Physical Chemistry Quantum Chemistry Quantum Mechanics Historical Background

Physical Chemistry Quantum Chemistry

Quantum Mechanics – Historical Background » Physics in the Late 19 th Century (prior to quantum mechanics (QM)) • Atoms are basic constituents of matter • Newton’s Laws apply universally • The world is deterministic » Physics was complete except for a few decimal places ! • Newtonian mechanics explained macroscopic behavior of matter --planetary motion, fluid flow, elasticity, etc. • Thermodynamics had its first two laws and most of their consequences • Basic statistical mechanics had been applied to chemical systems • Light was explained as an electromagnetic wave

» However there were several experiments that could not be explained by classical physics and the accepted dogma ! • • Blackbody radiation Photoelectric effect Discrete atomic spectra The electron as a subatomic particle » �Inescapable conclusions would result from these problems • Atoms are not the most microscopic objects • Newton’s laws do not apply to the microscopic world of the electron • OUTCOME �New Rules!!!

Quantum Mechanics! • Describes rules that apply to electrons in atoms and molecules • Non-deterministic, probabilistic ! A new philosophy of nature – �Explains unsolved problems of late 19 th century physics – �Explains bonding, structure, and reactivity in chemistry
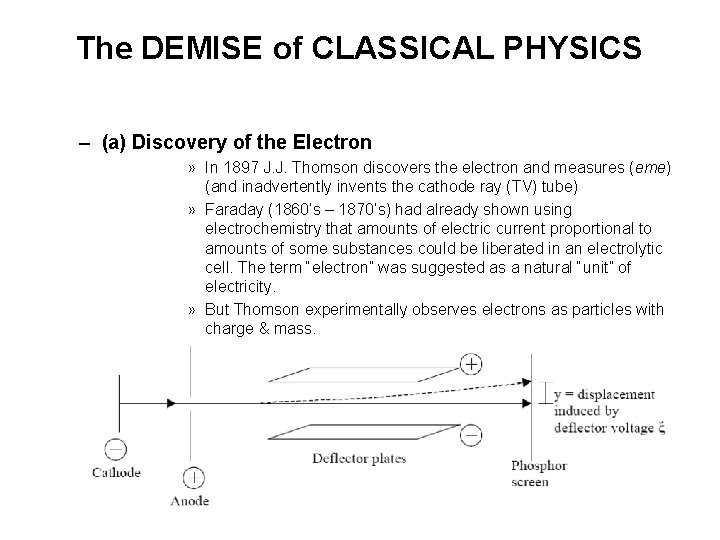
The DEMISE of CLASSICAL PHYSICS – (a) Discovery of the Electron » In 1897 J. J. Thomson discovers the electron and measures (eme) (and inadvertently invents the cathode ray (TV) tube) » Faraday (1860’s – 1870’s) had already shown using electrochemistry that amounts of electric current proportional to amounts of some substances could be liberated in an electrolytic cell. The term “electron” was suggested as a natural “unit” of electricity. » But Thomson experimentally observes electrons as particles with charge & mass.

» Thomson found that results are independent of (1) cathode material » (2) residual gas composition » �“electron” is a distinct particle, present in all materials! » Classical mechanics �force on electron due to deflector voltage:

