Physical Properties of Solutions Chapter 12 Copyright The




















- Slides: 38

Physical Properties of Solutions Chapter 12 Copyright © The Mc. Graw-Hill Companies, Inc. Permission required for reproduction or display.

12. 1 - Types of solutions A solution is a homogenous mixture of 2 or more substances The solute is(are) the substance(s) present in the smaller amount(s) The solvent is the substance present in the larger amount 12. 1

A saturated solution contains the maximum amount of a solute that will dissolve in a given solvent at a specific temperature. An unsaturated solution contains less solute than the solvent has the capacity to dissolve at a specific temperature. A supersaturated solution contains more solute than is present in a saturated solution at a specific temperature. Sodium acetate crystals rapidly form when a seed crystal is added to a supersaturated solution of sodium acetate. 12. 1

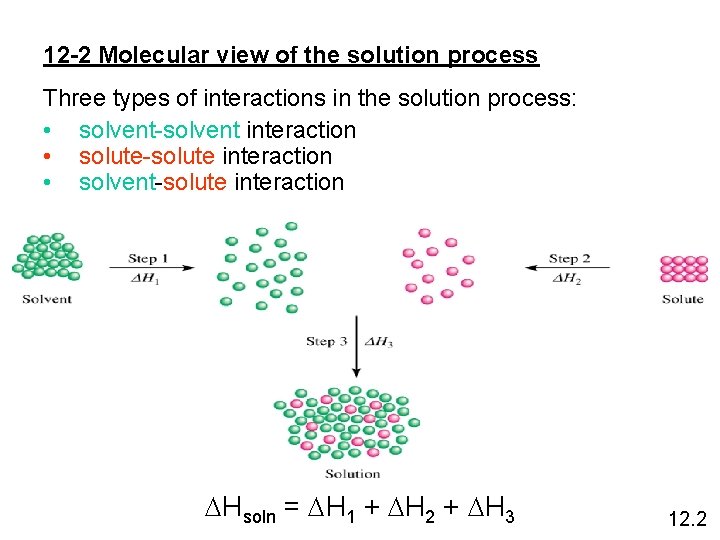
12 -2 Molecular view of the solution process Three types of interactions in the solution process: • solvent-solvent interaction • solute-solute interaction • solvent-solute interaction DHsoln = DH 1 + DH 2 + DH 3 12. 2

“like dissolves like” Two substances with similar intermolecular forces are likely to be soluble in each other. • non-polar molecules are soluble in non-polar solvents CCl 4 in C 6 H 6 • polar molecules are soluble in polar solvents C 2 H 5 OH in H 2 O • ionic compounds are more soluble in polar solvents Na. Cl in H 2 O or NH 3 (l) 12. 2

Worked Example 12. 1

12 -3 Concentration Units The concentration of a solution is the amount of solute present in a given quantity of solvent or solution. Percent by Mass mass of solute x 100% % by mass = mass of solute + mass of solvent mass of solute x 100% = mass of solution Mole Fraction (X) moles of A XA = sum of moles of all components 12. 3

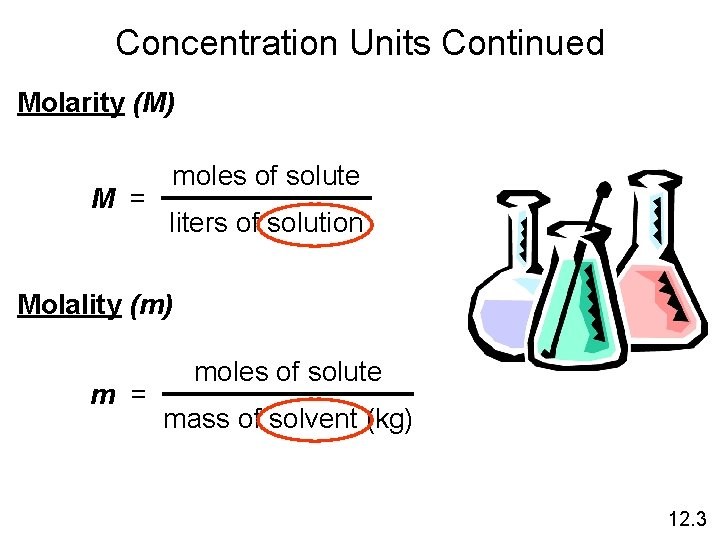
Concentration Units Continued Molarity (M) M = moles of solute liters of solution Molality (m) m = moles of solute mass of solvent (kg) 12. 3

