Physical Chemistry Table 1 A 1 Physical Chemistry

Physical Chemistry Table 1 A. 1 יחידות לחץ

Physical Chemistry Table 1 A. 2
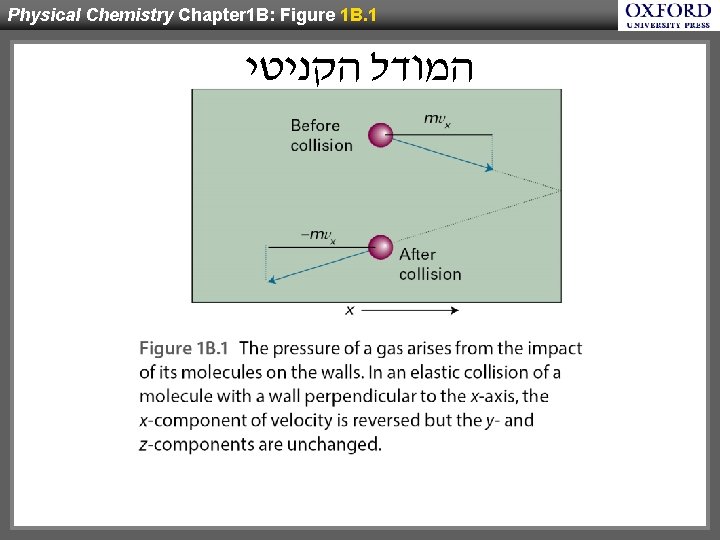
Physical Chemistry Chapter 1 B: Figure 1 B. 1 המודל הקניטי
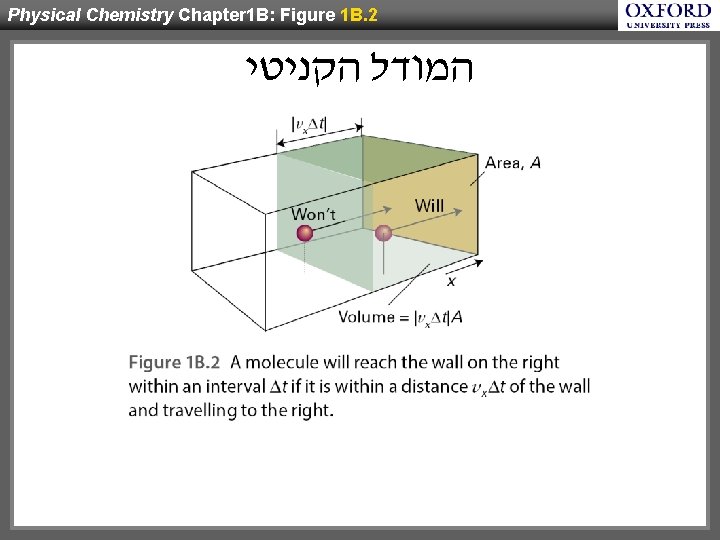
Physical Chemistry Chapter 1 B: Figure 1 B. 2 המודל הקניטי



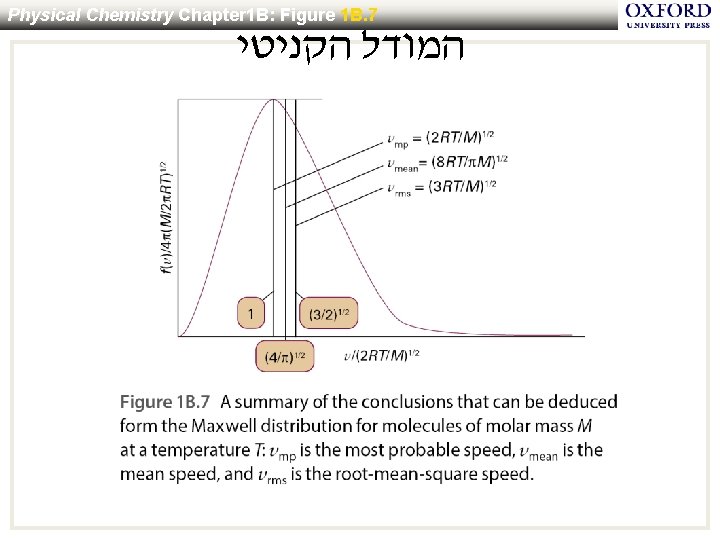
Physical Chemistry Chapter 1 B: Figure 1 B. 7 המודל הקניטי

