Physical Property A characteristic of matter that you



















- Slides: 32

Physical Property A characteristic of matter that you can observe without changing the identity of the matter. COLOR ODOR DENSITY STATE MELTING POINT VOLUME MASS LENGTH HEIGHT

Extensive and Intensive Properties An extensive property of a material depends upon how much matter is being considered. • mass • length • volume An intensive property of a material does not depend upon how much matter is being • density considered. • temperature • color 1. 6

Solids Particles packed close together and are fixed in position. though they may vibrate Incompressible. Retain their shape and volume when placed in a new container, don’t “flow” Low energy 5

Liquids Closely packed, some ability to move around. Also largely incompressible. take the shape of their container and “flow”; won’t escape or expand to fill the total container. Medium Energy 8

Gases Constantly movement, bumping into each other and the container. lots of empty space between the particles. on average High Energy 9

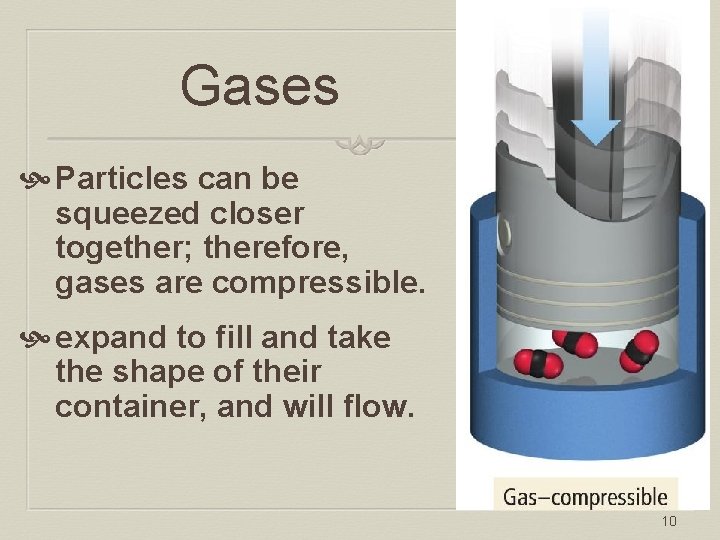
Gases Particles can be squeezed closer together; therefore, gases are compressible. expand to fill and take the shape of their container, and will flow. 10