Worked Example 12. 2


What is the molality of a 5. 86 M ethanol (C 2 H 5 OH) solution whose density is 0. 927 g/m. L? m = moles of solute mass of solvent (kg) M = moles of solute liters of solution Assume 1 L of solution: 5. 86 moles ethanol = 270 g ethanol 927 g of solution (1000 m. L x 0. 927 g/m. L) mass of solvent = mass of solution – mass of solute = 927 g – 270 g = 657 g = 0. 657 kg m = moles of solute mass of solvent (kg) = 5. 86 moles C 2 H 5 OH 0. 657 kg solvent = 8. 92 m 12. 3


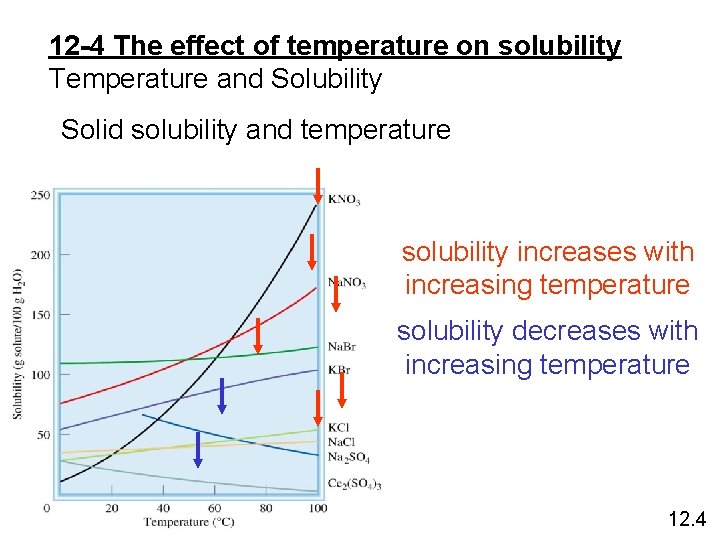
12 -4 The effect of temperature on solubility Temperature and Solubility Solid solubility and temperature solubility increases with increasing temperature solubility decreases with increasing temperature 12. 4

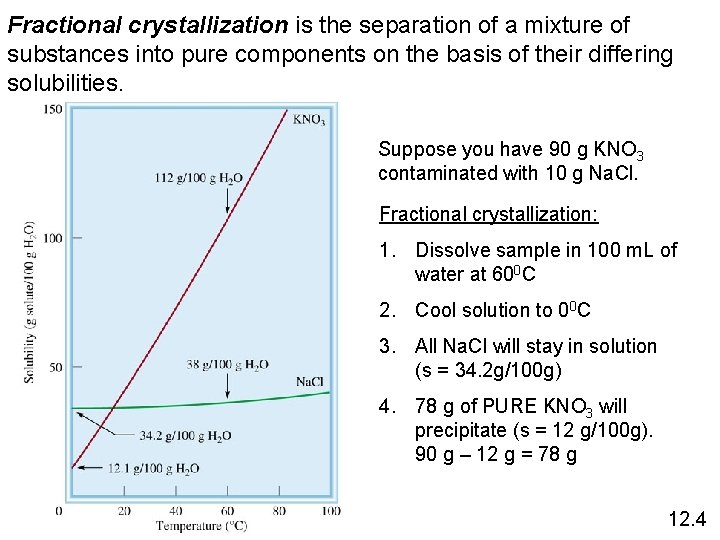
Fractional crystallization is the separation of a mixture of substances into pure components on the basis of their differing solubilities. Suppose you have 90 g KNO 3 contaminated with 10 g Na. Cl. Fractional crystallization: 1. Dissolve sample in 100 m. L of water at 600 C 2. Cool solution to 00 C 3. All Na. Cl will stay in solution (s = 34. 2 g/100 g) 4. 78 g of PURE KNO 3 will precipitate (s = 12 g/100 g). 90 g – 12 g = 78 g 12. 4