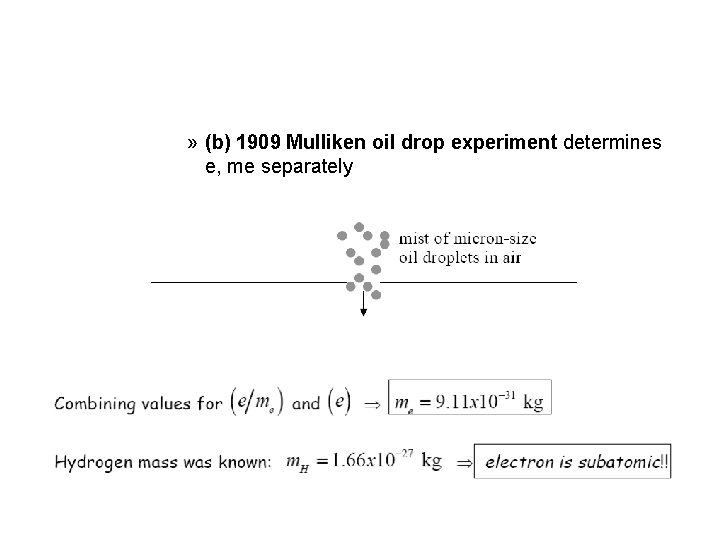
» (b) 1909 Mulliken oil drop experiment determines e, me separately
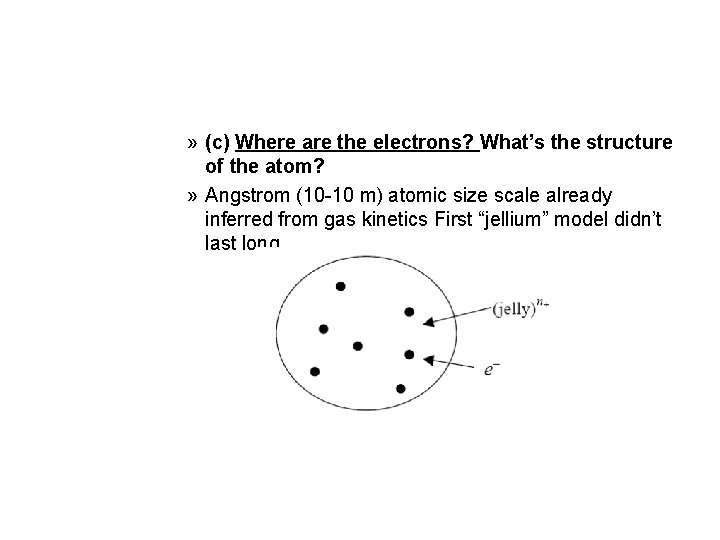
» (c) Where are the electrons? What’s the structure of the atom? » Angstrom (10 -10 m) atomic size scale already inferred from gas kinetics First “jellium” model didn’t last long

• Rutherford backscatterng experment » (1) He 2+ nucleus verysmall, << 10 -10 m (Rutherford estimated 10 -14 m) • (2) Au atoms are mostly empty!

» BUT model not consistent with classical electrodynamics: Accelerating charge emits radiation! (centripetal acceleration = v 2/r) And since light has energy, Emust be getting more negative with time » R must be getting smaller with time! » Electronspiralsintonucleusin~1010 s! » Also, as rdecreases, vshould increase Frequency �of emitted light = frequency of rotation » atom should emit light at all frequencies – that is it should produce a continuous spectrum

» BUT emission from atoms was known to be discrete, not continuous! » For the H atom, Rydberg showed that the spectrum was consistent with the simple formula:

Summary: Rutherford’s model of the atom • (1) Is not stable relative to collapse of electron into nucleus • (2) Does not yield discrete emission lines, • (3) Does not explain the Rydberg formula

The DEMISE of CLASSICAL PHYSICS (cont’d) • (a) Blackbody radiation --All things emit light when heated up! » Classically: (1) Radiation from a blackbody is the result of electrons oscillating with frequency � Oscillating charged parti antennae » (2) The electrons can oscillate (& radiate) equally well at any frequency • Rayleigh-Jeans Law for spectral density ρ(ν), where intensity of emitted light in frequency range from �ν to dν�ν+ is I(ν)~ ρ(ν) dν • k = Boltzmann’s constant [= R/NA (gas constant per molecule)] c = speed of light

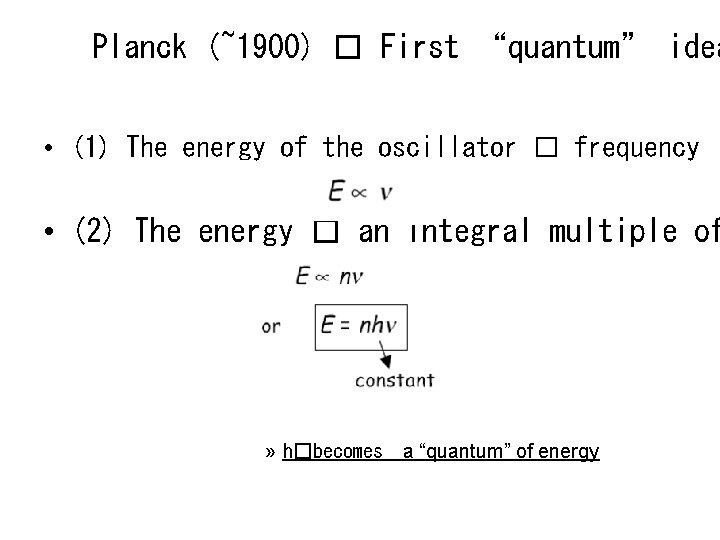
Planck (~1900) � First “quantum” idea • (1) The energy of the oscillator � frequency • (2) The energy � an integral multiple of » h�becomes a “quantum” of energy

