Physical Examination Physical exam BP 15192 P 97




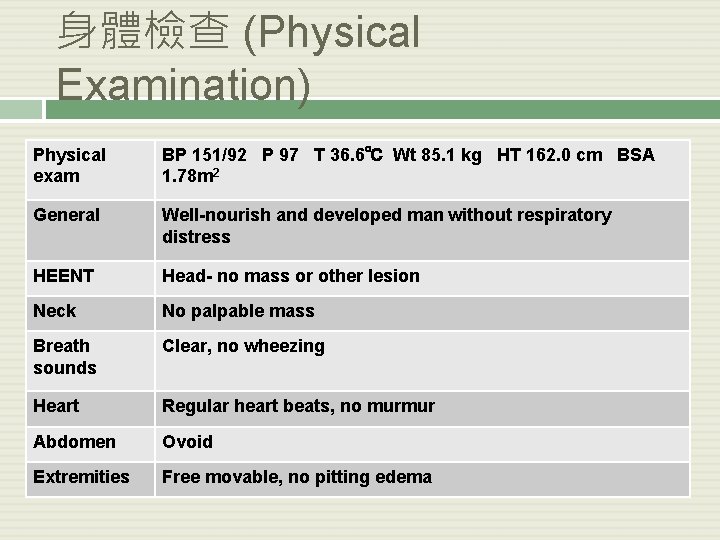
身體檢查 (Physical Examination) Physical exam BP 151/92 P 97 T 36. 6℃ Wt 85. 1 kg HT 162. 0 cm BSA 1. 78 m 2 General Well-nourish and developed man without respiratory distress HEENT Head- no mass or other lesion Neck No palpable mass Breath sounds Clear, no wheezing Heart Regular heart beats, no murmur Abdomen Ovoid Extremities Free movable, no pitting edema
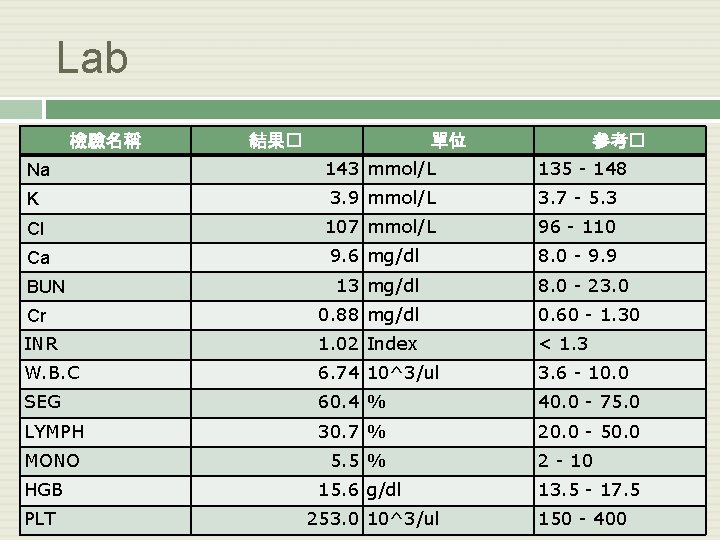
Lab 檢驗名稱 結果� 單位 參考� Na 143 mmol/L 135 - 148 K 3. 9 mmol/L 3. 7 - 5. 3 Cl 107 mmol/L 96 - 110 Ca 9. 6 mg/dl 8. 0 - 9. 9 BUN 13 mg/dl 8. 0 - 23. 0 Cr 0. 88 mg/dl 0. 60 - 1. 30 INR 1. 02 Index < 1. 3 W. B. C 6. 74 10^3/ul 3. 6 - 10. 0 SEG 60. 4 % 40. 0 - 75. 0 LYMPH 30. 7 % 20. 0 - 50. 0 MONO 5. 5 % HGB PLT 15. 6 g/dl 253. 0 10^3/ul 2 - 10 13. 5 - 17. 5 150 - 400
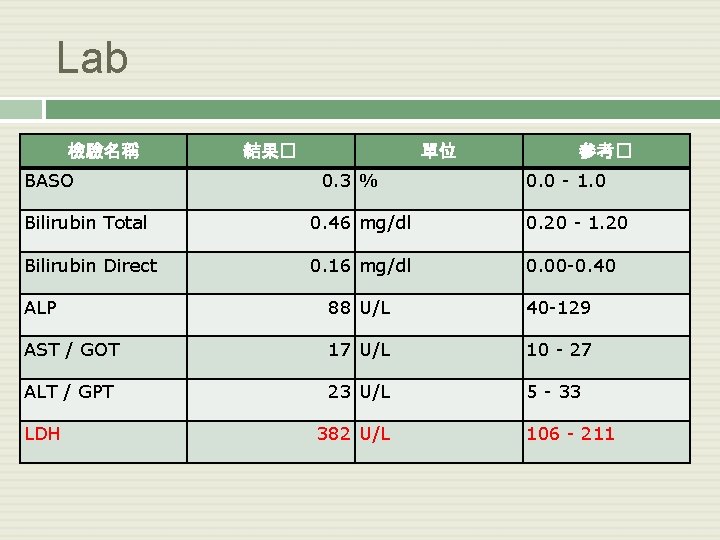
Lab 檢驗名稱 BASO 結果� 單位 0. 3 % 參考� 0. 0 - 1. 0 Bilirubin Total 0. 46 mg/dl 0. 20 - 1. 20 Bilirubin Direct 0. 16 mg/dl 0. 00 -0. 40 ALP 88 U/L 40 -129 AST / GOT 17 U/L 10 - 27 ALT / GPT 23 U/L 5 - 33 LDH 382 U/L 106 - 211



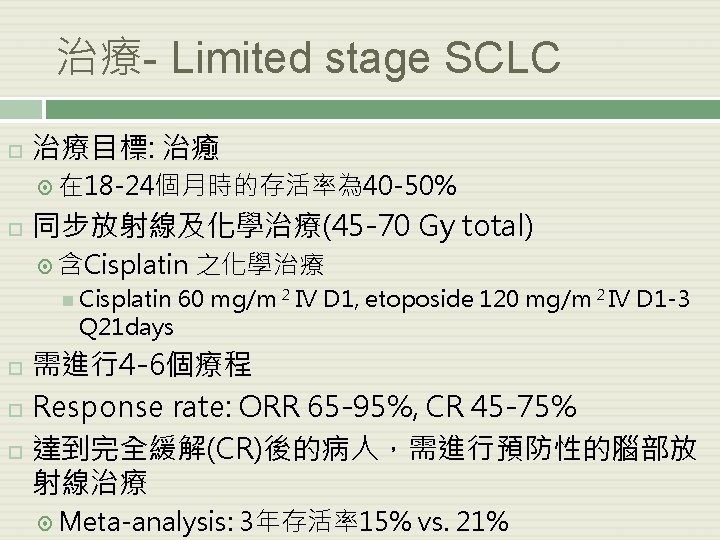
治療- Limited stage SCLC 治療目標: 治癒 在 18 -24個月時的存活率為 40 -50% 同步放射線及化學治療(45 -70 Gy total) 含Cisplatin Q 21 days 之化學治療 60 mg/m 2 IV D 1, etoposide 120 mg/m 2 IV D 1 -3 需進行4 -6個療程 Response rate: ORR 65 -95%, CR 45 -75% 達到完全緩解(CR)後的病人,需進行預防性的腦部放 射線治療 Meta-analysis: 3年存活率15% vs. 21%