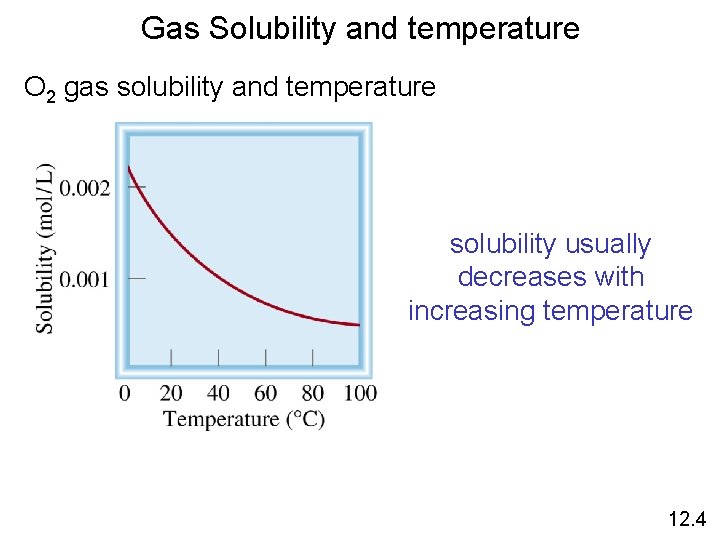
Gas Solubility and temperature O 2 gas solubility and temperature solubility usually decreases with increasing temperature 12. 4

12 -5 The effect of Pressure on the Solubility of Gases The solubility of a gas in a liquid is proportional to the pressure of the gas over the solution (Henry’s law). c is the concentration (M) of the dissolved gas c = k. P P is the pressure of the gas over the solution k is a constant for each gas (mol/L • atm) that depends only on temperature low P high P low c high c 12. 5

Worked Example 12. 6

12 -6 Colligative Properties of Nonelectrolyte Solutions Colligative properties are properties that depend only on the number of solute particles in solution and not on the nature of the solute particles. 1 -Vapor-Pressure Lowering P 1 = X 1 P 0 1 Raoult’s law P 10 = vapor pressure of pure solvent X 1 = mole fraction of the solvent If the solution contains only one solute: X 1 = 1 – X 2 P 10 - P 1 = DP = X 2 P 10 X 2 = mole fraction of the solute 12. 6

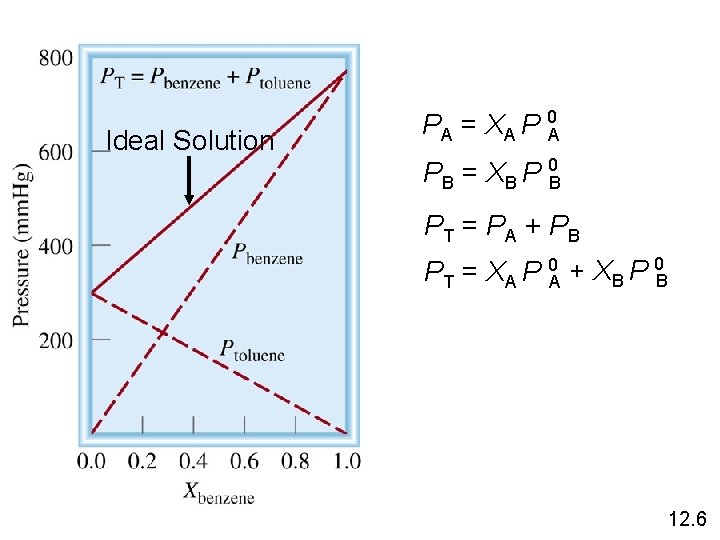
Ideal Solution PA = XA P 0 A PB = XB P 0 B PT = PA + PB PT = XA P 0 A + XB P 0 B 12. 6