» Planck used statistical mechanics (5. 62) to derive the expression for black body radiation

» (b) Photoelectric effect

» Einstein (1905) proposed: • (1) Light is made up of energy “packets: “photons” • (2) The energy of a photon is proportional to the light frequency E = hv h Planck’s constant

» New model of photoelectric effect: » Comparing to exp’t, value of “h” matches the one found by Planck! This was an extraordinary result !

» Summary: • (1) Structure of atom can’t be explained classically • (2) Discrete atomic spectra and Rydberg’s formula can’t be explained • (3) Blackbody radiation can be “explained” by quantifying energy of • oscillators E = h� • (4) Photoelectric effect can be “explained” by quantifying energy of light • E = h�

The ATOM of NIELS BOHR » Niels Bohr, a Danish physicist who established the Copenhagen school. – (a) Assumptions underlying the Bohr atom • (1) Atoms can exist in stable “states” without radiating. The states have discrete energies En, n= 1, 2, 3, . . . , where n= 1 is the lowest energy state (the most negative, relative to the dissociated atom at zero energy), n= 2 is the next lowest energy state, etc. The number “n” is an integer, a quantum number, that labels the state. • (2) Transitions between states can be made with the absorption or emission of a photon of frequency where ν = ΔE/ h.

» These two assumptions “explain” the discrete spectrum of atomic vapor emission. Each line in the spectrum corresponds to a transition between two particular levels. Thisisthebirthofmodernspectroscopy. » (3) Angular momentum is quantized: l = nh where h = h /2π • Angular momentum


» For H atom withn= 1, r=a 0= 5. 29 x 10 -11 m = 0. 529 Å (1 Å = 1010 m) » Take Rutherford’s energy and put in r, » Energies are quantized!!! » For H atom, emission spectrum
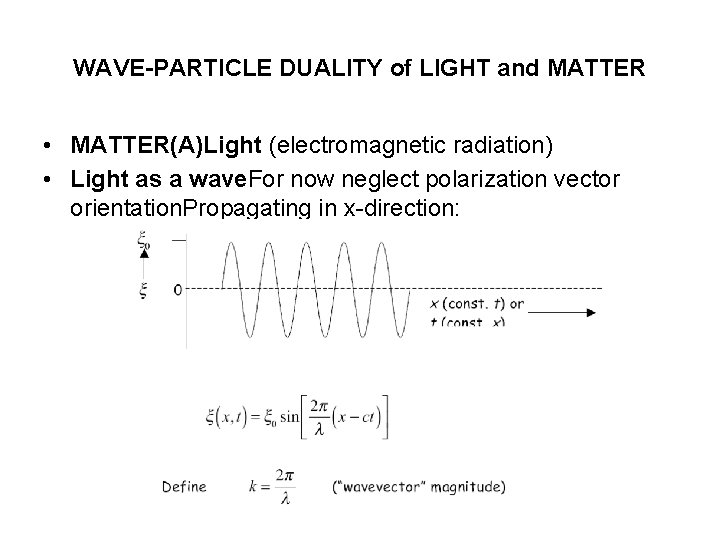
WAVE-PARTICLE DUALITY of LIGHT and MATTER • MATTER(A)Light (electromagnetic radiation) • Light as a wave. For now neglect polarization vector orientation. Propagating in x-direction:
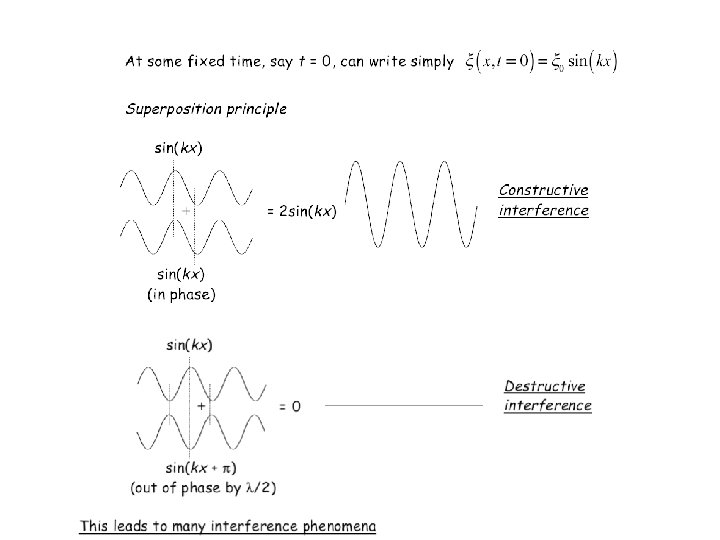

