2 or 3 outer shell electrons therefore Oxidation state? +2 or +3 28

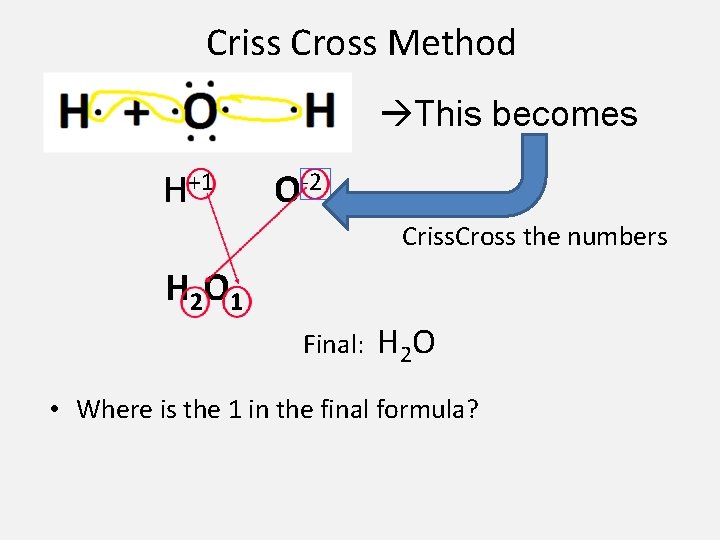
Criss-Cross Method • Determine the charges/oxidation #’s for each element. • By criss-crossing the charges of the elements you can easily write the chemical formula • Example: Hydrogen + Oxygen – H + O IONS = H+1 + O– 2 – Criss cross the oxidation # • just the numbers - not the + / - signs – Write the numbers as subscripts • bottom right of symbol

Criss Cross Method This becomes H+1 O-2 Criss. Cross the numbers H 2 O 1 Final: H 2 O • Where is the 1 in the final formula?

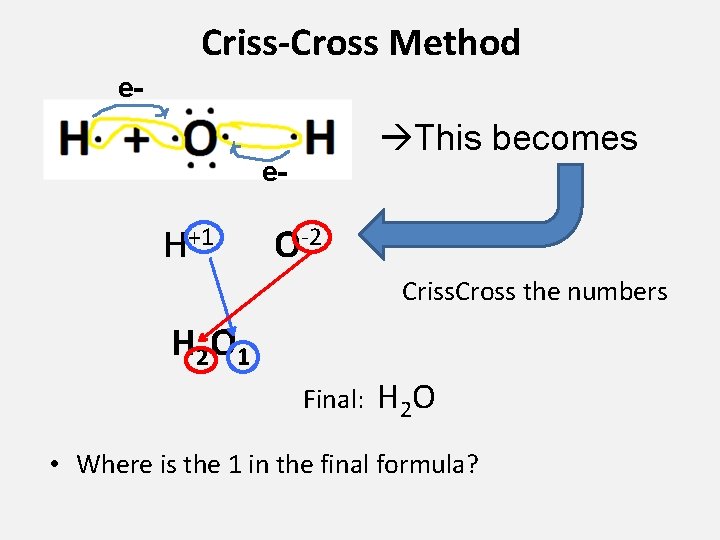
Criss-Cross Method e- This becomes e- H+1 O-2 Criss. Cross the numbers H 2 O 1 Final: H 2 O • Where is the 1 in the final formula?

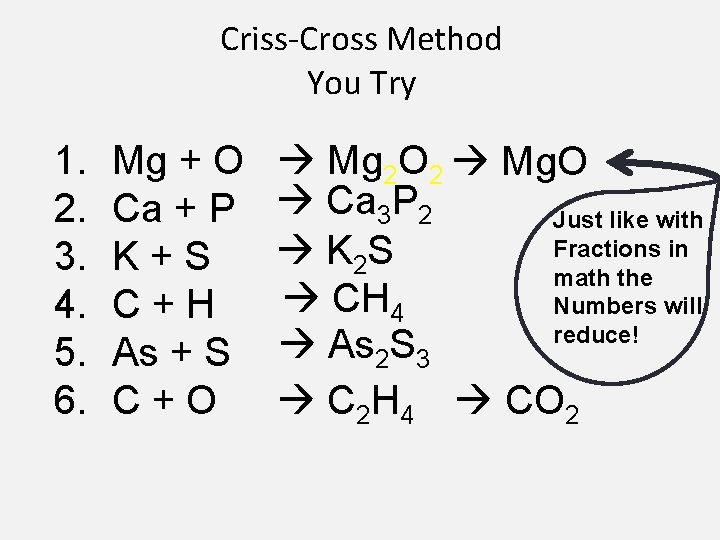
Criss-Cross Method - -You Try it! Mg + O Ca + P K+S C+H As + S C+O

Criss-Cross Method You Try 1. 2. 3. 4. 5. 6. Mg + O Ca + P K+S C+H As + S C+O Mg 2 O 2 Mg. O Ca 3 P 2 Just like with Fractions in K 2 S math the CH 4 Numbers will reduce! As 2 S 3 C 2 H 4 CO 2

Criss-Cross & Polyatomic Ions • Sodium + Sulfate – Na + SO 4 – Na+1 + (SO 4)-2 Do the Criss-Cross Na 2(SO 4)1 Final: Na 2(SO 4)

Chemical Bonding: • Three types of bonding http: //www. learner. org/interactives/periodic/groups_interactive • Ionic Bonding • Covalent • Metallic Bonding Animations and short clips AWESOME MOVIE ON BONDING 30 minutes long 35