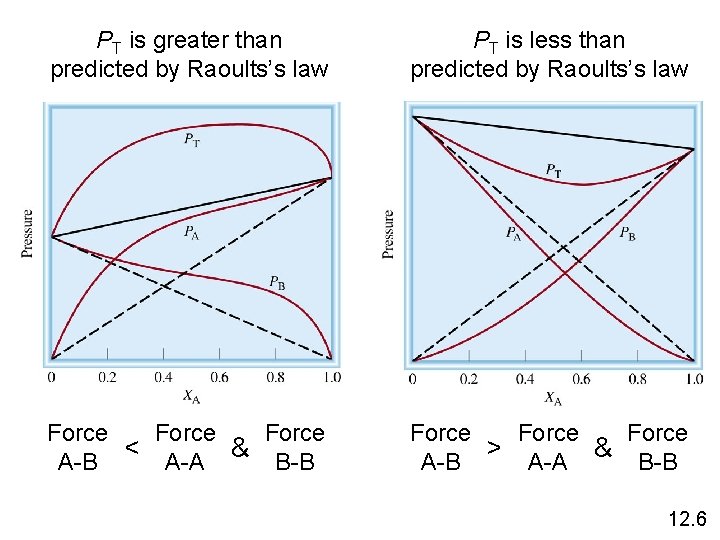
PT is greater than predicted by Raoults’s law PT is less than predicted by Raoults’s law Force < A-A & B-B A-B Force > A-A & B-B A-B 12. 6

Worked Example 12. 7

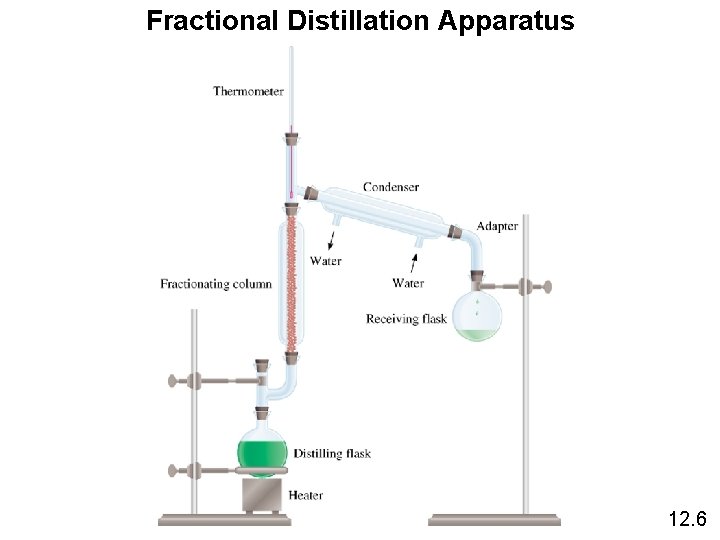
Fractional Distillation Apparatus 12. 6

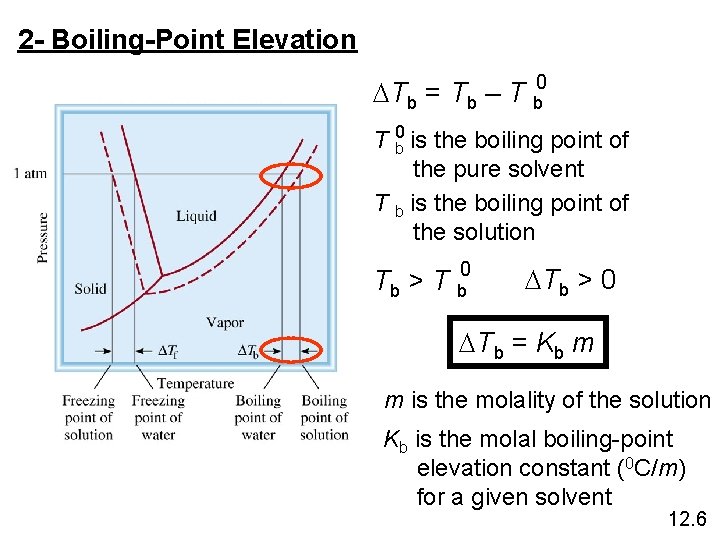
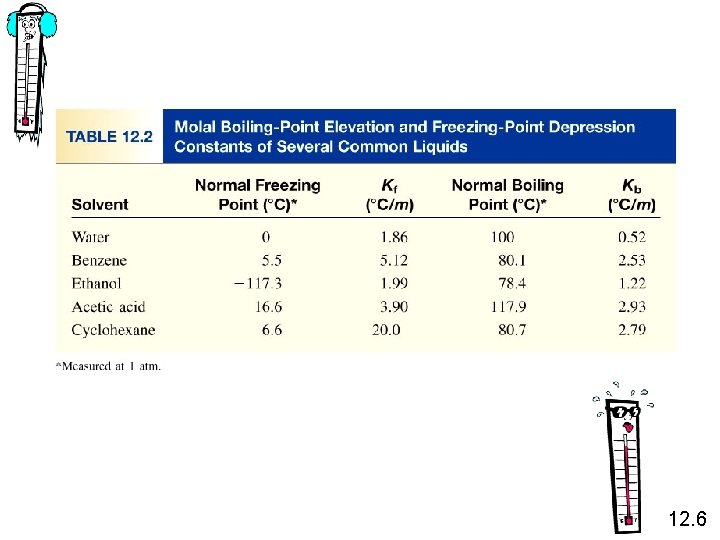
2 - Boiling-Point Elevation DTb = Tb – T b 0 is the boiling point of the pure solvent T b is the boiling point of the solution Tb > T b 0 DTb > 0 DTb = Kb m m is the molality of the solution Kb is the molal boiling-point elevation constant (0 C/m) for a given solvent 12. 6