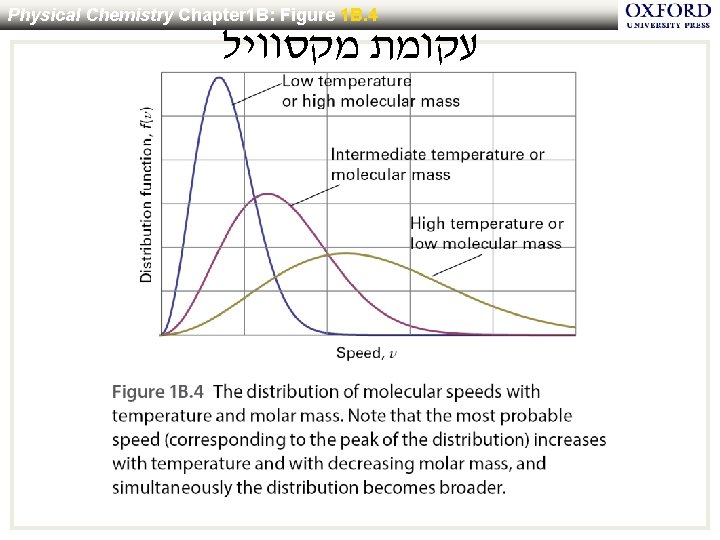
Physical Chemistry Chapter 1 B: Figure 1 B. 4 עקומת מקסוויל


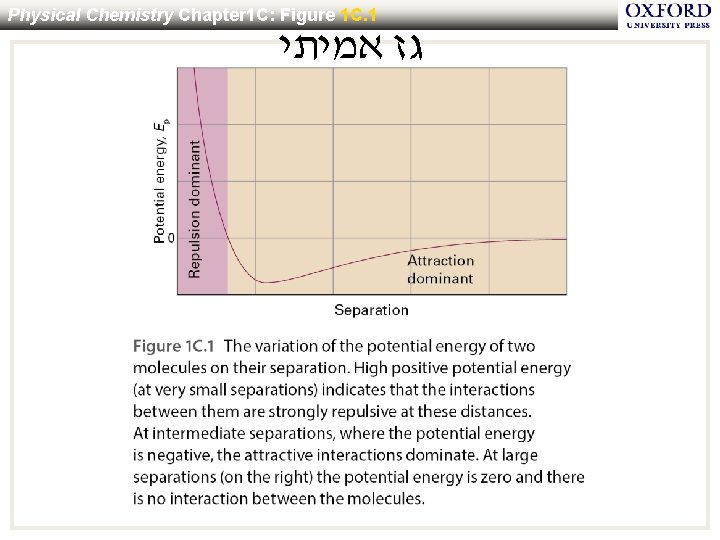
Physical Chemistry Chapter 1 C: Figure 1 C. 1 גז אמיתי