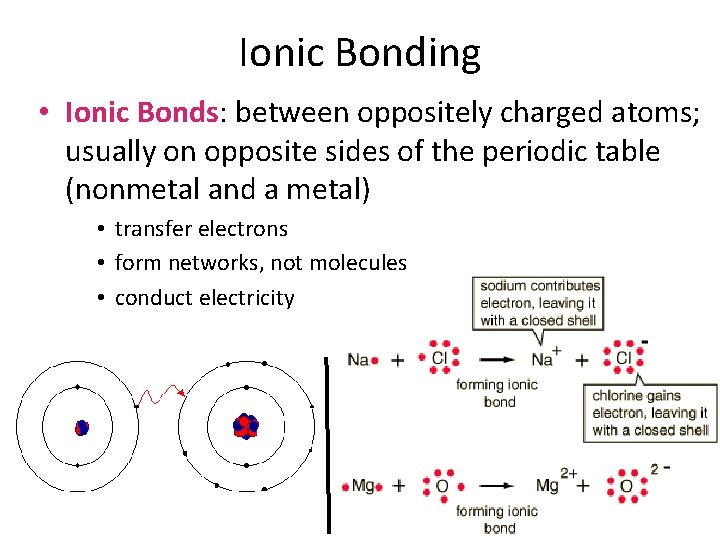
Ionic Bonding • Ionic Bonds: between oppositely charged atoms; usually on opposite sides of the periodic table (nonmetal and a metal) • transfer electrons • form networks, not molecules • conduct electricity

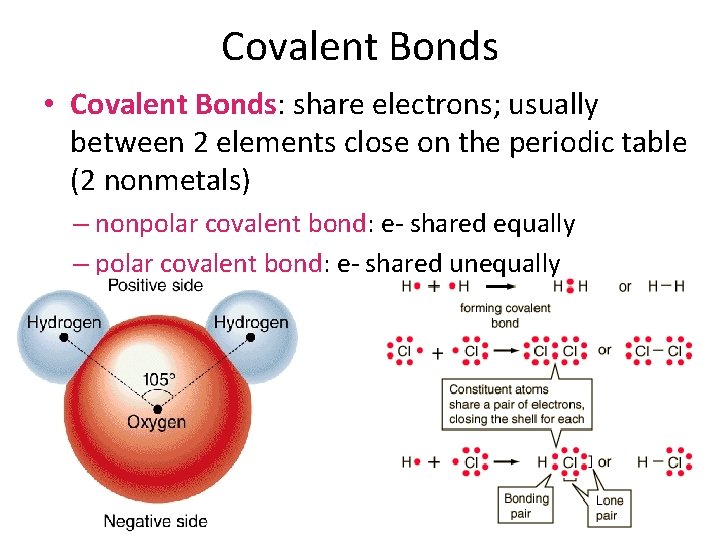
Covalent Bonds • Covalent Bonds: share electrons; usually between 2 elements close on the periodic table (2 nonmetals) – nonpolar covalent bond: e- shared equally – polar covalent bond: e- shared unequally

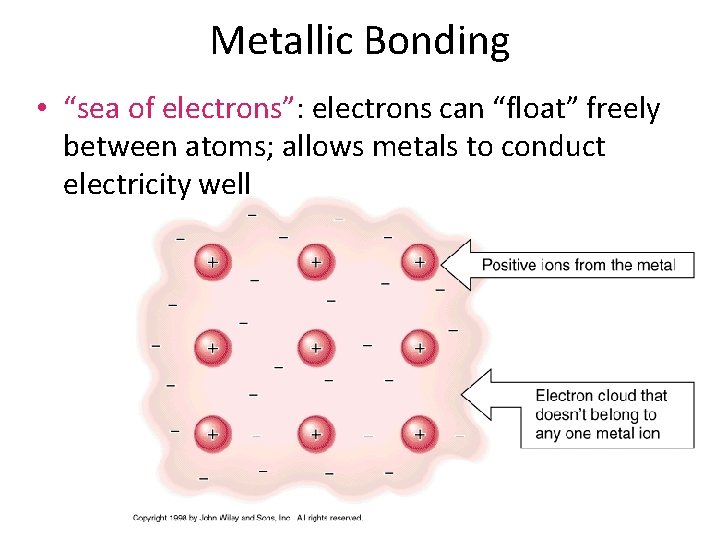
Metallic Bonding • “sea of electrons”: electrons can “float” freely between atoms; allows metals to conduct electricity well

Rules for naming and writing chemical formulas 1. Metal name comes first 2. Change the ending of the second element to “ide” • If you have two nonmetals, use the prefixes: Mono – 1 Penta - 5 Di - 2 Hexa - 6 Tri - 3 Hepta - 7 Tetra – 4 Octa - 8 39

Which of the following is the correct name of a combination of sodium and iodine? • a) sodium chloride • b) sodium iodide • c) sodium iodine • d) iodine sodium 40

Transition Metal Naming • Transition metals show their charges as Roman Numerals because they can change charge!! Fe. O = Fe+2 + O-2 Iron (II) Oxide Fe 2 O 3 = Fe+3 + O-2 Iron (III) Oxide

42

Chemical Reactions Types • Combustion: ALWAYS has O 2 as a REACTANT! – AB + O 2 AO + BO • Single-Replacement (single-displacement): • AX + B A + BX • Double-Replacement (double-displacement): • AX + BY AY + BX 43