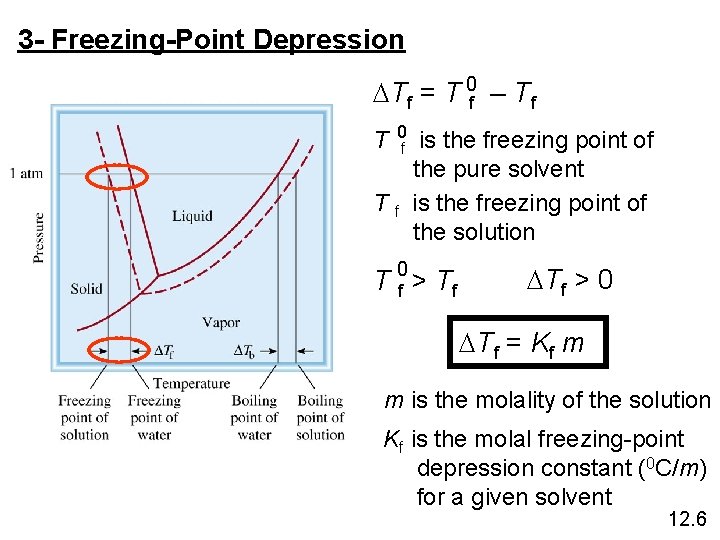
3 - Freezing-Point Depression DTf = T 0 f – Tf T 0 f is the freezing point of the pure solvent T f is the freezing point of the solution T 0 f > Tf DTf > 0 DTf = Kf m m is the molality of the solution Kf is the molal freezing-point depression constant (0 C/m) for a given solvent 12. 6

12. 6

What is the freezing point of a solution containing 478 g of ethylene glycol (antifreeze) in 3202 g of water? The molar mass of ethylene glycol is 62. 01 g. DTf = Kf m m = Kf water = 1. 86 0 C/m moles of solute mass of solvent (kg) 478 g x = 1 mol 62. 01 g 3. 202 kg solvent = 2. 41 m DTf = Kf m = 1. 86 0 C/m x 2. 41 m = 4. 48 0 C DTf = T 0 f – Tf Tf = T 0 f – DTf = 0. 00 0 C – 4. 48 0 C = -4. 48 0 C 12. 6

Worked Example 12. 8

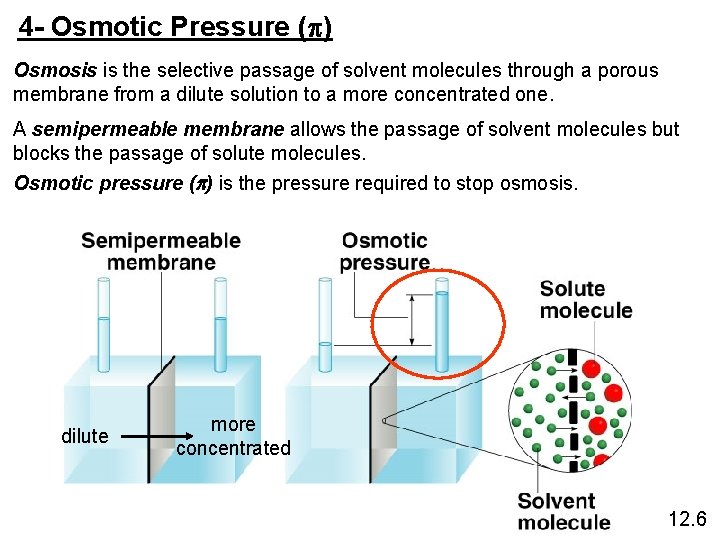
4 - Osmotic Pressure (p) Osmosis is the selective passage of solvent molecules through a porous membrane from a dilute solution to a more concentrated one. A semipermeable membrane allows the passage of solvent molecules but blocks the passage of solute molecules. Osmotic pressure (p) is the pressure required to stop osmosis. dilute more concentrated 12. 6