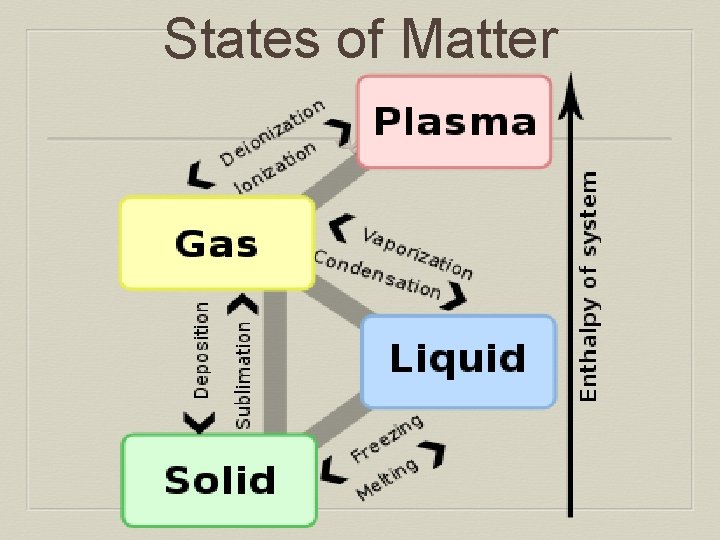
States of Matter

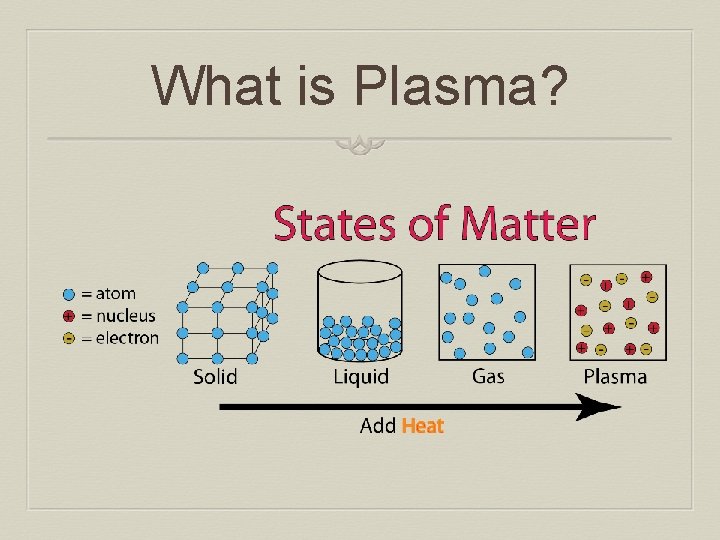
What is Plasma?

Chemical Property A chemical property is a characteristic that becomes evident during a chemical reaction. It’s any quality that can be established only by changing a substance's chemical identity. Does it combust? Does it tarnish? Does it react with Does it rust? water? oxygen? Does it ferment?

Chemical Changes in Matter The rusting of iron is a chemical change. The iron atoms in the nail combine with oxygen atoms from O 2 in the air to make a new substance, rust, with a different composition. 19

Common Chemical Changes • processes that cause changes in the matter that change its composition • rusting • processes that release lots of energy • burning C 3 H 8(g) + 5 O 2(g) → 3 CO 2(g) + 4 H 2 O(l)

Signs of chemical change Gas forms – bubbles or odor Precipitate (solid) forms – becomes cloudy or see sediment settle to the bottom Release/absorption of energy – change in temperature or gives off light Color change

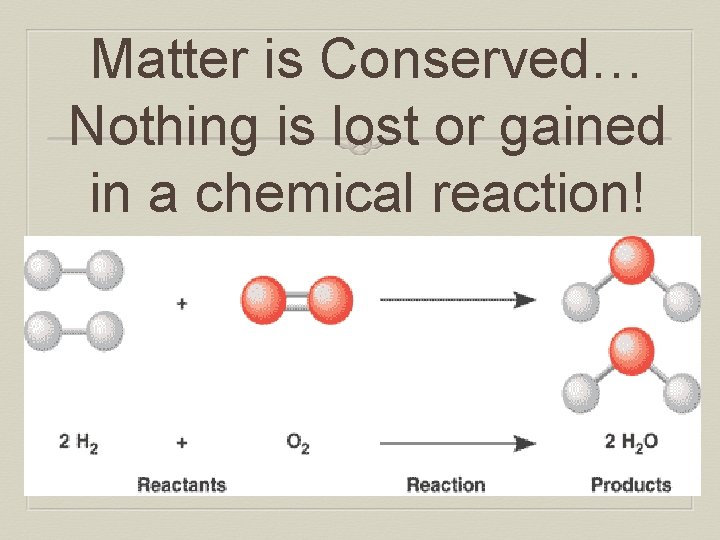
Matter is Conserved… Nothing is lost or gained in a chemical reaction!

Summary - Physical change Form of substance changes (size, state of matter) but not the composition Ex- change in state of matter Ex- dissolving sugar Ex- Crushing solid into powder

Summary Chemical Change Identity and composition of substance changes (new product forms) In a chemical reaction: Reactants products Atoms of reactants break apart and rearrange to join with different atoms to make the new product but all of the atoms originally there are still there at the end