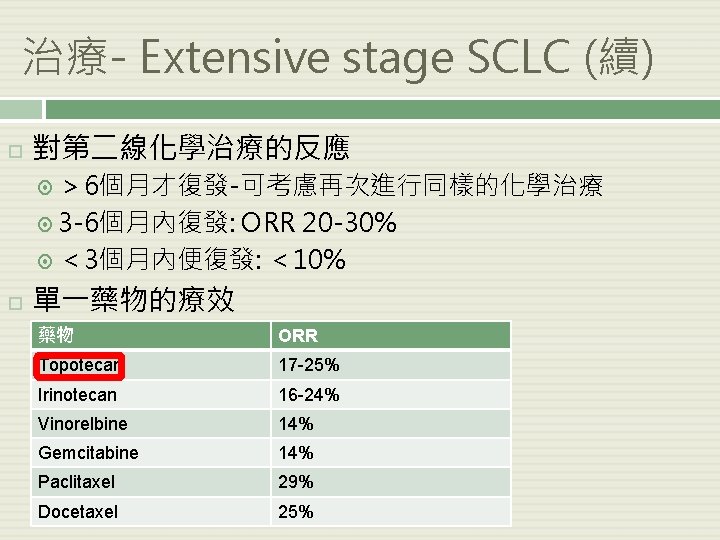
治療- Extensive stage SCLC (續) 對第二線化學治療的反應 > 6個月才復發-可考慮再次進行同樣的化學治療 3 -6個月內復發: ORR 20 -30% < 3個月內便復發: < 10% 單一藥物的療效 藥物 ORR Topotecan 17 -25% Irinotecan 16 -24% Vinorelbine 14% Gemcitabine 14% Paclitaxel 29% Docetaxel 25%

第二線化學治療-topotecan Topotecan 1. 5 mg/m 2 IV daily *5 days, Q 21 days = Topotecan 2. 3 mg/m 2 PO daily *5 days, Q 21 days 常見副作用: 白血球數下降、中性球數下降、血小板減少、 貧血、噁心、嘔吐、掉髮 依腎功能調整劑量: Clcr> 50 ml/min : 75% Clcr=10 -50 ml/min : 50% Clcr< 10 ml/min : 25%

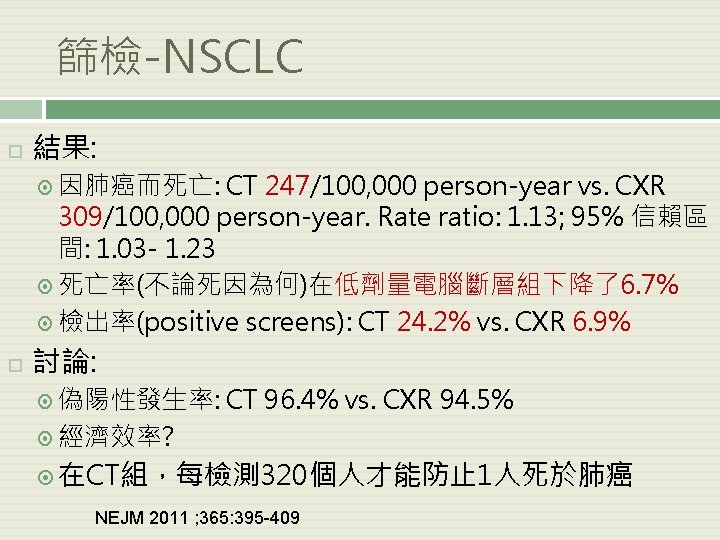
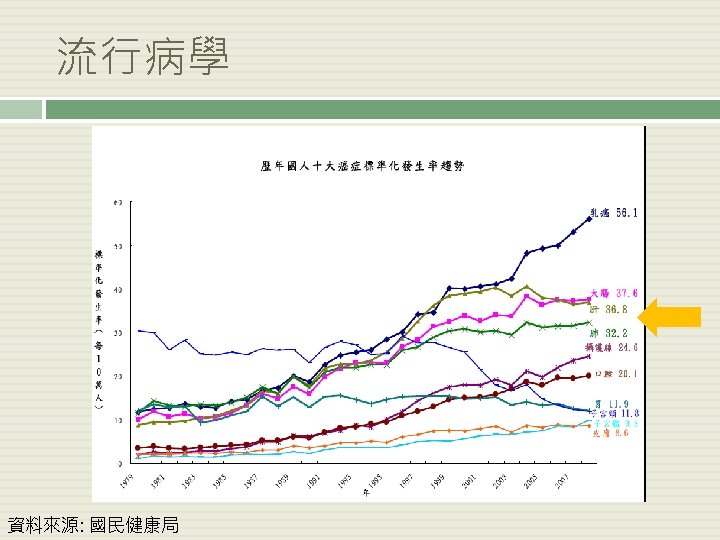
篩檢-NSCLC 結果: 因肺癌而死亡: CT 247/100, 000 person-year vs. CXR 309/100, 000 person-year. Rate ratio: 1. 13; 95% 信賴區 間: 1. 03 - 1. 23 死亡率(不論死因為何)在低劑量電腦斷層組下降了6. 7% 檢出率(positive screens): CT 24. 2% vs. CXR 6. 9% 討論: 偽陽性發生率: CT 96. 4% vs. CXR 94. 5% 經濟效率? 在CT組,每檢測 320個人才能防止 1人死於肺癌 NEJM 2011 ; 365: 395 -409
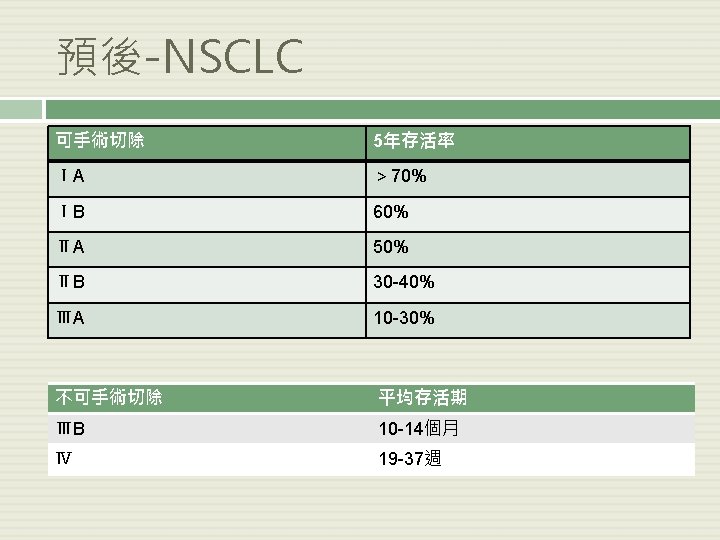
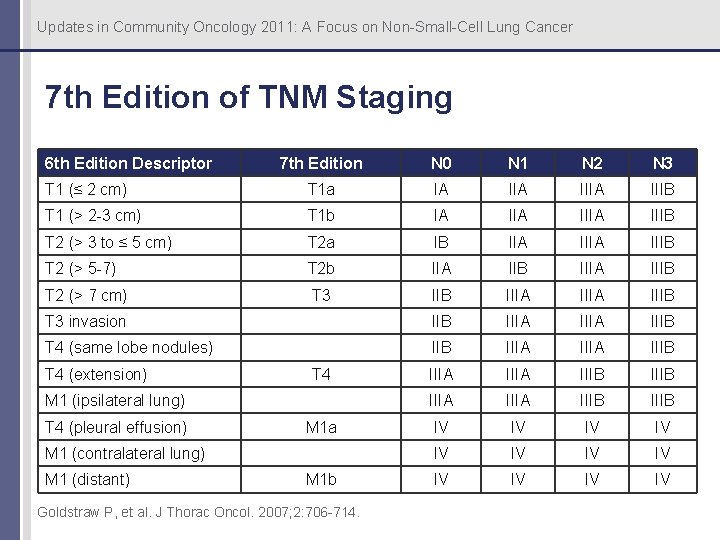
Updates in Community Oncology 2011: A Focus on Non-Small-Cell Lung Cancer 7 th Edition of TNM Staging 6 th Edition Descriptor 7 th Edition N 0 N 1 N 2 N 3 T 1 (≤ 2 cm) T 1 a IA IIIA IIIB T 1 (> 2 -3 cm) T 1 b IA IIIA IIIB T 2 (> 3 to ≤ 5 cm) T 2 a IB IIA IIIB T 2 (> 5 -7) T 2 b IIA IIB IIIA IIIB T 2 (> 7 cm) T 3 IIB IIIA IIIB T 3 invasion IIB IIIA IIIB T 4 (same lobe nodules) IIB IIIA IIIB IIIA IIIB IV IV IV IV T 4 (extension) T 4 M 1 (ipsilateral lung) T 4 (pleural effusion) M 1 a M 1 (contralateral lung) M 1 (distant) M 1 b Goldstraw P, et al. J Thorac Oncol. 2007; 2: 706 -714.
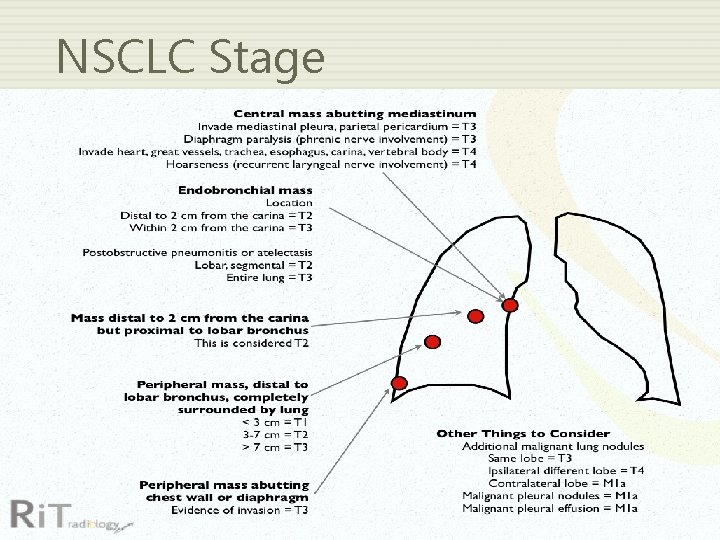
NSCLC Stage