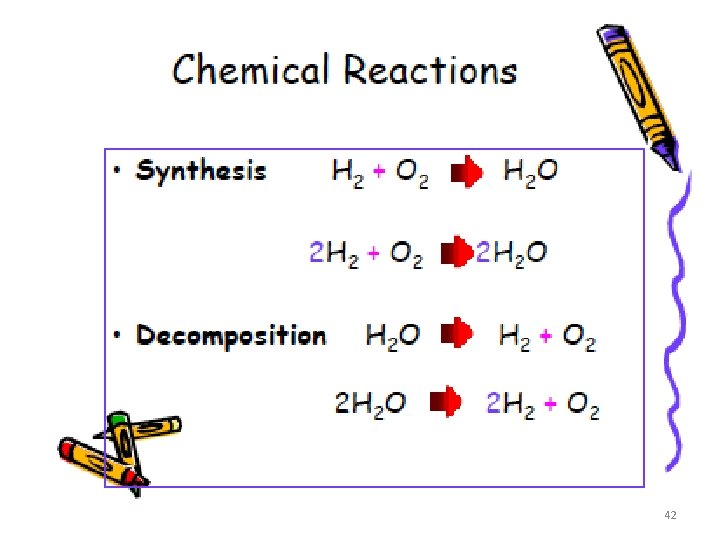
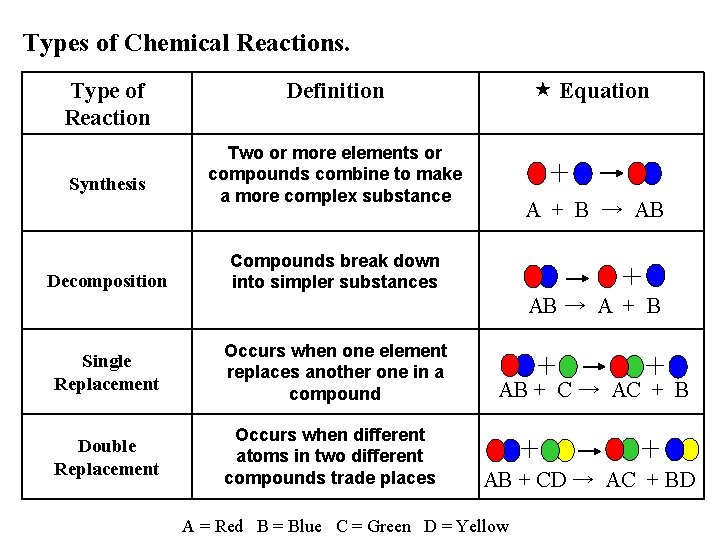
Types of Chemical Reactions. Type of Reaction Synthesis Decomposition Equation Definition Two or more elements or compounds combine to make a more complex substance A + B → AB Compounds break down into simpler substances AB → A + B Single Replacement Occurs when one element replaces another one in a compound Double Replacement Occurs when different atoms in two different compounds trade places AB + C → AC + B AB + CD → AC + BD A = Red B = Blue C = Green D = Yellow

Identifying Chemical Reactions Use colored pencils to circle the common atoms or compounds in each equation to help you determine the type of reaction it illustrates. Use the code below to classify each reaction. S = Synthesis D = Decomposition ____ P + O 2 → P 4 O 10 ____ Hg. O → ____ Cl 2 + Hg + Na. Br → SR = Single Replacement DR = Double Replacement ____ Mg + O 2 Na. Cl + Br 2 O 2 → ____ Al 2 O 3 Mg. O → Al ____ H 2 + N 2 → + NH 3 O 2

____ Na ____ Hg. O + Br 2 → + Cl 2 → Na. Br ____ Cu. Cl 2 + Hg. Cl + O 2 ____ C + ____ KCl. O 3 → KCl + ____ Ba. Cl 2 + Na 2 SO 4 → O 2 Na. Cl + ____ S 8 Ba. SO 4 + H 2 S → H 2 → F 2 → Cu. S + CH 4 SF 6 HCl

Acids & Bases • The strength of an acid or base depends on how many acid or base particles dissociate into ions in water. • Strong Acid/Base - + – 100% ions in water – strong electrolyte – HCl, HNO 3, Na. OH, Li. OH • Weak Acid/Base • few ions in water • weak electrolyte • HC 2 H 3 O 2, NH 3 - + 47

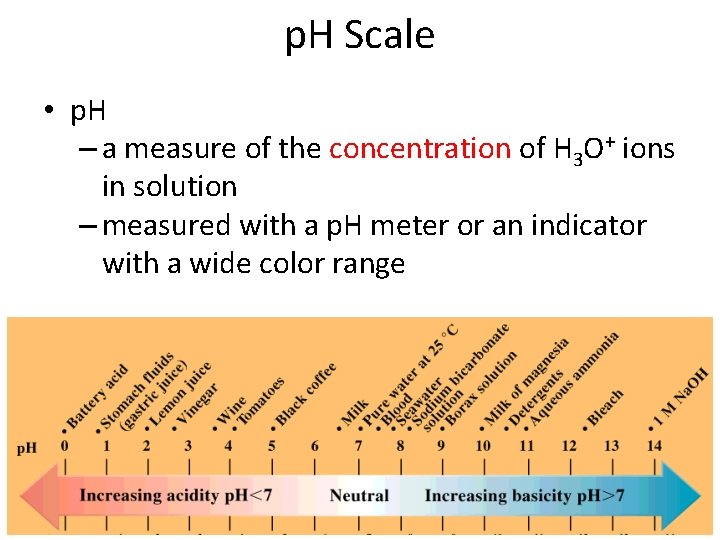
p. H Scale • p. H – a measure of the concentration of H 3 O+ ions in solution – measured with a p. H meter or an indicator with a wide color range

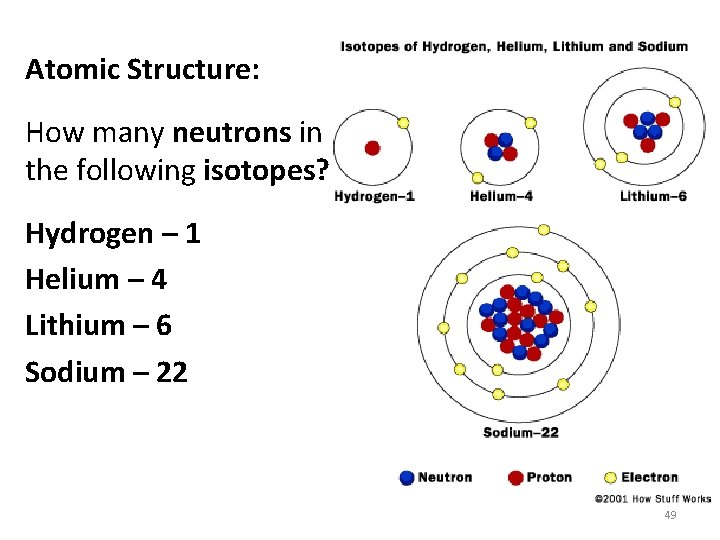
Atomic Structure: How many neutrons in the following isotopes? Hydrogen – 1 Helium – 4 Lithium – 6 Sodium – 22 49