Young’s 2 -slit experiment
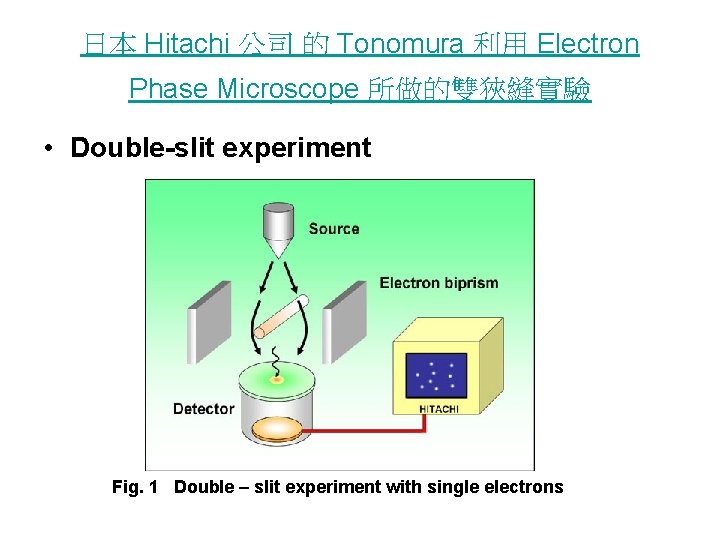
日本 Hitachi 公司 的 Tonomura 利用 Electron Phase Microscope 所做的雙狹縫實驗 • Double-slit experiment Fig. 1 Double – slit experiment with single electrons