輔助性化療-NSCLC Stage Ⅱ-ⅢA: Cisplatin 為主的化學治療, 可增加 5年存活率 Vinorelbine + cisplatin 的組合在研究論文中最常被使用 4 cycles 存活危險比(survival hazard Ratio): 有接受輔助性化療: 沒接受輔助性化療=0. 69 (0. 52 -0. 91) Grade toxicity ¾=33%-75% Age consideration 65 -75: no difference VS <65 >75, few studies J Clin Oncol 2007; 25: 5506 -5518
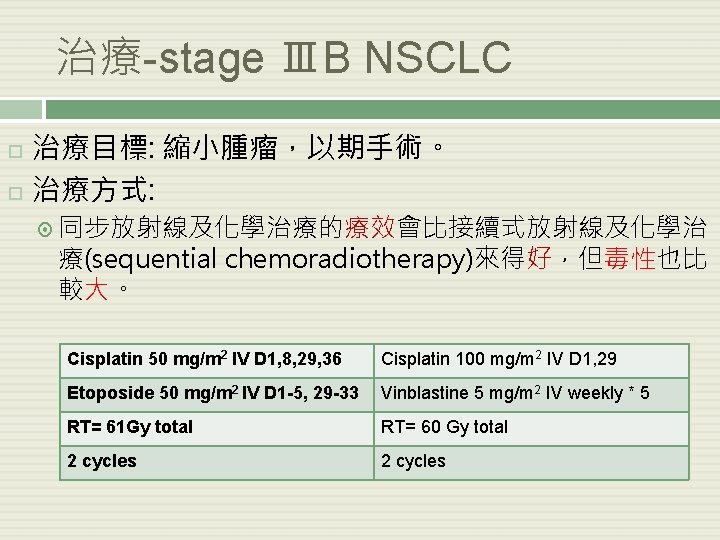
治療-stage ⅢB NSCLC 治療目標: 縮小腫瘤,以期手術。 治療方式: 同步放射線及化學治療的療效會比接續式放射線及化學治 療(sequential chemoradiotherapy)來得好,但毒性也比 較大。 Cisplatin 50 mg/m 2 IV D 1, 8, 29, 36 Cisplatin 100 mg/m 2 IV D 1, 29 Etoposide 50 mg/m 2 IV D 1 -5, 29 -33 Vinblastine 5 mg/m 2 IV weekly * 5 RT= 61 Gy total RT= 60 Gy total 2 cycles

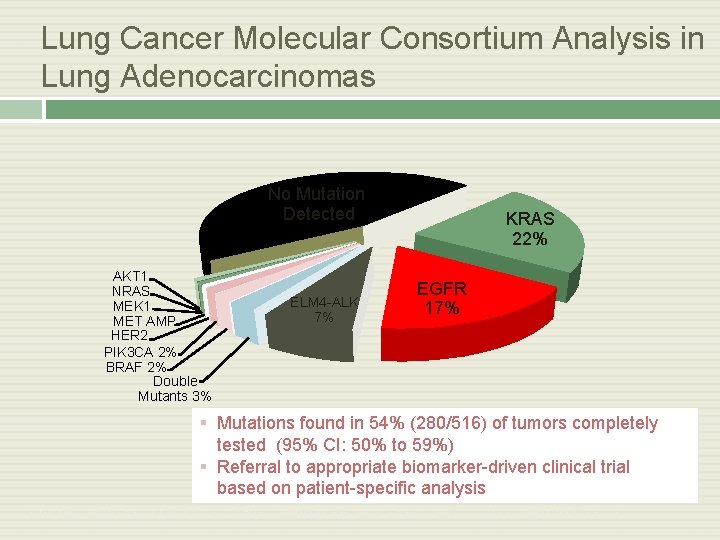
Lung Cancer Molecular Consortium Analysis in Lung Adenocarcinomas No Mutation Detected AKT 1 NRAS MEK 1 MET AMP HER 2 PIK 3 CA 2% BRAF 2% Double Mutants 3% ELM 4 -ALK 7% KRAS 22% EGFR 17% § Mutations found in 54% (280/516) of tumors completely tested (95% CI: 50% to 59%) § Referral to appropriate biomarker-driven clinical trial based on patient-specific analysis Kris MG, et al. ASCO 2011. CRA 7506. Johnson BE, et al. IASLC WCLC 2011. Abstract O 16. 01
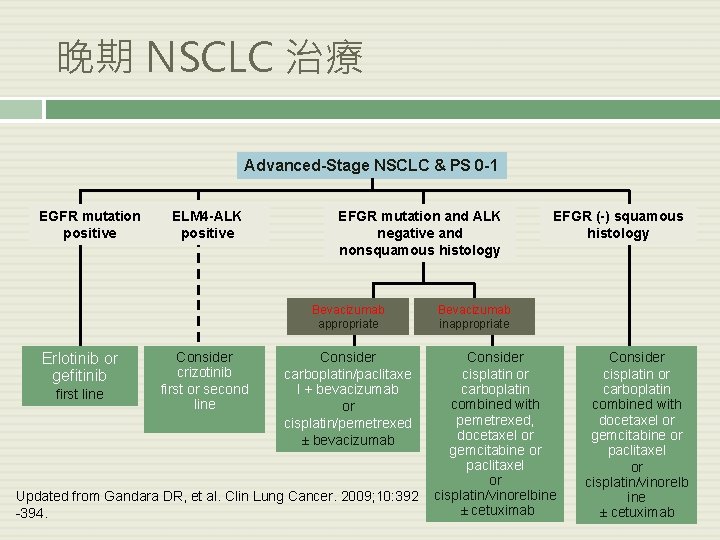
晚期 NSCLC 治療 Advanced-Stage NSCLC & PS 0 -1 EGFR mutation positive ELM 4 -ALK positive EFGR mutation and ALK negative and nonsquamous histology Bevacizumab appropriate Erlotinib or gefitinib first line Consider crizotinib first or second line Consider carboplatin/paclitaxe l + bevacizumab or cisplatin/pemetrexed ± bevacizumab Updated from Gandara DR, et al. Clin Lung Cancer. 2009; 10: 392 -394. EFGR (-) squamous histology Bevacizumab inappropriate Consider cisplatin or carboplatin combined with pemetrexed, docetaxel or gemcitabine or paclitaxel or cisplatin/vinorelbine ± cetuximab Consider cisplatin or carboplatin combined with docetaxel or gemcitabine or paclitaxel or cisplatin/vinorelb ine ± cetuximab
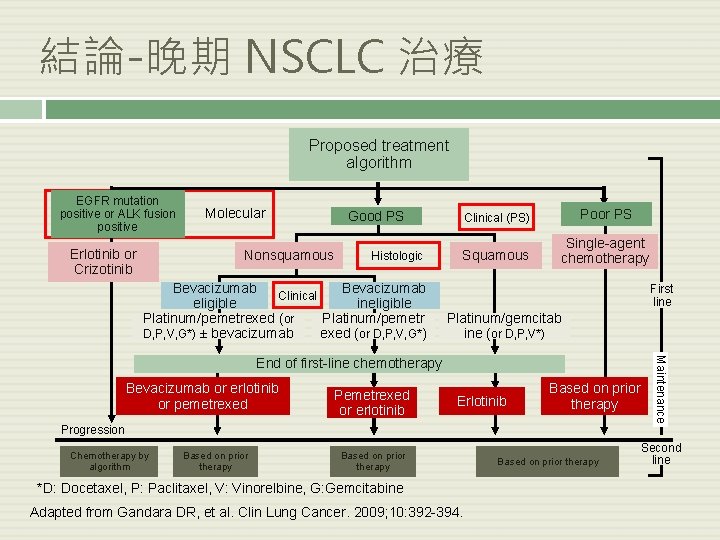
結論-晚期 NSCLC 治療 Proposed treatment algorithm EGFR mutation positive or ALK fusion positive Erlotinib or Crizotinib Molecular Nonsquamous Clinical (PS) Poor PS Squamous Single-agent chemotherapy Good PS Histologic Bevacizumab Clinical eligible ineligible Platinum/pemetrexed (or Platinum/pemetr D, P, V, G*) ± bevacizumab exed (or D, P, V, G*) First line Platinum/gemcitab ine (or D, P, V*) Bevacizumab or erlotinib or pemetrexed Pemetrexed or erlotinib Erlotinib Based on prior therapy Progression Chemotherapy by algorithm Based on prior therapy *D: Docetaxel, P: Paclitaxel, V: Vinorelbine, G: Gemcitabine Adapted from Gandara DR, et al. Clin Lung Cancer. 2009; 10: 392 -394. Based on prior therapy Maintenance End of first-line chemotherapy Second line