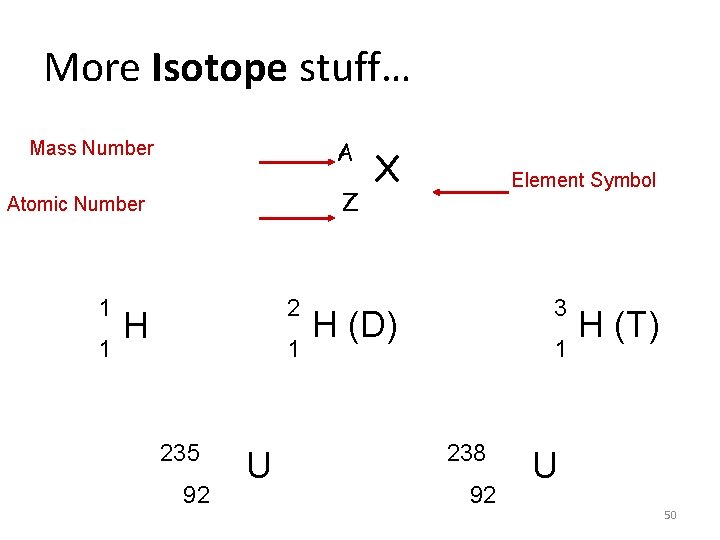
More Isotope stuff… A Mass Number Z Atomic Number 1 1 2 H 1 235 92 U X Element Symbol 3 H (D) 1 238 92 H (T) U 50
![Radioactive Decay Radioactive decay results in the emission or release of either an Radioactive Decay Radioactive decay results in the emission [or release] of either: • an](https://slidetodoc.com/presentation_image_h2/9fb0e561d0cdacade92d1dd10c0d577f/image-51.jpg)
Radioactive Decay Radioactive decay results in the emission [or release] of either: • an alpha particle (a), • a negative beta particle (electron) (b-), • a positive beta particle (positron) (b+), • or a gamma ray (g). In a nuclear reaction the MASS and ATOMIC NUMBER must be the SAME on both sides of the equations

Alpha Decay An alpha particle is identical to that of a helium nucleus. It contains two protons and two neutrons. A X Z A-4 4 Y He + Z-2 2 unstable atom alpha particle more stable atom

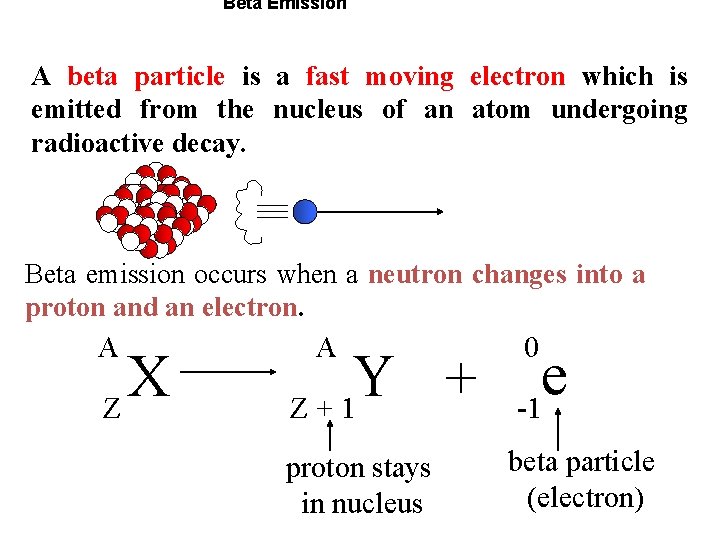
Beta Emission A beta particle is a fast moving electron which is emitted from the nucleus of an atom undergoing radioactive decay. Beta emission occurs when a neutron changes into a proton and an electron. A X Z A 0 Y + Z+1 e -1 proton stays in nucleus beta particle (electron)

Gamma Decay • When atoms decay by emitting a or b particles to form a new atom, the nuclei of the new atom may still have too much energy to be completely stable. These unstable atoms will emit gamma rays to release that energy. • There is no change in mass or atomic number A X Z A Z X + 0 g 0

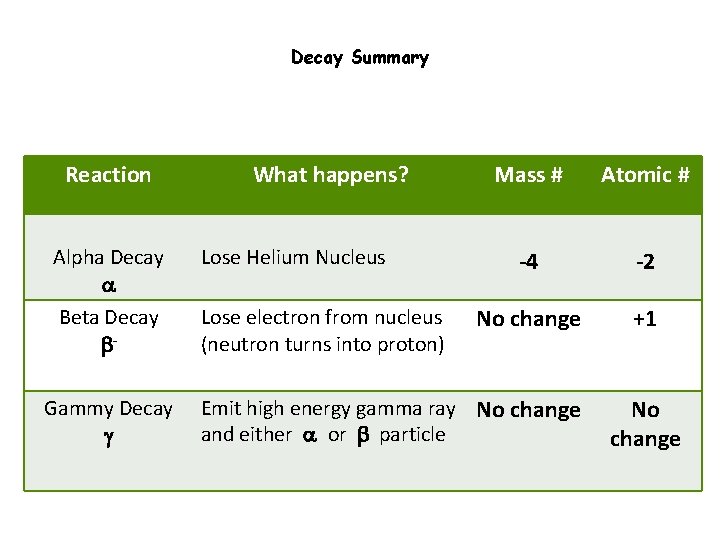
Decay Summary Reaction Alpha Decay a Beta Decay b. Gammy Decay g What happens? Lose Helium Nucleus Lose electron from nucleus (neutron turns into proton) Mass # Atomic # -4 -2 No change +1 Emit high energy gamma ray No change and either a or b particle No change