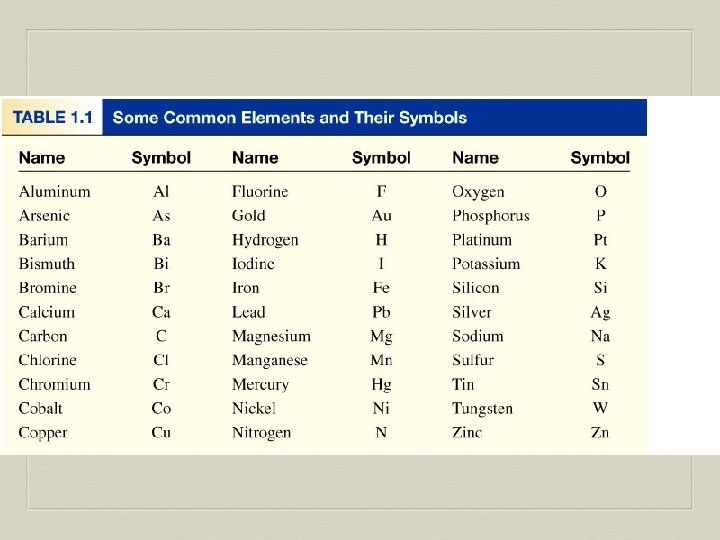
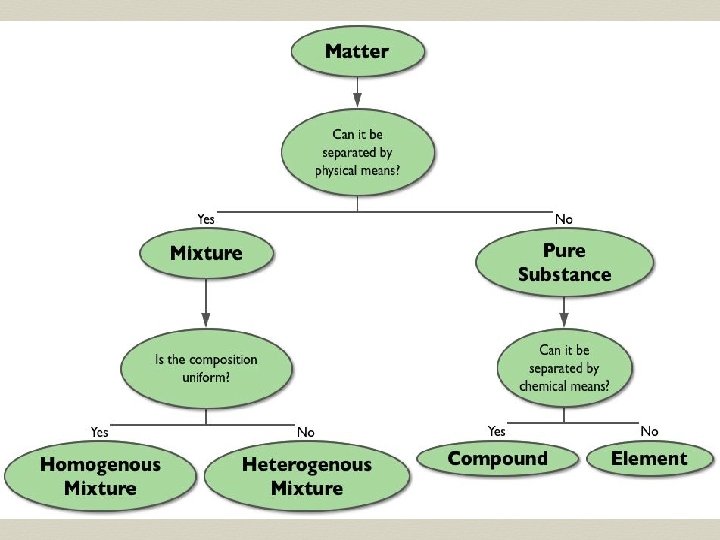
Elements An element (atom) is a pure substance that cannot be separated into simpler substances by chemical means. ² 118 elements have been identified ² 82 elements occur naturally on Earth ²gold, aluminum, lead, oxygen, carbon 1. 4


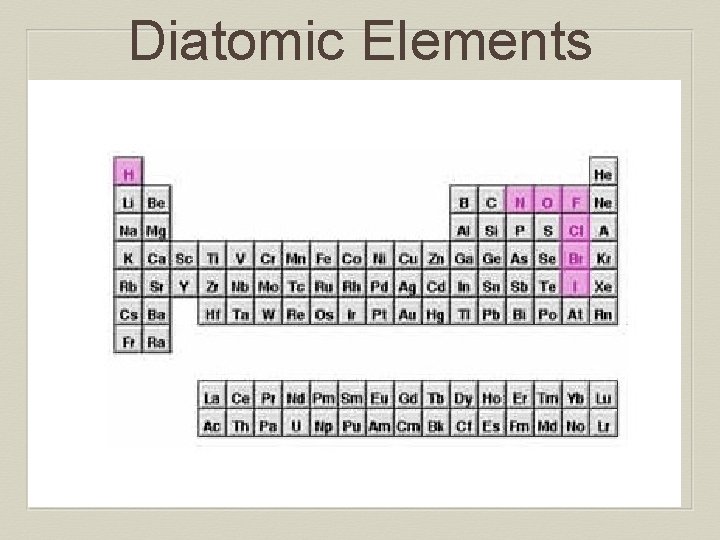
Diatomic Elements A pure substance containing two atoms of the same element combined: • Seven elements: • H, N, O, F, Cl, Br, I • Tend to be stable • Can be chemically broken apart