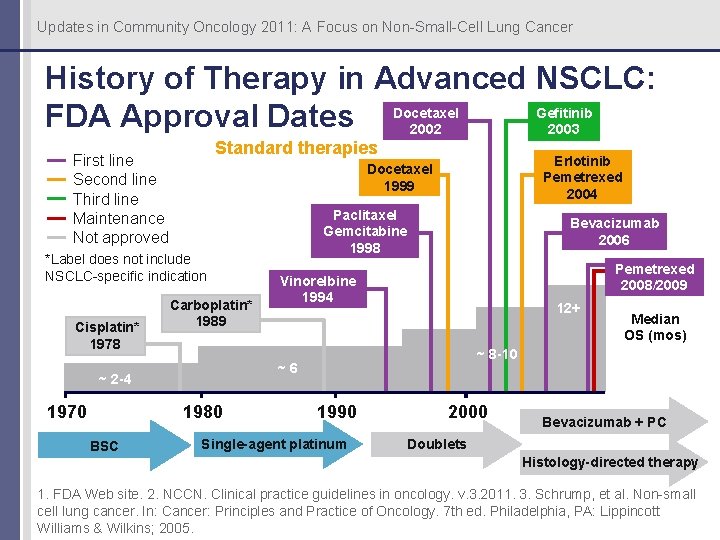
Updates in Community Oncology 2011: A Focus on Non-Small-Cell Lung Cancer History of Therapy in Advanced NSCLC: Gefitinib FDA Approval Dates Docetaxel 2003 2002 Standard therapies First line Second line Third line Maintenance Not approved *Label does not include NSCLC-specific indication Cisplatin* 1978 Carboplatin* 1989 1980 BSC Paclitaxel Gemcitabine 1998 Bevacizumab 2006 Pemetrexed 2008/2009 Vinorelbine 1994 12+ Median OS (mos) ~ 8 -10 ~6 ~ 2 -4 1970 Erlotinib Pemetrexed 2004 Docetaxel 1999 1990 Single-agent platinum 2000 Bevacizumab + PC Doublets Histology-directed therapy 1. FDA Web site. 2. NCCN. Clinical practice guidelines in oncology. v. 3. 2011. 3. Schrump, et al. Non-small cell lung cancer. In: Cancer: Principles and Practice of Oncology. 7 th ed. Philadelphia, PA: Lippincott Williams & Wilkins; 2005.

Updates in Community Oncology 2011: A Focus on Non-Small-Cell Lung Cancer Agents With Activity in Advanced NSCLC Older Agents* Newer Agents* § § § § § Carboplatin Cisplatin Etoposide Ifosfamide Mitomycin C Vinblastine Vindesine *Not all drugs listed have FDA approval. Docetaxel Gemcitabine Irinotecan Paclitaxel Topotecan Vinorelbine Pemetrexed Gefitinib Erlotinib Bevacizumab Cetuximab Crizotinib

第一 線化療-晚期 NSCLC 含白金類的化療組合為首選 Overall response rate (ORR): 25 -35% Time to progression (TTP): 4 -6個月 平均存活期: 8 -10個月 一年存活率: 30 -40% NCCN practice guideline: Non-small cell lung cancer v. 2012
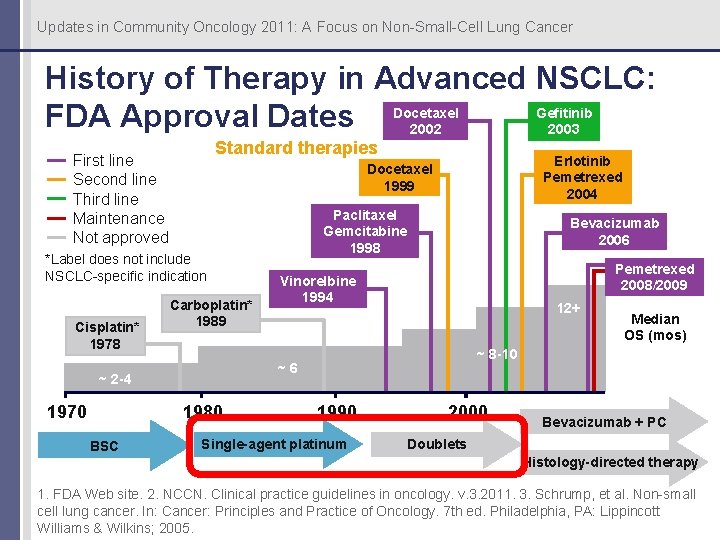
Updates in Community Oncology 2011: A Focus on Non-Small-Cell Lung Cancer History of Therapy in Advanced NSCLC: Gefitinib FDA Approval Dates Docetaxel 2003 2002 Standard therapies First line Second line Third line Maintenance Not approved *Label does not include NSCLC-specific indication Cisplatin* 1978 Carboplatin* 1989 1980 BSC Paclitaxel Gemcitabine 1998 Bevacizumab 2006 Pemetrexed 2008/2009 Vinorelbine 1994 12+ Median OS (mos) ~ 8 -10 ~6 ~ 2 -4 1970 Erlotinib Pemetrexed 2004 Docetaxel 1999 1990 Single-agent platinum 2000 Bevacizumab + PC Doublets Histology-directed therapy 1. FDA Web site. 2. NCCN. Clinical practice guidelines in oncology. v. 3. 2011. 3. Schrump, et al. Non-small cell lung cancer. In: Cancer: Principles and Practice of Oncology. 7 th ed. Philadelphia, PA: Lippincott Williams & Wilkins; 2005.

Updates in Community Oncology 2011: A Focus on Non-Small-Cell Lung Cancer First-line Treatment: 2 Agents Are More Effective Than 1 § Meta-analysis: 65 trials (N = 13, 601) between 1980 -2001 – Compared efficacy of – Doublet vs single-agent regimens – Triplet vs doublet regimens Survival Outcome Doublet vs Single-Agent Regimens Triplet vs Doublet Regimens 1 -yr OS Doublet > single-agent § OR: 0. 80; 95% CI: 0. 70 -0. 91; P <. 001 § 5% absolute benefit Triplet = doublet § OR: 1. 01; 95% CI: 0. 85 -1. 21; P =. 88 Median OS Doublet > single-agent § MR: 0. 83; 95% CI: 0. 79 -0. 89; P <. 001 Triplet = doublet § MR: 1. 00; 95% CI: 0. 94 -1. 06; P =. 97 Delbaldo C, et al. JAMA. 2004; 292: 470 -484.