Fig. Single electron events build up to from an interference pattern in 2 the double-slit experiments.
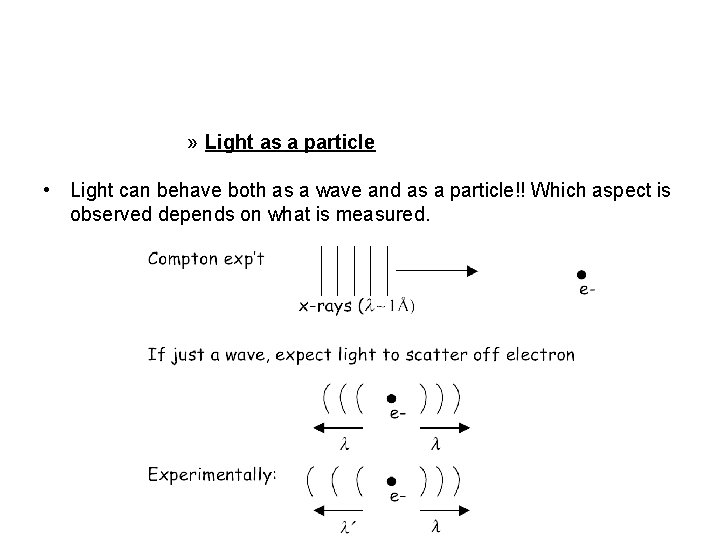
» Light as a particle • Light can behave both as a wave and as a particle!! Which aspect is observed depends on what is measured.
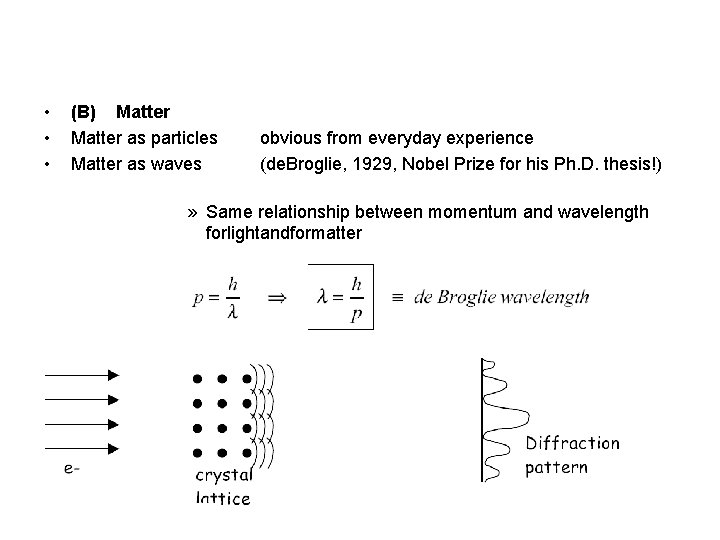
• • • (B) Matter as particles Matter as waves obvious from everyday experience (de. Broglie, 1929, Nobel Prize for his Ph. D. thesis!) » Same relationship between momentum and wavelength forlightandformatter
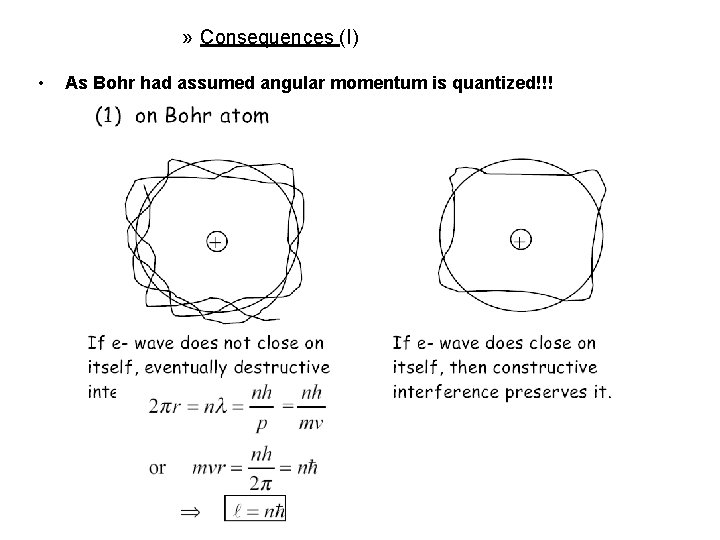
» Consequences (I) • As Bohr had assumed angular momentum is quantized!!!

WAVE-PARTICLE DUALITY of MATTER » Consequences (II) » Heisenberg Uncertainty Principle » Consider diffraction through a single slit
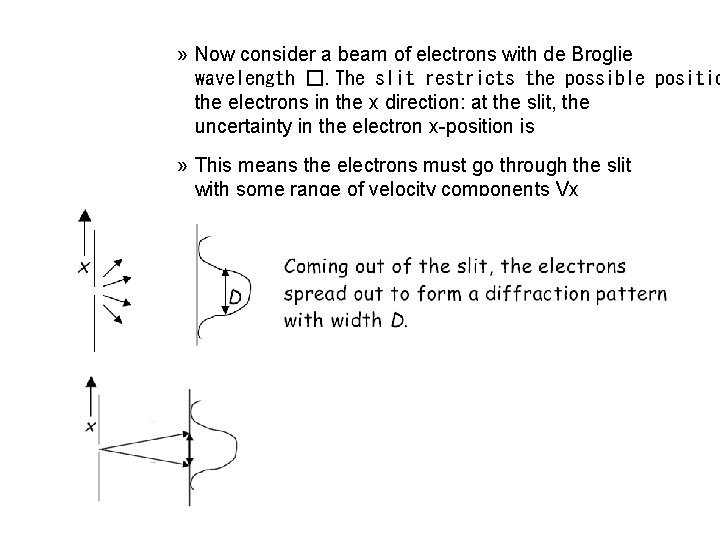
» Now consider a beam of electrons with de Broglie wavelength �. The slit restricts the possible positio the electrons in the x direction: at the slit, the uncertainty in the electron x-position is » This means the electrons must go through the slit with some range of velocity components Vx

» So the position and momentum of a particle cannot both be determined with arbitrary position! Knowing one quantity with high precision means that the other must necessarily be imprecise!