Physical Chemistry Chapter 1 C: Figure 1 C. 2 גז אמיתי
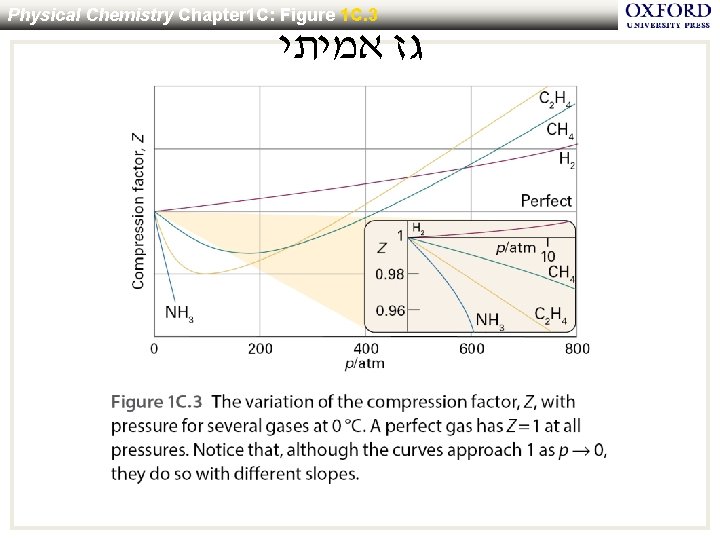
Physical Chemistry Chapter 1 C: Figure 1 C. 3 גז אמיתי
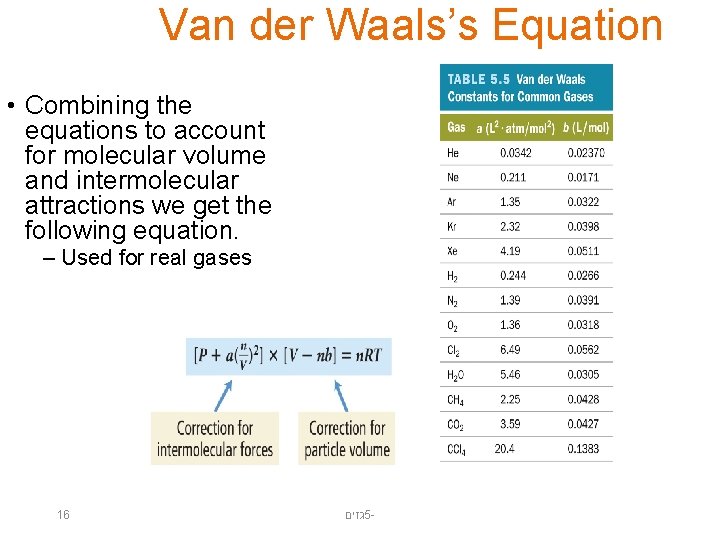
Van der Waals’s Equation • Combining the equations to account for molecular volume and intermolecular attractions we get the following equation. – Used for real gases 16 גזים 5 -

Real Gases • A plot of PV/RT versus P for 1 mole of a gas shows the difference between real and ideal gases. • It reveals a curve that shows the PV/RT ratio for a real gas is generally lower than ideal for “low” pressures—meaning that the most important factor is the intermolecular attractions. • It reveals a curve that shows the PV/RT ratio for a real gas is generally higher than ideal for “high” pressures—meaning that the most important factor is the molecular volume. 17 גזים 5 -
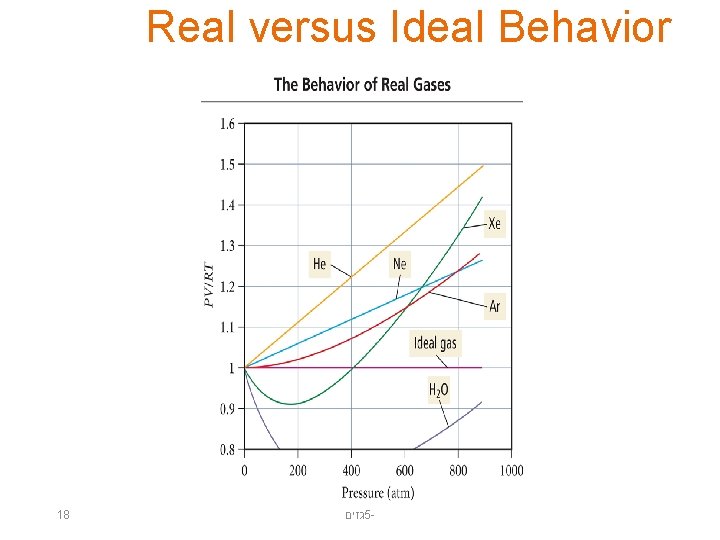
Real versus Ideal Behavior 18 גזים 5 -

Van der Waals’s Equation If SO 2 were an ideal gas, the pressure at 0. 0°C exerted by 1. 000 mole occupying 22. 41 L would be 1. 000 atm. Use the van der Waals equation to estimate the pressure of this volume of 1. 000 mol SO 2 at 0. 0°C. גזים 5 - © 2017 Cengage Learning. All Rights Reserved. May not be copied, scanned, or duplicated, in whole or in part, except for use as permitted in a license distributed with a certain product or service or otherwise 5 | 19

Van der Waals’s Equation Rearranging the van der Waal’s equation to give P Substituting values of R, T, V, a, and b כוחות משיכה גזים 5 - © 2017 Cengage Learning. All Rights Reserved. May not be copied, scanned, or duplicated, in whole or in part, except for use as permitted in a license distributed with a certain product or service or otherwise 5 | 20
- Slides: 20