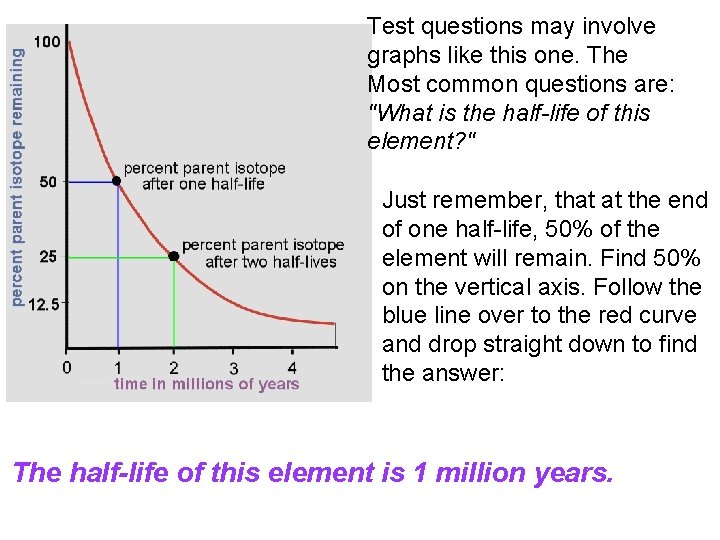
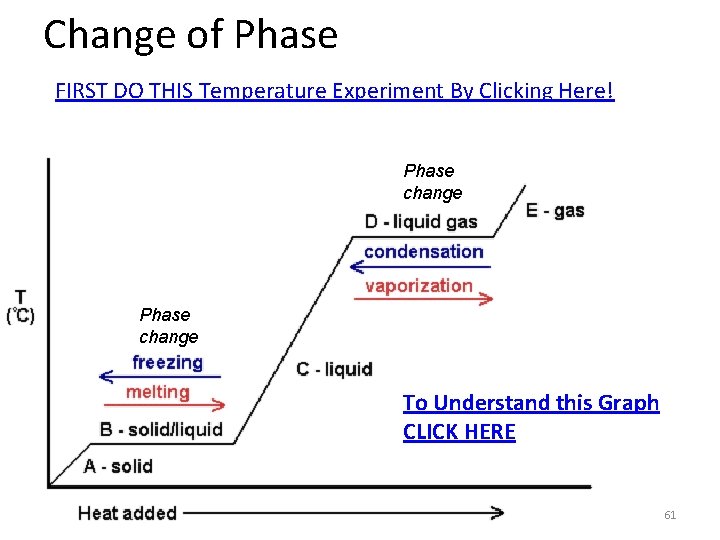
Test questions may involve graphs like this one. The Most common questions are: "What is the half-life of this element? " Just remember, that at the end of one half-life, 50% of the element will remain. Find 50% on the vertical axis. Follow the blue line over to the red curve and drop straight down to find the answer: The half-life of this element is 1 million years.

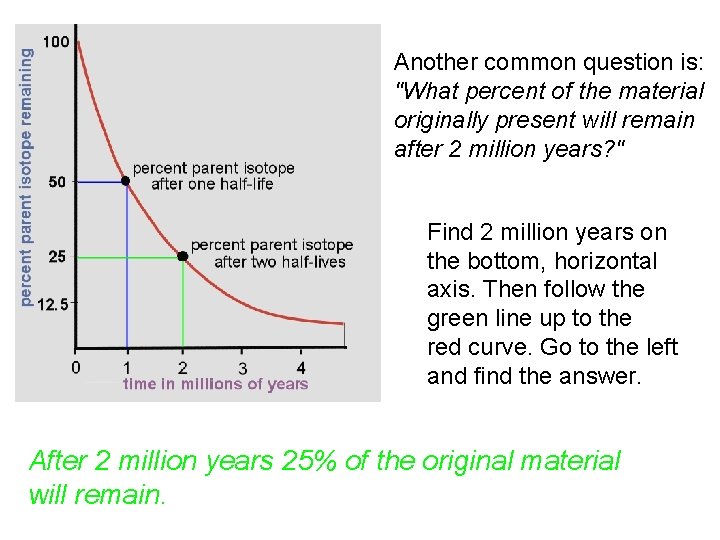
Another common question is: "What percent of the material originally present will remain after 2 million years? " Find 2 million years on the bottom, horizontal axis. Then follow the green line up to the red curve. Go to the left and find the answer. After 2 million years 25% of the original material will remain.

Nuclear Energy Essential Questions: • Why is it worth the RISK? ? • Tremendous OUTPUT of ENERGY!! • Fission or Fusion? • Fission! Atoms of U-235 are split • Use the link to see how Nuclear Fusion works: • Fusion! Atoms are joined or fused together. http: //science. howstuffworks. com/fusion-reactor. htm/printable 58

Heat energy: • Direction of Energy Flow? • HOT toward COLD 59

States of Matter • Watch what happens to molecules when heated by doing cool virtual experiments. • Click on the icon below to get started: 60

Change of Phase FIRST DO THIS Temperature Experiment By Clicking Here! Phase change To Understand this Graph CLICK HERE 61

Chemical or Physical Change? Chemical Change • New substances formed with new properties • Examples: • Rusting • Gas forming during a reaction (bubbles) • A precipitant forming during a reaction Physical Change • No new substances formed • Examples: • Ice melting • Water evaporating • Dry ice subliming into Carbon dioxide • Salt or sugar dissolving in water 62