Diatomic Elements

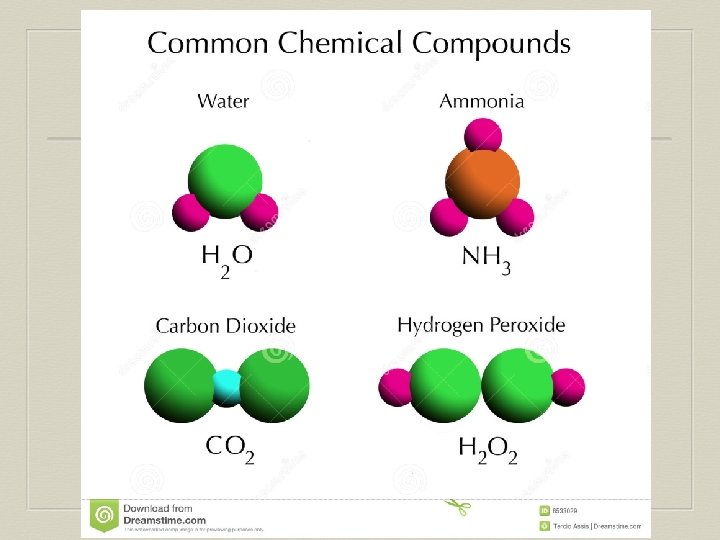
Compounds A pure substance containing two or more different elements: • Distinct physical & chemical properties • Can be chemically broken down to other substances • Unique structure & ratio of


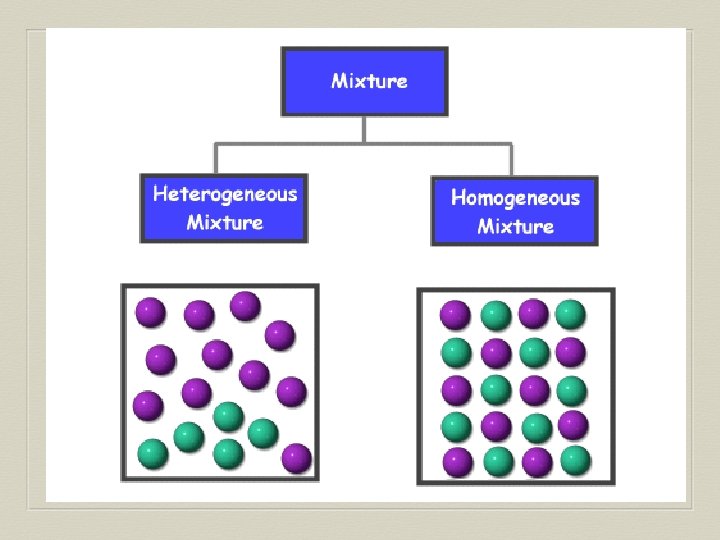
Mixtures Any combination of two or more different substances: • Not considered “pure” • Each part retains its distinct physical & chemical properties • Can be physically broken down to other substances

Homogeneous Mixture A mixture that is the same throughout. • Different substances are evenly spread out • Similar properties throughout • Also referred to as a

Heterogenous Mixture A mixture with areas of different concentrations of each substance. • Substances are clumped or layered together • Different areas may have different properties



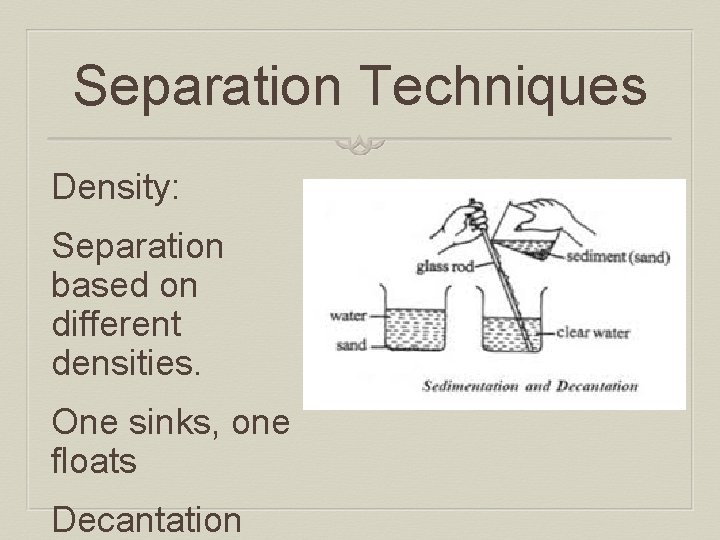
Separation Techniques Density: Separation based on different densities. One sinks, one floats Decantation

