Physical Properties Section 2 2 Physical Properties G














- Slides: 15

Physical Properties (Section 2. 2)

Physical Properties G Is any characteristic of a material that can be observed or measured without changing the composition of the substances in the material G Viscosity, conductivity, malleability, hardness, melting point, boiling point, and density are examples of physical properties

Viscosity G Viscosity is the tendency of a liquid to keep from flowing (its resistance to flowing) G The greater the viscosity, the slower the liquid moves G Thick liquids like corn syrup and honey have a high viscosity G Thin liquids like vinegar and water have a low viscosity G The viscosity of a liquid usually decreases

Conductivity G A material’s ability to allow heat to flow G Materials that have a high conductivity, such as metals, are called good conductors G If a material is a good conductor of heat it is usually also a good conductor of electricity G Wood is not a good conductor

Malleability G Is the ability of a solid to be hammered without shattering G Most metals are malleable G Solids that shatter when struck are brittle

Hardness G One way to compare the hardness of two materials is to see which of the materials can scratch the other G Diamond is the hardest known material Tip of drill is coated in diamond

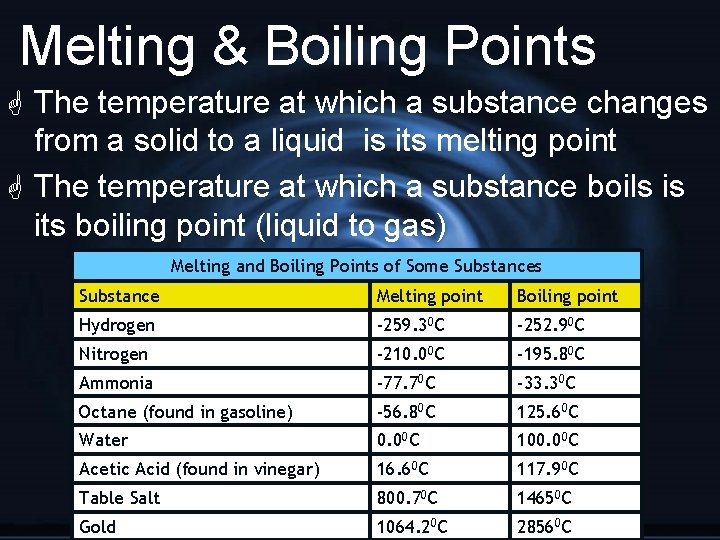
Melting & Boiling Points G The temperature at which a substance changes from a solid to a liquid is its melting point G The temperature at which a substance boils is its boiling point (liquid to gas) Melting and Boiling Points of Some Substances Substance Melting point Boiling point Hydrogen -259. 30 C -252. 90 C Nitrogen -210. 00 C -195. 80 C Ammonia -77. 70 C -33. 30 C Octane (found in gasoline) -56. 80 C 125. 60 C Water 0. 00 C 100. 00 C Acetic Acid (found in vinegar) 16. 60 C 117. 90 C Table Salt 800. 70 C 14650 C Gold 1064. 20 C 28560 C

Density G Is the ratio of the mass of a substance to its volume G Density = Mass Volume G Can be used to test the purity of a substance G Silver has a density of 10. 5 g/cm 3 at room temperature but if you have a coin with a density of 9. 9 g/cm 3 at room temperature it must not be pure

Using Properties to Identify Materials G Step 1: decide which properties to test G Step 2: do tests on a sample of the unknown G Step 3: compare the results with the data reported for the known materials When a car is involved in a hit-and-run accident detectives use paint chips from the scene of the crime to identify the make and model of the vehicle using a database of the different paint mixtures

Using Properties to Choose Materials G People don’t consider just one property when choosing a material for a particular application G Example G You wouldn’t want shoelaces made of wood or steel G Shoelaces need to be flexible, durable, and

Separating Mixtures G Some properties can be used to separate mixtures G Filtration G Uses a porous barrier to separate a solid from a liquid G Good for heterogeneous

G Distillation G Uses differences in the boiling points of the substances involved G Crystallization G Results in the formation of pure solid particles of a substance from a solution containing the dissolved substance

G Chromatography G Parts of a mixture are separated based on how fast they travel through a medium

Recognizing Physical Changes G A physical change occurs when some of the properties of a material change, but the substances in the material remain the same G Change in the state of matter

Physical Changes G Crumpling a piece of paper changes the size and shape of the paper and slicing a tomato changes the size and shape of the tomato but it does not change their composition G Some physical changes can be reversed such as melting ice and then freezing it again G Some cannot be reversed (You can’t put the sliced