Physical Properties Boiling Point Physical Properties Boiling Point


Physical Properties: Boiling Point

Physical Properties Boiling Point – the temperature at which a substance turns from a liquid to a gas

Physical Properties Boiling Point When a liquid becomes a gas it is called boiling or vaporization. What happens? At a certain temperature called the boiling point, the molecules will gain enough energy to break free and become a gas. This Photo by Unknown Author is licensed under CC BY The boiling point for water is 100 degrees C (212 degrees F).

Physical Properties: Melting Point
Physical Properties Melting Point – the temperature at which a substance turns from a solid into a liquid

The temperature at which a solid turns into a liquid is called the melting point. Physical Properties As the energy in the molecules increases from a rise in temperature, the molecules start moving faster. Eventually, they have enough energy to break free of their rigid structure and start moving around more easily and the matter becomes a liquid. The melting point for water is 0 degrees C (32 degrees F).


Physical Properties: Freezing Point This Photo by Unknown Author is licensed under CC BY
Physical Properties Freezing Point – the temperature at which a substance turns from a liquid to a solid

The temperature at which a liquid turns into a solid is called the freezing point. Physical Properties As the energy in the molecules decreases from a fall in temperature, the molecules start moving slower. Eventually, they don’t have enough energy to move easily and they are confined to a rigid structure. The matter becomes a solid. The freezing point for water is 0 degrees C (32 degrees F).

Physical Properties What do you notice about the melting point and freezing point?
Physical Properties The temperature of a substances freezing point and the temperature of a substances melting point are the same temperature!
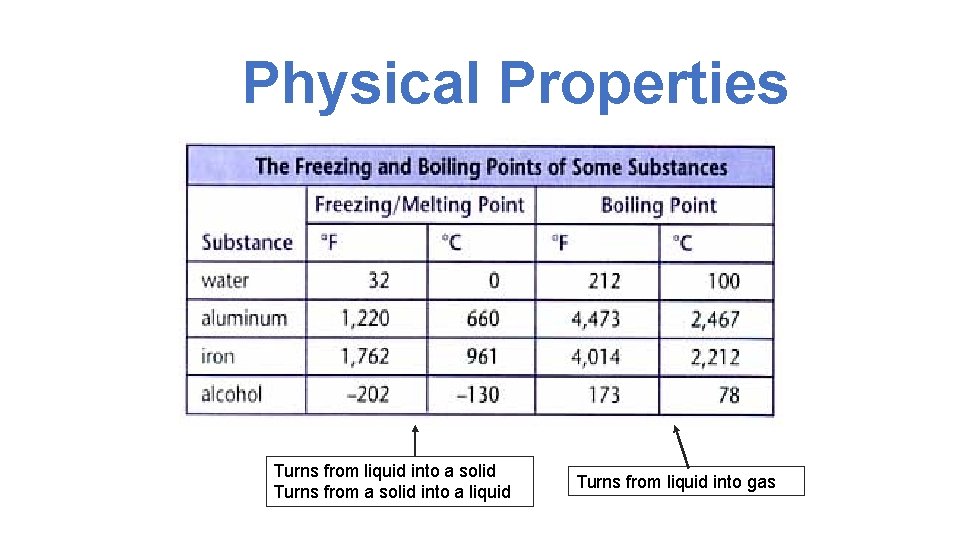
Physical Properties Turns from liquid into a solid Turns from a solid into a liquid Turns from liquid into gas

You Do! Answer the question in today’s Exit Ticket.
- Slides: 15