臨床試驗 ECOG 1594: 4組白金類化療組合比較 ECOG 4599: Carbo/Pac 該不該加 Bevacizumab? JMDB: Cis + Pemetrexed vs. Cis + Gemcitabine
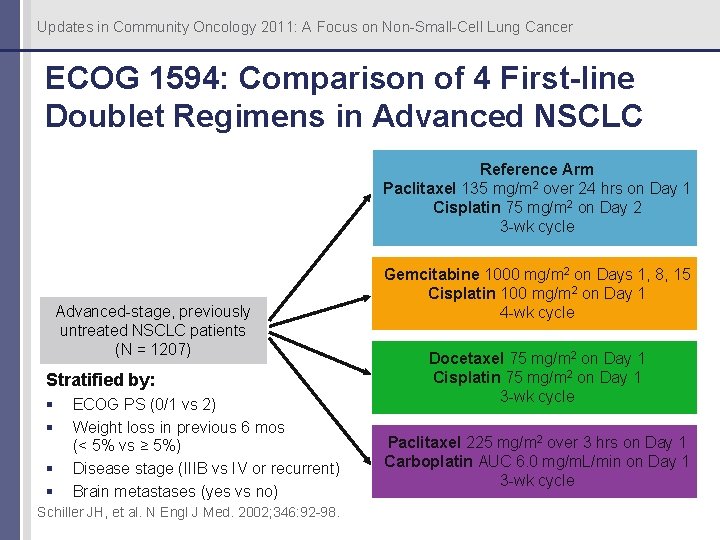
Updates in Community Oncology 2011: A Focus on Non-Small-Cell Lung Cancer ECOG 1594: Comparison of 4 First-line Doublet Regimens in Advanced NSCLC Reference Arm Paclitaxel 135 mg/m 2 over 24 hrs on Day 1 Cisplatin 75 mg/m 2 on Day 2 3 -wk cycle Advanced-stage, previously untreated NSCLC patients (N = 1207) Stratified by: § § ECOG PS (0/1 vs 2) Weight loss in previous 6 mos (< 5% vs ≥ 5%) Disease stage (IIIB vs IV or recurrent) Brain metastases (yes vs no) Schiller JH, et al. N Engl J Med. 2002; 346: 92 -98. Gemcitabine 1000 mg/m 2 on Days 1, 8, 15 Cisplatin 100 mg/m 2 on Day 1 4 -wk cycle Docetaxel 75 mg/m 2 on Day 1 Cisplatin 75 mg/m 2 on Day 1 3 -wk cycle Paclitaxel 225 mg/m 2 over 3 hrs on Day 1 Carboplatin AUC 6. 0 mg/m. L/min on Day 1 3 -wk cycle

ECOG 1594 -platinum doublets
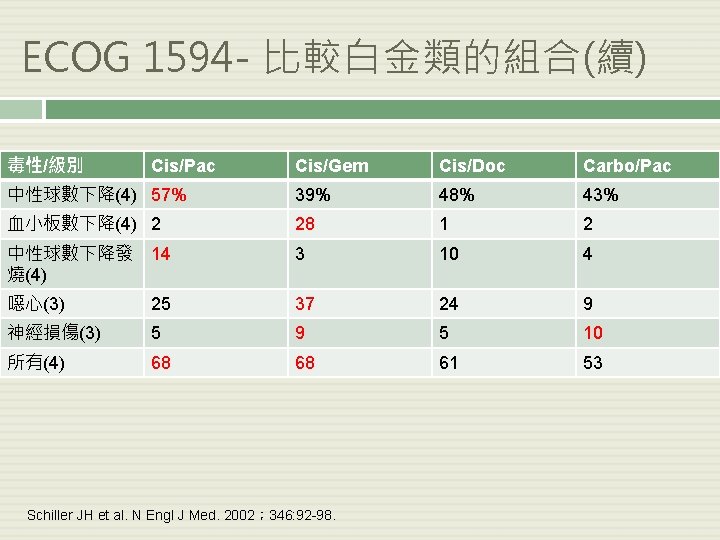
ECOG 1594 - 比較白金類的組合(續) 毒性/級別 Cis/Pac Cis/Gem Cis/Doc Carbo/Pac 中性球數下降(4) 57% 39% 48% 43% 血小板數下降(4) 2 28 1 2 中性球數下降發 14 燒(4) 3 10 4 噁心(3) 25 37 24 9 神經損傷(3) 5 9 5 10 所有(4) 68 68 61 53 Schiller JH et al. N Engl J Med. 2002; 346: 92 -98.