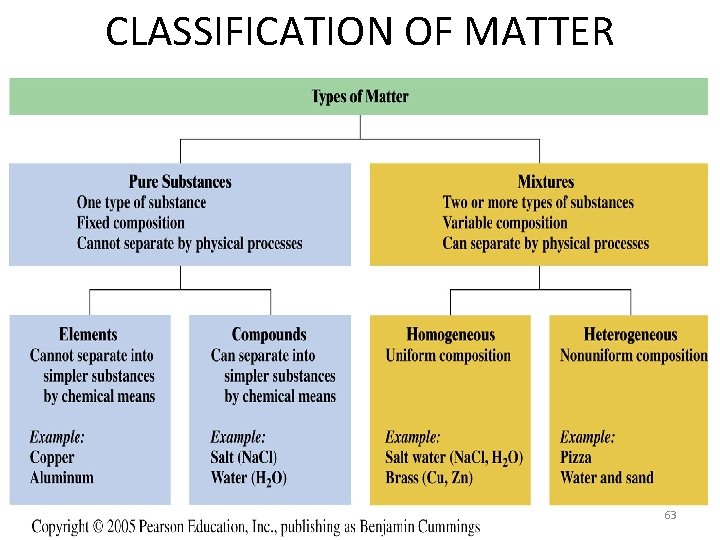
CLASSIFICATION OF MATTER 63

ELEMENTS • contain only one type of atom • building blocks of matter • 115 known elements today, 90 which occur naturally • Found on periodic table – The first letter is always capitalized, the second letter is always lower case • Fluorine is F, not f • Cobalt is Co, not CO (which is carbon monoxide) 64

Compounds - 2 or more elements chemically combined to form a new substance with new properties Properties – The way a chemical substance looks and behaves 65

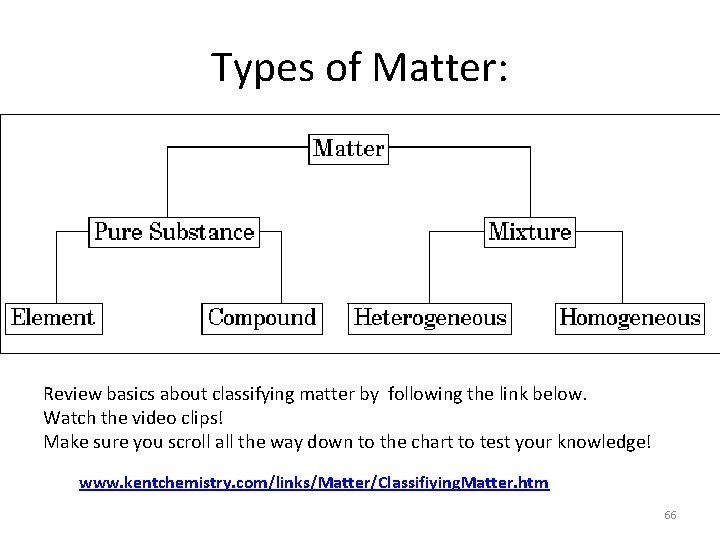
Types of Matter: Review basics about classifying matter by following the link below. Watch the video clips! Make sure you scroll all the way down to the chart to test your knowledge! www. kentchemistry. com/links/Matter/Classifiying. Matter. htm 66

Mixtures and Pure Substances – A mixture has unlike parts and a composition that varies from sample to sample • A heterogeneous mixture has physically distinct parts with different properties. • A homogeneous mixture is the same throughout the sample – Pure substances are substances with a fixed composition 67

Matter comes in 3 phases Solid Gas Liquid 68

Solid Definite Shape Definite Volume 69

Liquid Indefinite Shape – takes the shape of the container Definite Volume 70

Gas Indefinite Shape – takes the shape of the container. Indefinite Volume – can expand can be compressed. 71

Plasma • • • Plasma: a high energy gaseous state of matter. It is very unstable. Particles are moving extremely fast, free energy Most abundant phase of matter Only present under extremely hot/energetic situations • Example: Sun 72

CHANGES IN STATE • Melting: Solid to liquid • Boiling: Liquid to gas • Sublimation: Solid to gas – The above three require input of energy • Condensation: Gas to liquid • Freezing: Liquid to solid • Deposition: Gas to solid – The above three release energy 73

74

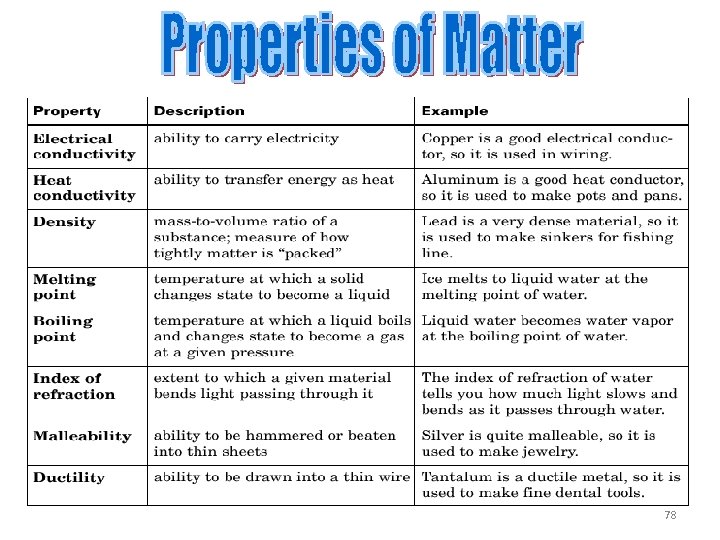
PHYSICAL PROPERTIES • Characterize the physical state and physical behavior of a substance {appearance, smell, feel} • Each substance has unique physical properties • Examples – Sulfur appears as a yellow powder – The boiling point of water is 100 o. C – Carbon monoxide is odorless 75