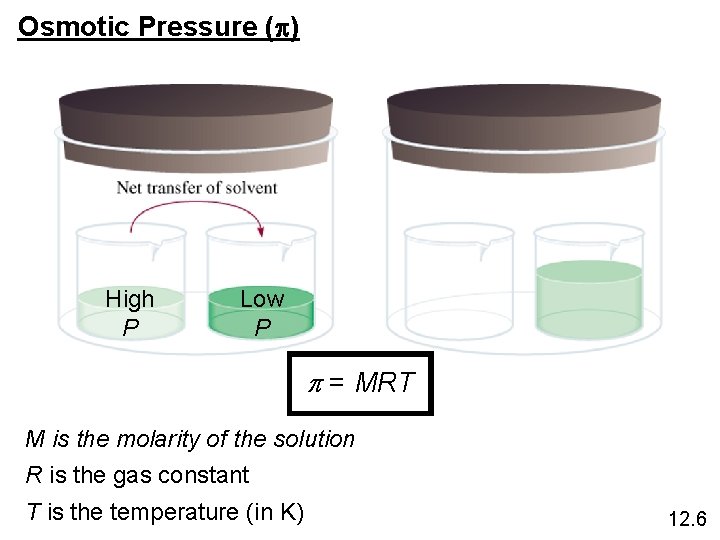
Osmotic Pressure (p) High P Low P p = MRT M is the molarity of the solution R is the gas constant T is the temperature (in K) 12. 6

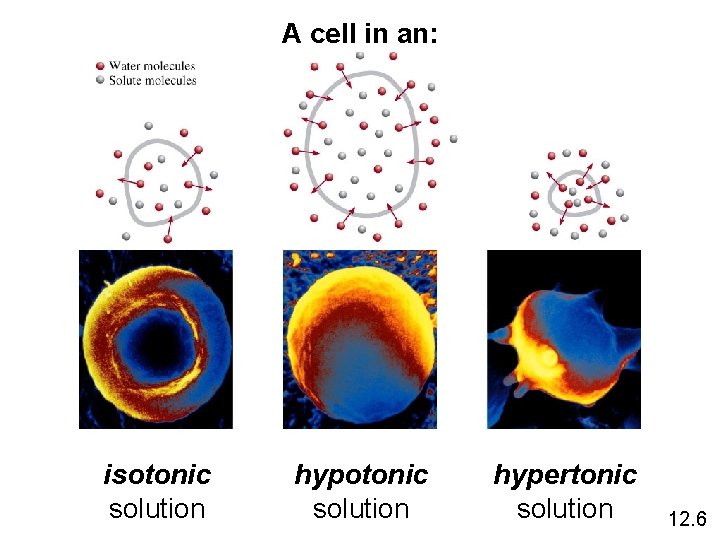
A cell in an: isotonic solution hypertonic solution 12. 6

Worked Example 12. 9

Colligative Properties of Nonelectrolyte Solutions Colligative properties are properties that depend only on the number of solute particles in solution and not on the nature of the solute particles. Vapor-Pressure Lowering P 1 = X 1 P 10 Boiling-Point Elevation DTb = Kb m Freezing-Point Depression DTf = Kf m Osmotic Pressure (p) p = MRT 12. 6

12 -7 Colligative Properties of Electrolyte Solutions 0. 1 m Na. Cl solution 0. 1 m Na+ ions & 0. 1 m Cl- ions Colligative properties are properties that depend only on the number of solute particles in solution and not on the nature of the solute particles. 0. 1 m Na. Cl solution van’t Hoff factor (i) = 0. 2 m ions in solution actual number of particles in soln after dissociation number of formula units initially dissolved in soln i should be nonelectrolytes Na. Cl Ca. Cl 2 1 2 3 12. 7

Colligative Properties of Electrolyte Solutions Boiling-Point Elevation DTb = i Kb m Freezing-Point Depression DTf = i Kf m Osmotic Pressure (p) p = i. MRT 12. 7