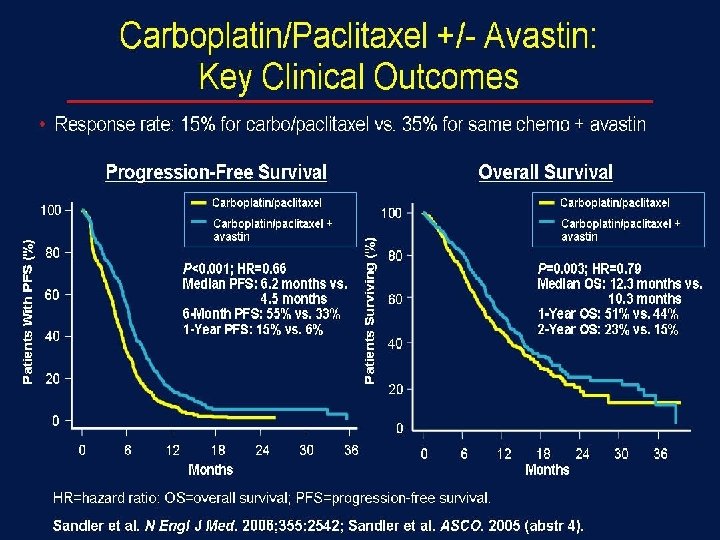

ECOG 4599 總結 符合以下條件者,使用Cis 可增加存活率: + Pac + Bevacizumab 非鱗狀上皮細胞癌之非小細胞肺癌病人 ECOG PS =0 -1 Sandler A, et al. N Engl J Med. 2006; 355: 2542 -2550.
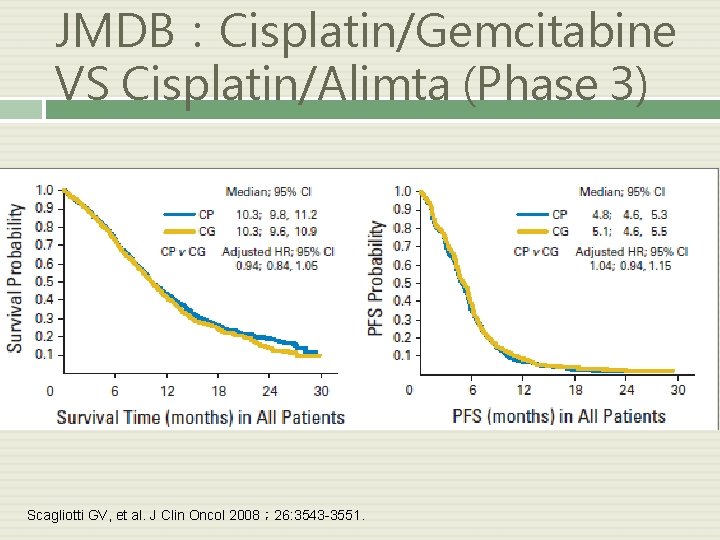
JMDB:Cisplatin/Gemcitabine VS Cisplatin/Alimta (Phase 3) Scagliotti GV, et al. J Clin Oncol 2008; 26: 3543 -3551.
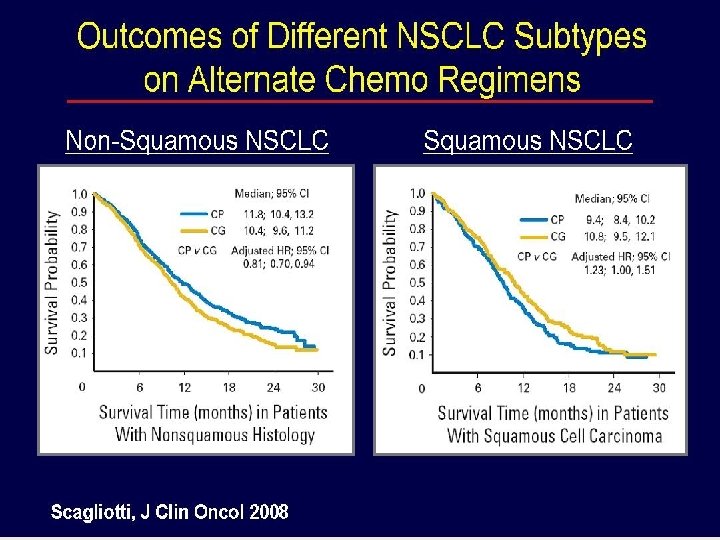
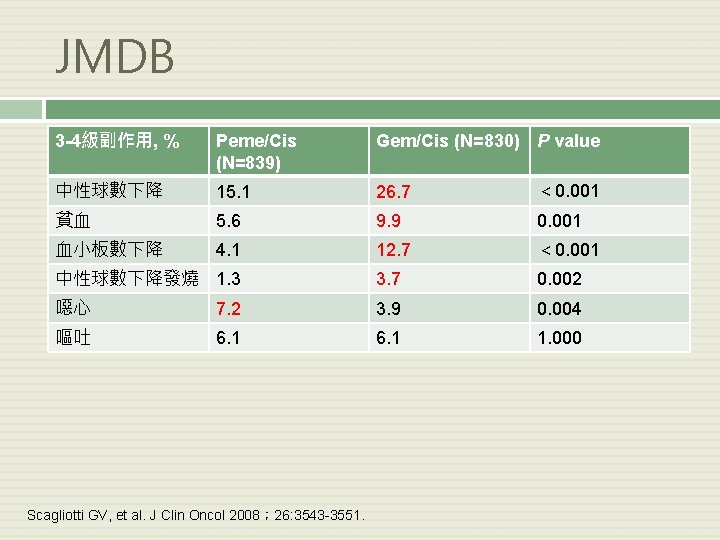
JMDB 3 -4級副作用, % Peme/Cis (N=839) Gem/Cis (N=830) P value 中性球數下降 15. 1 26. 7 < 0. 001 貧血 5. 6 9. 9 0. 001 血小板數下降 4. 1 12. 7 < 0. 001 中性球數下降發燒 1. 3 3. 7 0. 002 噁心 7. 2 3. 9 0. 004 嘔吐 6. 1 1. 000 Scagliotti GV, et al. J Clin Oncol 2008; 26: 3543 -3551.

JMDB 總結 整體存活率: Cis + Peme = Cis + Gem 鱗狀上皮細胞癌: : Cis + Peme < Cis + Gem 非鱗狀上皮細胞癌: : Cis + Peme > Cis + Gem 毒性: Cis + Peme < Cis + Gem Scagliotti GV, et al. J Clin Oncol 2008; 26: 3543 -3551.

Updates in Community Oncology 2011: A Focus on Non-Small-Cell Lung Cancer Chemotherapy Today and the Need for Targeted Therapies § Doublet chemotherapy for 4 -6 cycles is standard § Select chemotherapy based on histology § Future selection by other markers (ie, ERCC 1) § There is a need for “targeted” chemotherapy and other agents – Antiangiogenesis: VEGF targeted (bevacizumab, etc) – EGFR-targeted antibody (cetuximab), TKI (erlotinib, etc) – Newer targets (ALK and others) – Recent identification of “driver mutations” in 50% of NSCLC adenocarcinomas

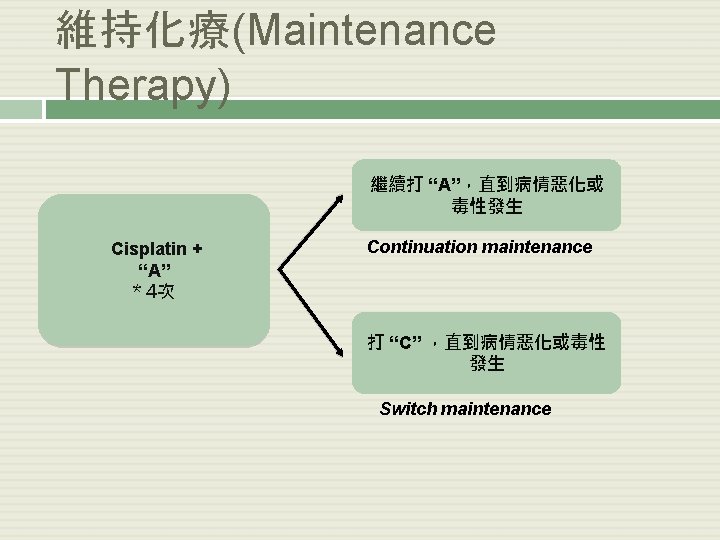
維持化療(Maintenance Therapy) 繼續打 “A”,直到病情惡化或 毒性發生 Cisplatin + “A” * 4次 Continuation maintenance 打 “C” ,直到病情惡化或毒性 發生 Switch maintenance
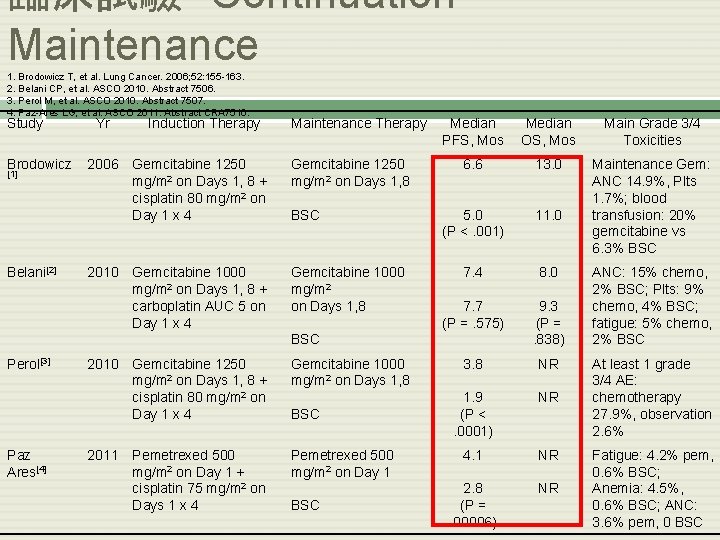
臨床試驗- Continuation Maintenance 1. Brodowicz T, et al. Lung Cancer. 2006; 52: 155 -163. 2. Belani CP, et al. ASCO 2010. Abstract 7506. 3. Perol M, et al. ASCO 2010. Abstract 7507. 4. Paz-Ares LG, et al. ASCO 2011. Abstract CRA 7510. Study Brodowicz [1] Belani[2] Yr Induction Therapy Maintenance Therapy 2006 Gemcitabine 1250 mg/m 2 on Days 1, 8 + cisplatin 80 mg/m 2 on Day 1 x 4 Gemcitabine 1250 mg/m 2 on Days 1, 8 2010 Gemcitabine 1000 mg/m 2 on Days 1, 8 + carboplatin AUC 5 on Day 1 x 4 Gemcitabine 1000 mg/m 2 on Days 1, 8 BSC Median PFS, Mos Median OS, Mos Main Grade 3/4 Toxicities 6. 6 13. 0 5. 0 (P <. 001) 11. 0 Maintenance Gem: ANC 14. 9%, Plts 1. 7%; blood transfusion: 20% gemcitabine vs 6. 3% BSC 7. 4 8. 0 7. 7 (P =. 575) 9. 3 (P =. 838) 3. 8 NR 1. 9 (P <. 0001) NR 4. 1 NR 2. 8 (P =. 00006) NR BSC Perol[3] Paz Ares[4] 2010 Gemcitabine 1250 mg/m 2 on Days 1, 8 + cisplatin 80 mg/m 2 on Day 1 x 4 Gemcitabine 1000 mg/m 2 on Days 1, 8 2011 Pemetrexed 500 mg/m 2 on Day 1 + cisplatin 75 mg/m 2 on Days 1 x 4 Pemetrexed 500 mg/m 2 on Day 1 BSC ANC: 15% chemo, 2% BSC; Plts: 9% chemo, 4% BSC; fatigue: 5% chemo, 2% BSC At least 1 grade 3/4 AE: chemotherapy 27. 9%, observation 2. 6% Fatigue: 4. 2% pem, 0. 6% BSC; Anemia: 4. 5%, 0. 6% BSC; ANC: 3. 6% pem, 0 BSC
![臨床試驗-Switch Maintenance Agent/Control Arm N PFS Salvage Treatment, % OS Fidias[1] Docetaxel Delayed docetaxel 臨床試驗-Switch Maintenance Agent/Control Arm N PFS Salvage Treatment, % OS Fidias[1] Docetaxel Delayed docetaxel](http://slidetodoc.com/presentation_image/207fa670aae0c1fb81812606a359de6a/image-62.jpg)
臨床試驗-Switch Maintenance Agent/Control Arm N PFS Salvage Treatment, % OS Fidias[1] Docetaxel Delayed docetaxel 309 5. 7 mos HR: 0. 63 2. 7 mos P =. 001 63 12. 3 9. 7 HR: 0. 80 P =. 085 Ciuleanu[2] Pemetrexed Placebo 663 4. 0 mos HR: 0. 50 2. 6 mos P <. 0001 67 13. 4 10. 6 HR: 0. 79 P =. 012 Cappuzzo[3] Erlotinib Placebo 889 12. 3 wks HR: 0. 71 11. 1 wks P <. 0001 72 12. 0 11. 0 HR: 0. 81 P =. 0088 Miller[4] Erlotinib + bevacizumab Placebo + bevacizumab 768 4. 8 mos HR: 0. 72 3. 8 mos P =. 001 55. 5 15. 9 13. 9 HR: 0. 90 P =. 2686 Perol[5] Erlotinib Observation 310 2. 9 mos HR: 0. 82 1. 9 mos P =. 002 81. 9 NA NA HR: . 91 Zhang[6] Gefitinib Placebo 296 4. 8 mos HR: 0. 42 2. 6 mos P <. 0001 58. 8 18. 7 16. 9 HR: . 84 P =. 2608 1. Fidias P, et al. J Clin Oncol. 2010; 28: 5116 -5123. 2. Ciuleanu T, et al. Lancet. 2009; 374: 1432 -1440. 3. Cappuzzo F, et al. Lancet Oncol. 2010; 11: 521 -529. 4. Miller VA, et al. ASCO 2009. Abstract LBA 8002. 5. Perol M, et al. ASCO 2010. Abstract 7507. 6. Zhang L, et al. ASCO 2011. Abstract LBA 7511.