76

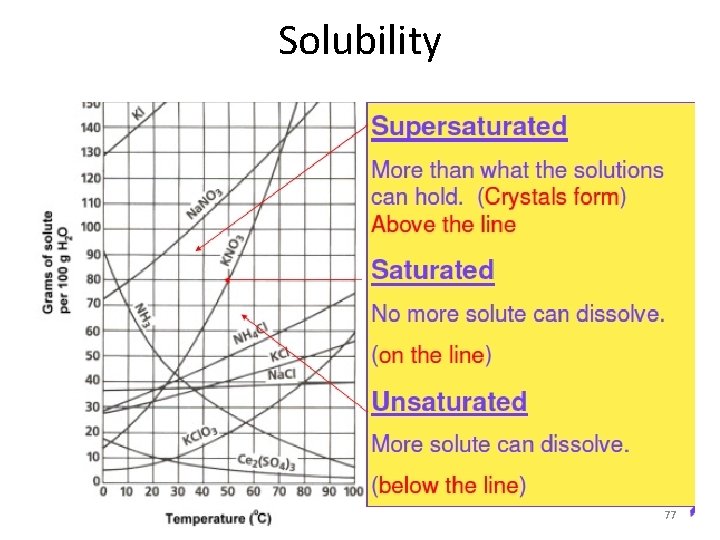
Solubility 77

78

Where is Magnetism Concentrated? Magnetism is strongest at the POLES 79

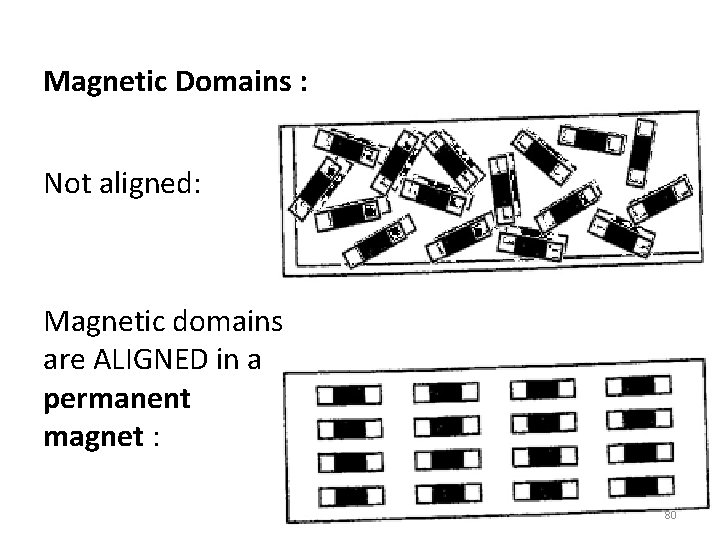
Magnetic Domains : Not aligned: Magnetic domains are ALIGNED in a permanent magnet : 80

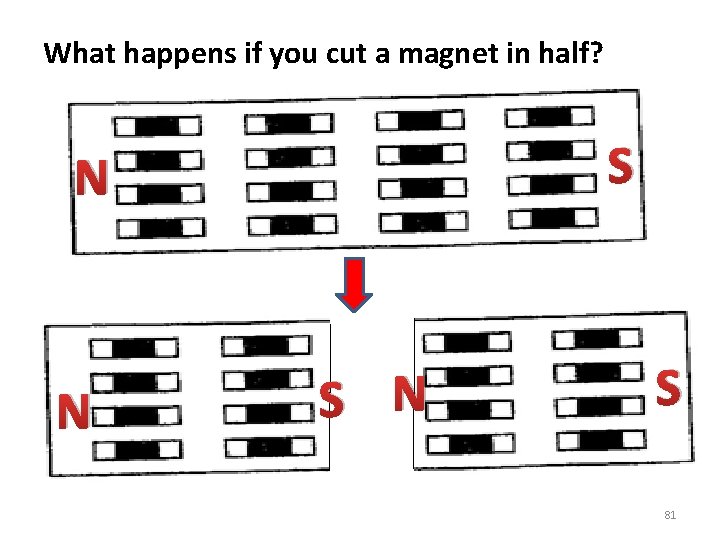
What happens if you cut a magnet in half? S N N S 81

