Physical Properties of Solids Melting PointSublimation Point Hardness

Physical Properties of Solids Melting Point/Sublimation Point Hardness Malleable or brittle Solubility Heating curves Polarity Electrical Conductivity of solid Conductivity in solution

Ionic Solids High melting point Hard but brittle Strong electrostatic attraction forces between particles Non-conductors as solids, most conduct when molten and when dissolved Most are soluble Examples: Na. Cl, Mg. O, Na. OH


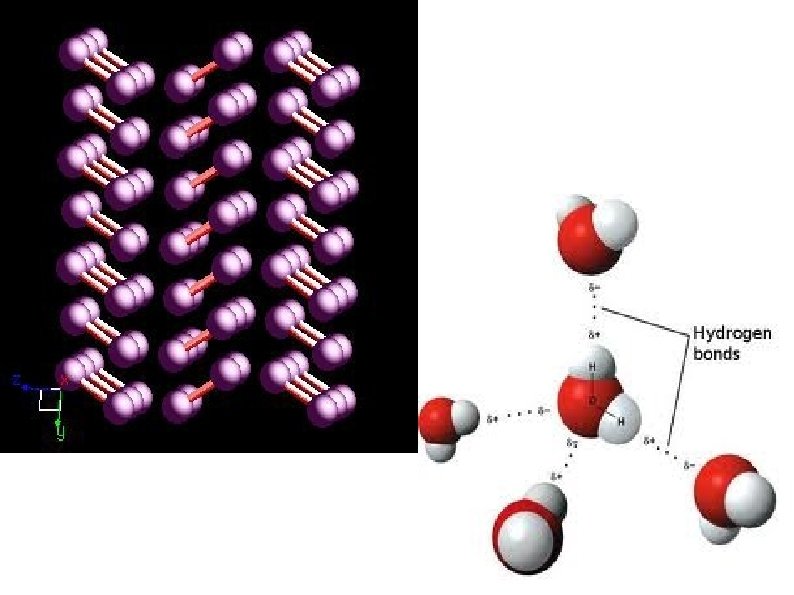
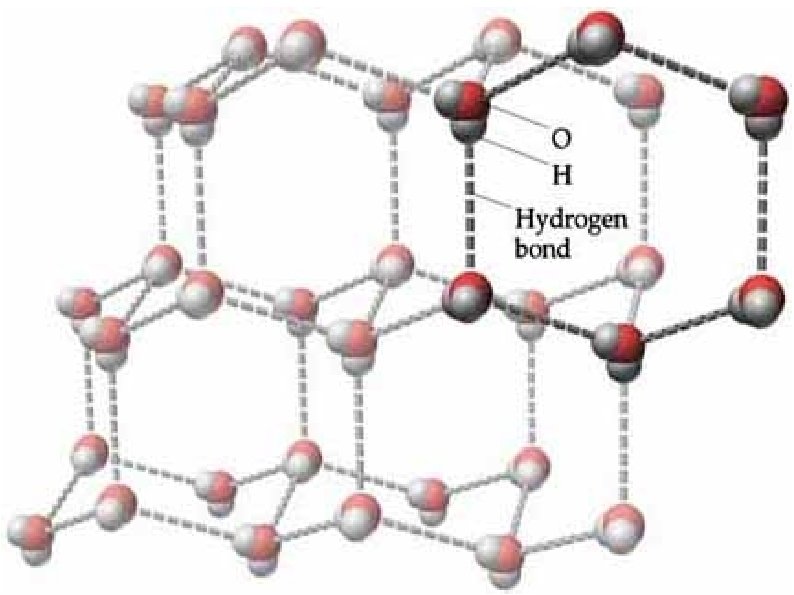
Molecular Solids Low melting point Soft Weak Van der Waals Forces or Hydrogen bonds Non-conductors May be soluble Examples: I 2 H 2 O Sulfur CO 2

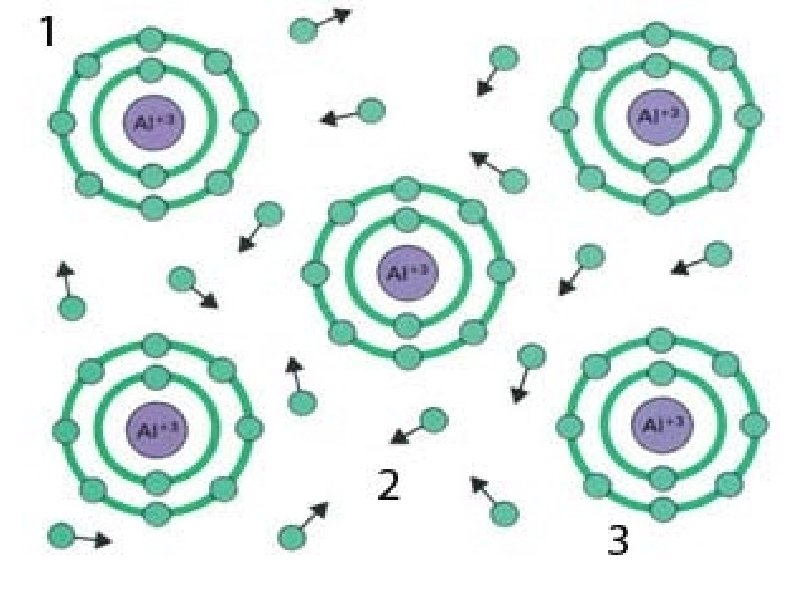
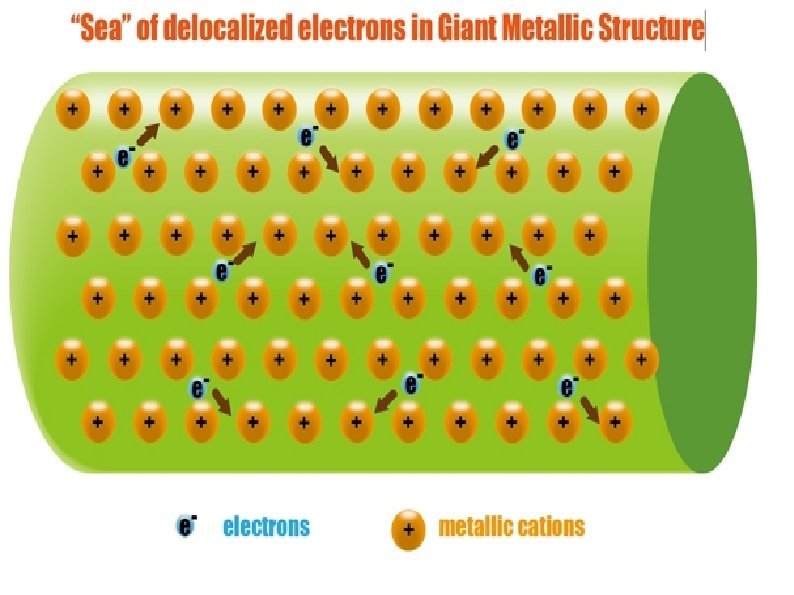
Metallic Solids Moderate to high melting point Hardness varies from soft to hard Malleable and ductile Cations and delocalized electrons form metallic bonds Conductors as solid and molten (liquid) Examples: Na, Cu, Al, Zn, Fe, Ag

Network Solids High to very high melting or sublimation point Most are hard to very hard Strong covalent bond network Most are non-conductors and insoluable Examples: C (graphite), C (diamond), Si. O 2


Type of Solid Ionic Molecular Metallic Network Type of structural particle Type of Bond Melting Point Hardness Electrical conductivity Solubility Examples
- Slides: 12