» The conventional statement of the Heisenberg Uncertainty Principle is » Implications for atomic structure » Apply Uncertainty Principle to e-in H atom

» Basically, if we know the e-is in the atom, then we can’t know its velocity at all! » Bohr had assumed the electron was a particle with a known position and velocity. To complete the picture of atomic structure, the wavelike properties of the electron had to be included. » So how do we properly represent where the particle is? ?

» Schrodinger (1933 Nobel Prize) » A particle in a “stable” or time-independent state can be represented mathematically as a wave, by a “wavefunction” �(x) (in 1 -D) which is a solution to • the differential equation
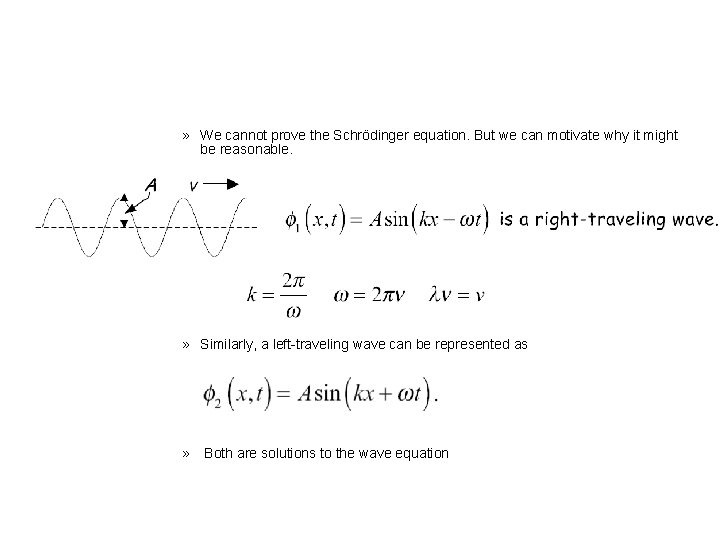
» We cannot prove the Schrödinger equation. But we can motivate why it might be reasonable. » Similarly, a left-traveling wave can be represented as » Both are solutions to the wave equation

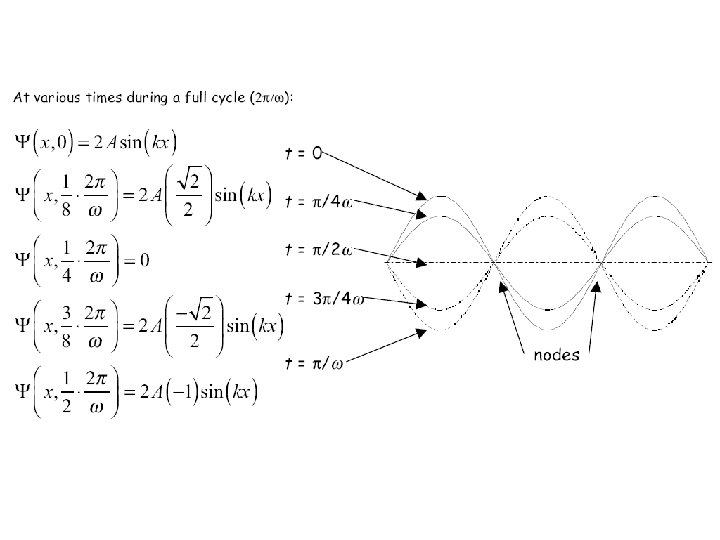

» As in a vibrating violin string, the node positions are independent of time. Only the amplitude of the fixed waveform oscillates with time. » More generally, we can write wave equation solutions in the form

» We now have the outline of: » a physicalpictureinvolving wave and particle duality of light and matter ! » a quantitativetheoryallowing calculations of stable states and their properties !


- Slides: 46