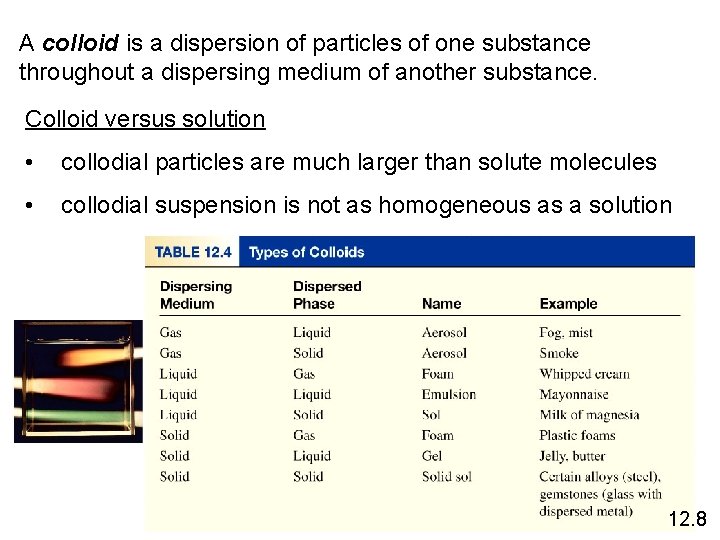
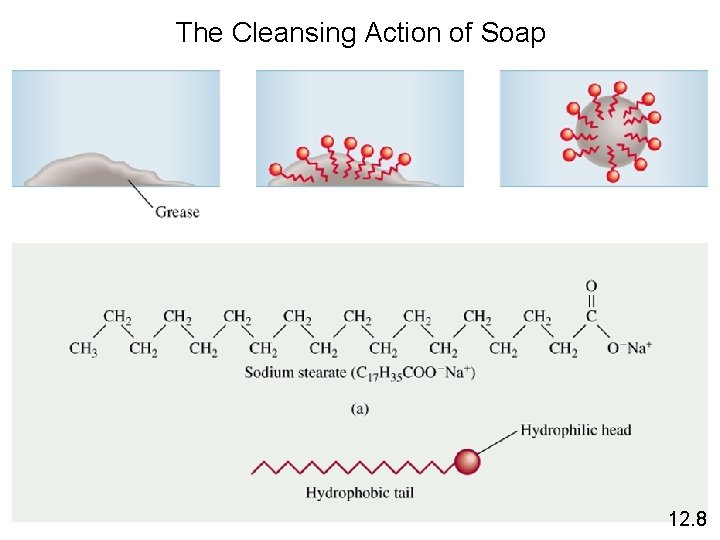
A colloid is a dispersion of particles of one substance throughout a dispersing medium of another substance. Colloid versus solution • collodial particles are much larger than solute molecules • collodial suspension is not as homogeneous as a solution 12. 8

The Cleansing Action of Soap 12. 8

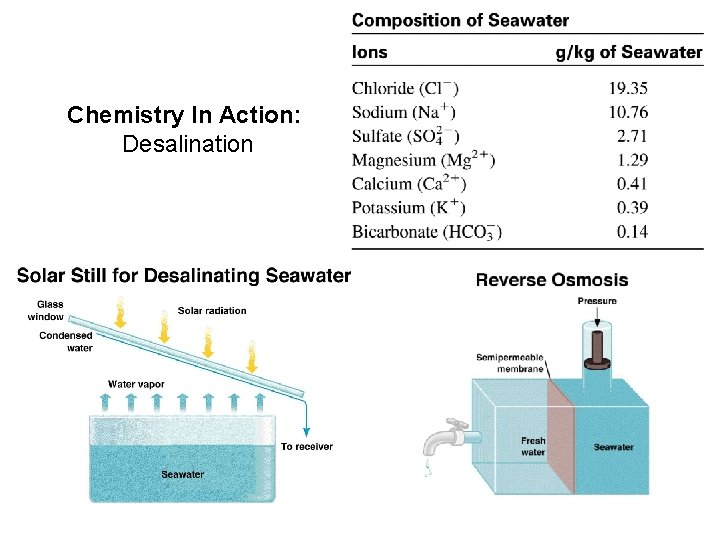
Chemistry In Action: Desalination