總結-維持化療(Maintenance Therapy) 非鱗狀上皮細胞+EGFR (-) or 不明 Continuation maintenance : Bevacizumab (category 1) or cetuximab (category 1) Pemetrexed or gemcitabine Switch maintenance: Pemetrexed or erlotinib 鱗狀上皮細胞 Continuation Cetuximab Switch maintenance: (category 1) or gemcitabine mainyenance: Docetaxel (category 2 B) or erlotinib NCCN guidelines version 2. 2012 Non-small cell lung cancer


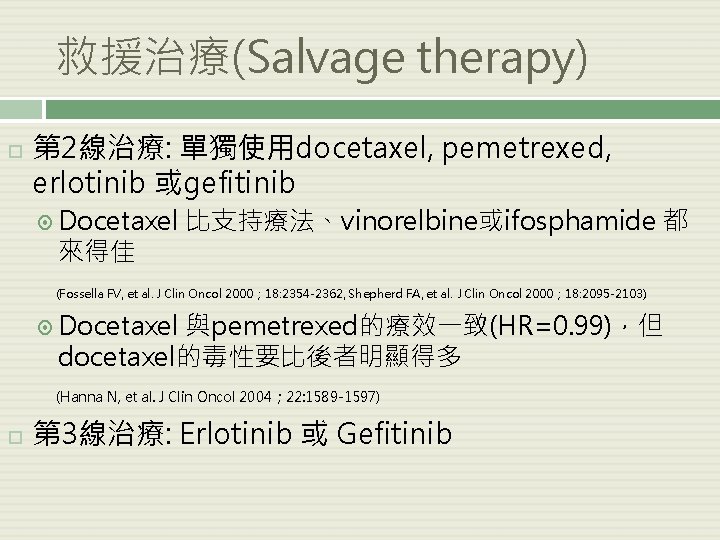
救援治療(Salvage therapy) 第 2線治療: 單獨使用docetaxel, pemetrexed, erlotinib 或gefitinib Docetaxel 來得佳 比支持療法、vinorelbine或ifosphamide 都 (Fossella FV, et al. J Clin Oncol 2000; 18: 2354 -2362, Shepherd FA, et al. J Clin Oncol 2000; 18: 2095 -2103) Docetaxel 與pemetrexed的療效一致(HR=0. 99),但 docetaxel的毒性要比後者明顯得多 (Hanna N, et al. J Clin Oncol 2004; 22: 1589 -1597) 第 3線治療: Erlotinib 或 Gefitinib
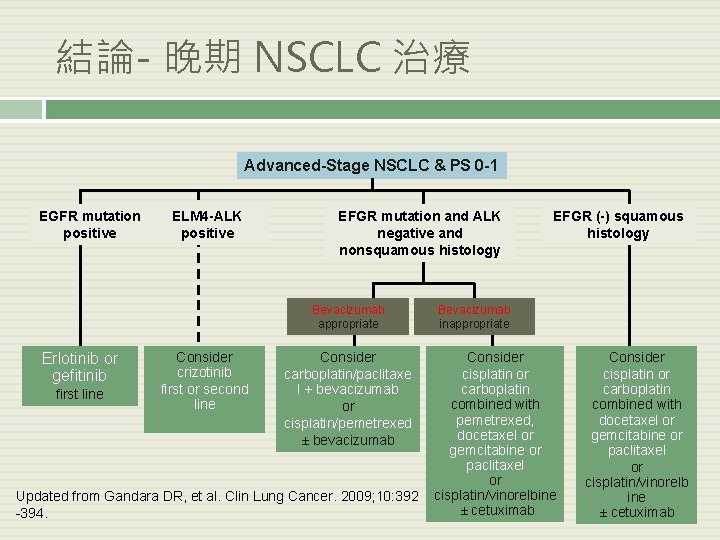
結論- 晚期 NSCLC 治療 Advanced-Stage NSCLC & PS 0 -1 EGFR mutation positive ELM 4 -ALK positive EFGR mutation and ALK negative and nonsquamous histology Bevacizumab appropriate Erlotinib or gefitinib first line Consider crizotinib first or second line Consider carboplatin/paclitaxe l + bevacizumab or cisplatin/pemetrexed ± bevacizumab Updated from Gandara DR, et al. Clin Lung Cancer. 2009; 10: 392 -394. EFGR (-) squamous histology Bevacizumab inappropriate Consider cisplatin or carboplatin combined with pemetrexed, docetaxel or gemcitabine or paclitaxel or cisplatin/vinorelbine ± cetuximab Consider cisplatin or carboplatin combined with docetaxel or gemcitabine or paclitaxel or cisplatin/vinorelb ine ± cetuximab
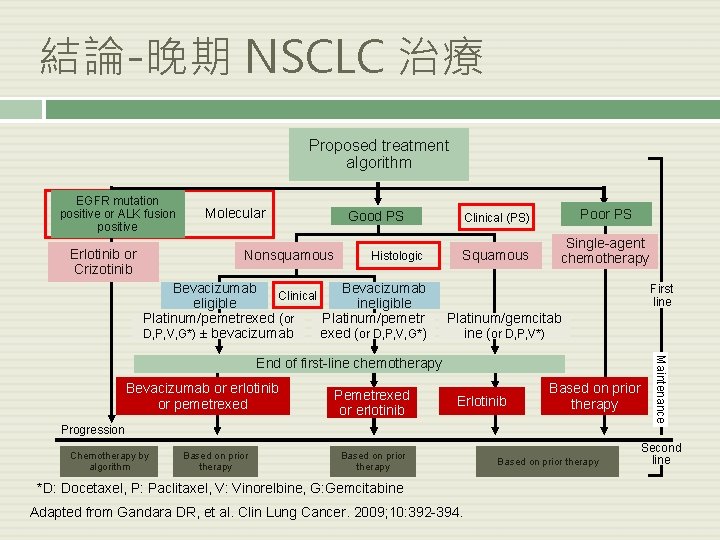
結論-晚期 NSCLC 治療 Proposed treatment algorithm EGFR mutation positive or ALK fusion positive Erlotinib or Crizotinib Molecular Nonsquamous Clinical (PS) Poor PS Squamous Single-agent chemotherapy Good PS Histologic Bevacizumab Clinical eligible ineligible Platinum/pemetrexed (or Platinum/pemetr D, P, V, G*) ± bevacizumab exed (or D, P, V, G*) First line Platinum/gemcitab ine (or D, P, V*) Bevacizumab or erlotinib or pemetrexed Pemetrexed or erlotinib Erlotinib Based on prior therapy Progression Chemotherapy by algorithm Based on prior therapy *D: Docetaxel, P: Paclitaxel, V: Vinorelbine, G: Gemcitabine Adapted from Gandara DR, et al. Clin Lung Cancer. 2009; 10: 392 -394. Based on prior therapy Maintenance End of first-line chemotherapy Second line