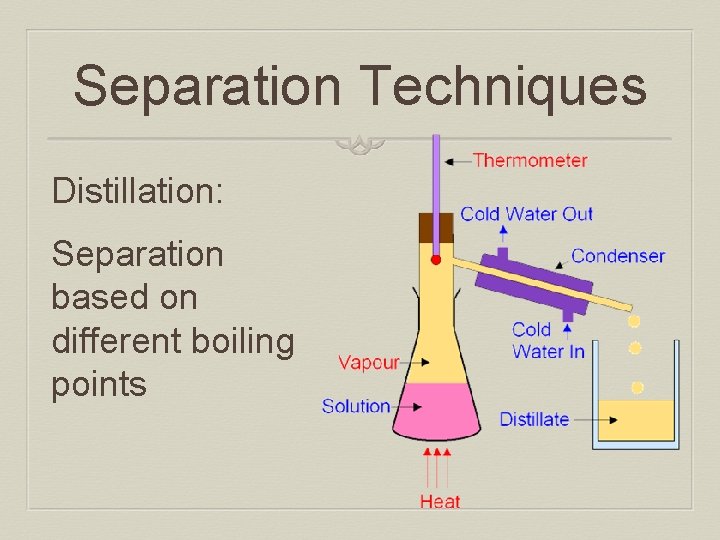
Separation Techniques Distillation: Separation based on different boiling points

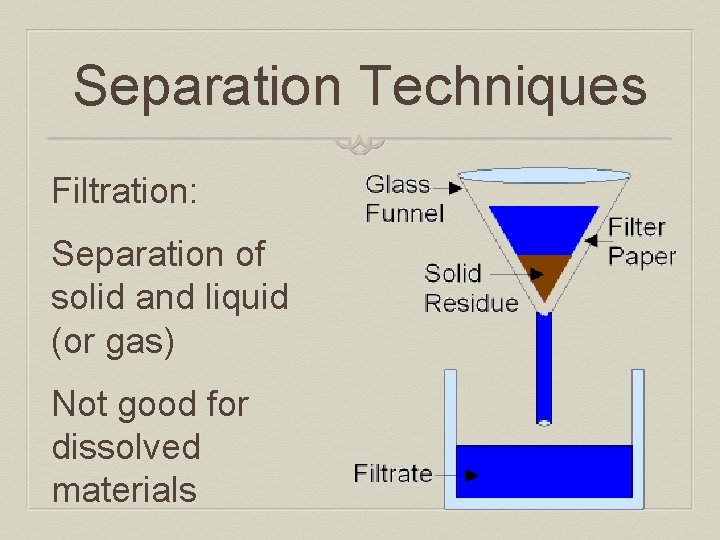
Separation Techniques Filtration: Separation of solid and liquid (or gas) Not good for dissolved materials

Separation Techniques Evaporation: Dry mixture until one substance completely evaporates

Separation Techniques Sifting : Separate small and large particles

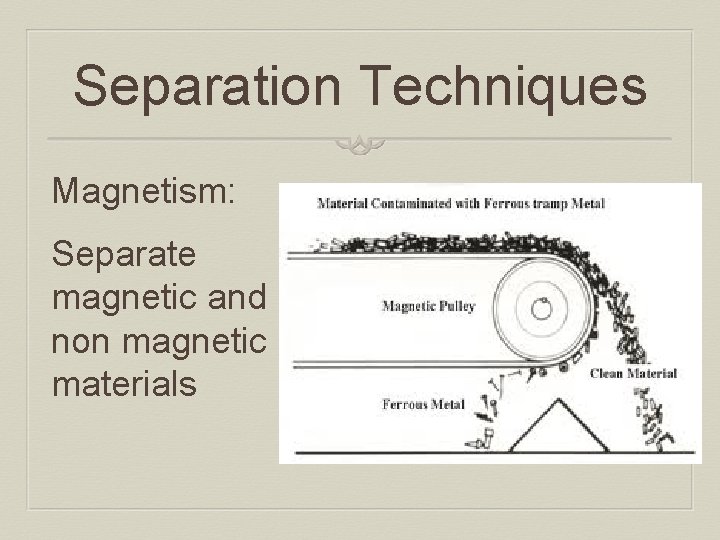
Separation Techniques Magnetism: Separate magnetic and non magnetic materials