Electricity and Magnetism: Electromagnetism 82

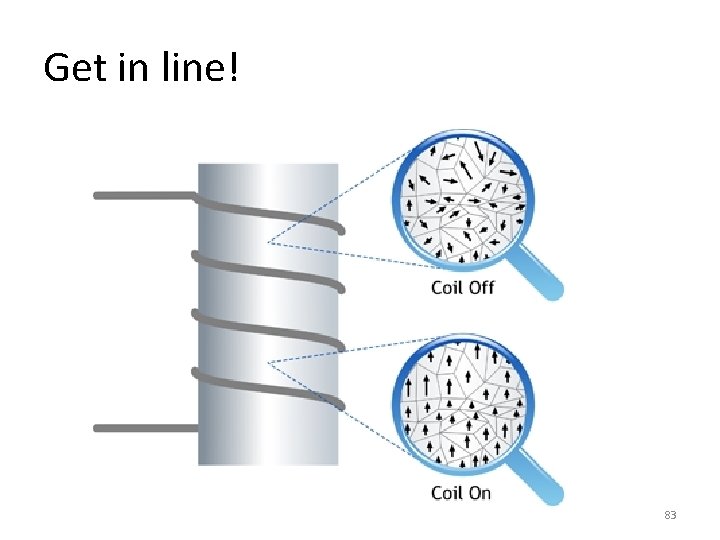
Get in line! 83

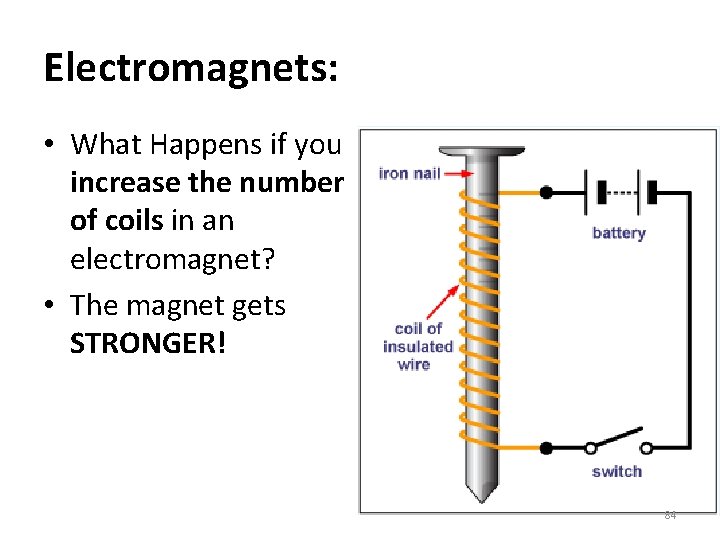
Electromagnets: • What Happens if you increase the number of coils in an electromagnet? • The magnet gets STRONGER! 84

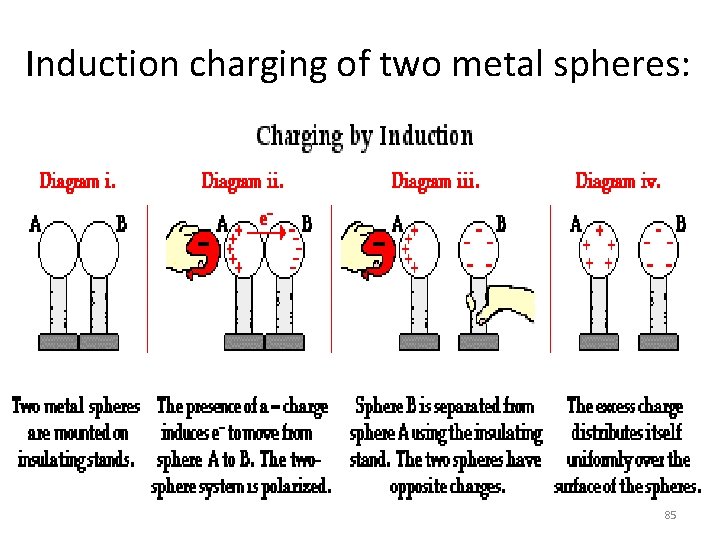
Induction charging of two metal spheres: 85

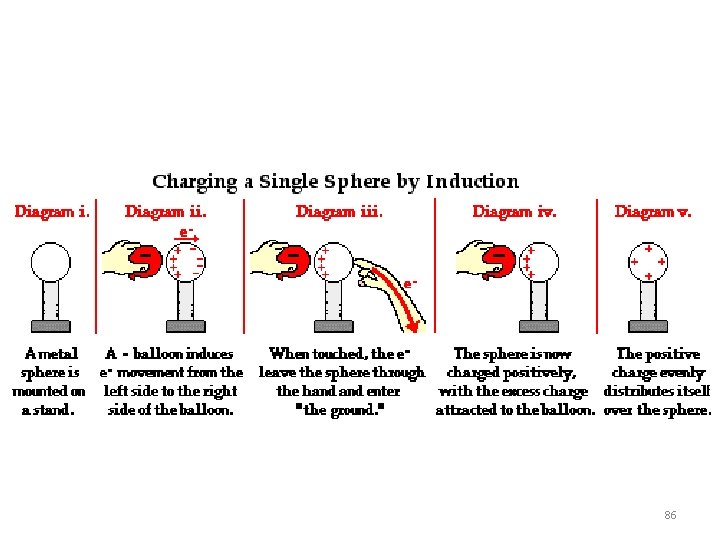
86

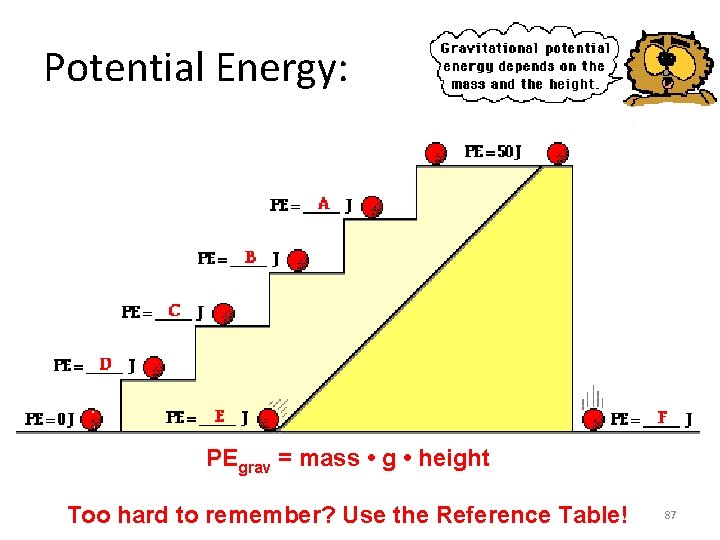
Potential Energy: PEgrav = mass • g • height Too hard to remember? Use the Reference Table! 87

Answer key: A: PE = 40 J (since the same mass is elevated to 4/5 -ths height of the top stair) B: PE = 30 J (since the same mass is elevated to 3/5 -ths height of the top stair) C: PE = 20 J (since the same mass is elevated to 2/5 -ths height of the top stair) D: PE = 10 J (since the same mass is elevated to 1/5 -ths height of the top stair) E and F: PE = 0 J (since the same mass is at the same zero height position as shown for the bottom stair Note since PEgrav = m * • g • h Doubling of the height will result in a doubling of the gravitational potential energy. • A tripling of the height will result in a tripling of the gravitational potential energy. 88

Physics Portion • Make a given table that lists the information you are given. BE SURE to include the item you are to find! • USE the Reference sheet! Find the equation that fits what you have. • Put the item you need to find on one side of the equals sign. • Add the other numbers and punch in the calculator. • Double check the answer from the calculator! 89