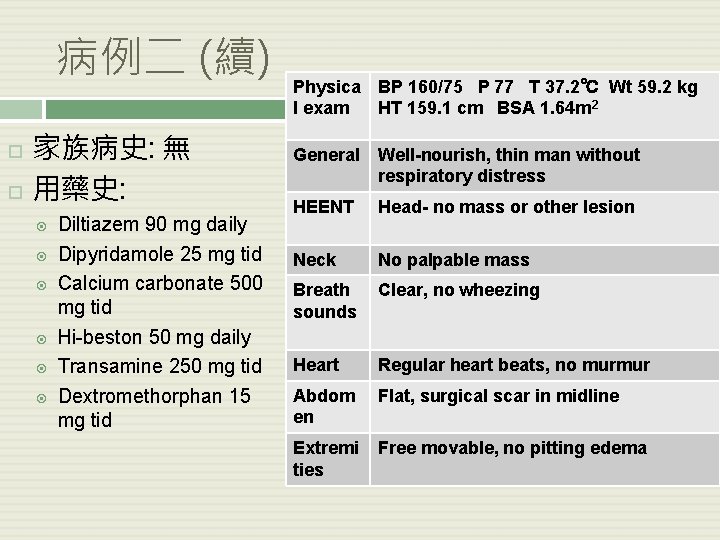
病例二 (續) 家族病史: 無 用藥史: Diltiazem 90 mg daily Dipyridamole 25 mg tid Calcium carbonate 500 mg tid Hi-beston 50 mg daily Transamine 250 mg tid Dextromethorphan 15 mg tid Physica BP 160/75 P 77 T 37. 2℃ Wt 59. 2 kg l exam HT 159. 1 cm BSA 1. 64 m 2 General Well-nourish, thin man without respiratory distress HEENT Head- no mass or other lesion Neck No palpable mass Breath sounds Clear, no wheezing Heart Regular heart beats, no murmur Abdom en Flat, surgical scar in midline Extremi ties Free movable, no pitting edema
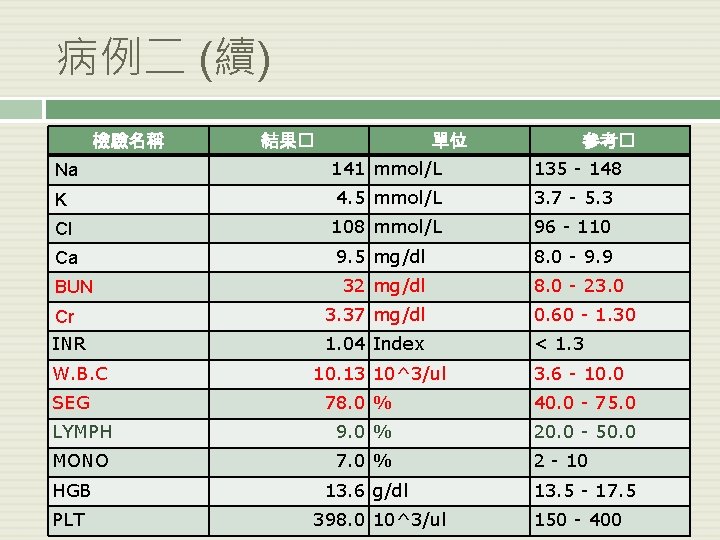
病例二 (續) 檢驗名稱 結果� 單位 參考� Na 141 mmol/L 135 - 148 K 4. 5 mmol/L 3. 7 - 5. 3 Cl 108 mmol/L 96 - 110 Ca 9. 5 mg/dl 8. 0 - 9. 9 BUN 32 mg/dl 8. 0 - 23. 0 Cr 3. 37 mg/dl 0. 60 - 1. 30 INR 1. 04 Index < 1. 3 W. B. C SEG 10. 13 10^3/ul 3. 6 - 10. 0 78. 0 % 40. 0 - 75. 0 LYMPH 9. 0 % 20. 0 - 50. 0 MONO 7. 0 % 2 - 10 HGB PLT 13. 6 g/dl 398. 0 10^3/ul 13. 5 - 17. 5 150 - 400
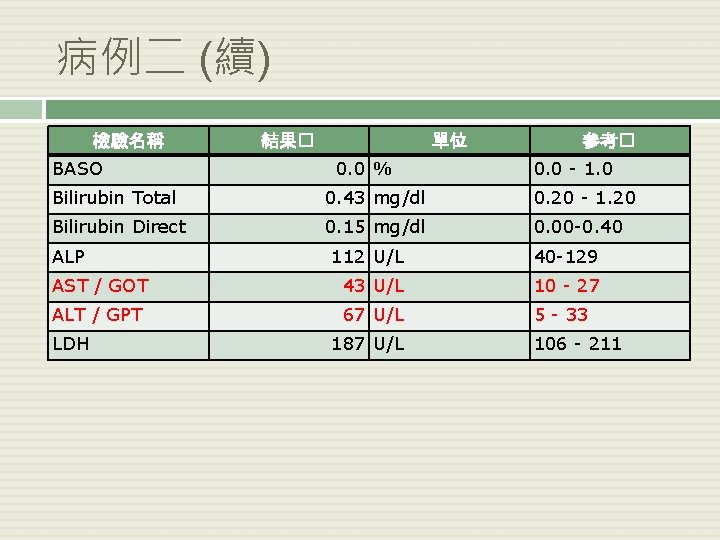
病例二 (續) 檢驗名稱 BASO 結果� 單位 0. 0 % 參考� 0. 0 - 1. 0 Bilirubin Total 0. 43 mg/dl 0. 20 - 1. 20 Bilirubin Direct 0. 15 mg/dl 0. 00 -0. 40 ALP 112 U/L 40 -129 AST / GOT 43 U/L 10 - 27 ALT / GPT 67 U/L 5 - 33 LDH 187 U/L 106 - 211


病例二- 治療 第 4期NSCLC 2011/6/14 CT顯示腫 瘤增大 2012/2/14 腦部MRI 發現有腦部轉移 • Docetaxel + carboplatin * 6次 (2010/12/27 -2011/4/18) • Docetaxel (2011/5/5 -2011/6/20) • Gefitinib (2011/7/4 -2012/2/27) • EGFR mutation positive • Erlotinib (2012/3/12 - 2012/8/9) • 緩和性腦部放射線治療 3600 c. Gy,分13次照射 (2012/2/16 - 2012/3/6)



練習題(續) 以下哪項化療方式最適合這位病人? Paclitaxel/carboplatin B. Bevacizumab/pemetrexed/carboplatin C. Gemcitabine/carboplatin 答案 : B A.





- Slides: 82