PHYSICAL CHEMISTRY ProgramBSc SemesterV Topic Elementary Quantum Mechanics

PHYSICAL CHEMISTRY Program-BSc Semester-V Topic: Elementary Quantum Mechanics (Part-1)

CONTENTS ØBlack body radiation ØRayleigh-Jeans Law ØStefan Boltzmann Law ØWien’s Displacement Law Planck’s Law

Black body radiation ØA black body is an ideal body which completely absorbs the radiant energy falling on it. ØThe electromagnetic radiation emitted by a black body is called blackbody radiation. A black body is a perfect radiator as well as perfect absorber of radiant energy.
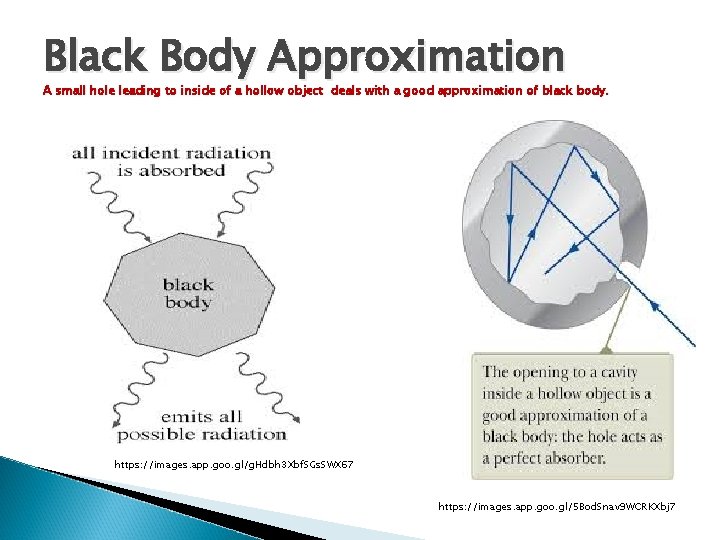
Black Body Approximation A small hole leading to inside of a hollow object deals with a good approximation of black body. https: //images. app. goo. gl/g. Hdbh 3 Xbf. SGs. SWX 67 https: //images. app. goo. gl/5 Bod. Snav 9 WCRKXbj 7

Rayleigh-Jeans Law Rayleigh-Jeans law based on classical physics: The law matched experimental results well at longer wavelength. https: //images. app. goo. gl/aaf. Lax. Honeehvn 5 BA
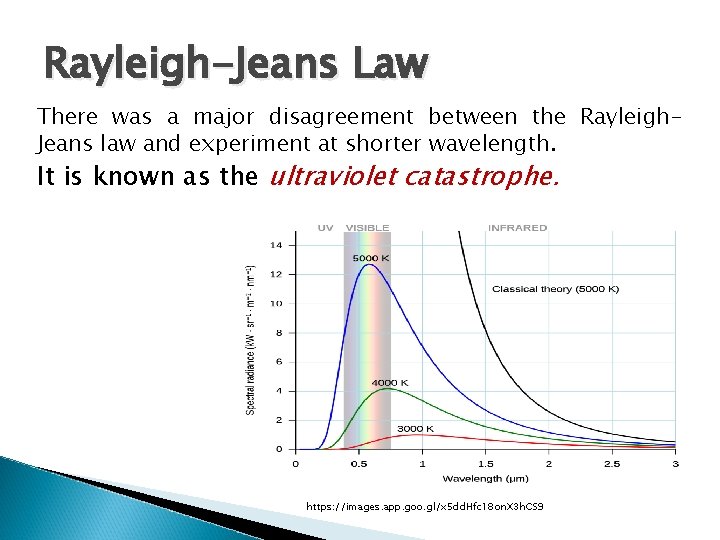
Rayleigh-Jeans Law There was a major disagreement between the Rayleigh. Jeans law and experiment at shorter wavelength. It is known as the ultraviolet catastrophe. https: //images. app. goo. gl/x 5 dd. Hfc 18 on. X 3 h. CS 9

Stefan Boltzmann Law Ø The total power of the emitted radiation increases with temperature. ØEnergy radiated by blackbody is proportional to temperature to the fourth power. P=s. Ae T 4 Joseph Stefan https: //images. app. goo. gl/2 eig. VWGptvyaw. Vak 6 Ø where s = 5. 67 * 10 -8 J s-1 m-2 K-4, Known as the Stefan-Boltzmann constant. https: //images. app. goo. gl/R 9 r. W 6 WVq. Dh 9 X 2 x. Ao 6
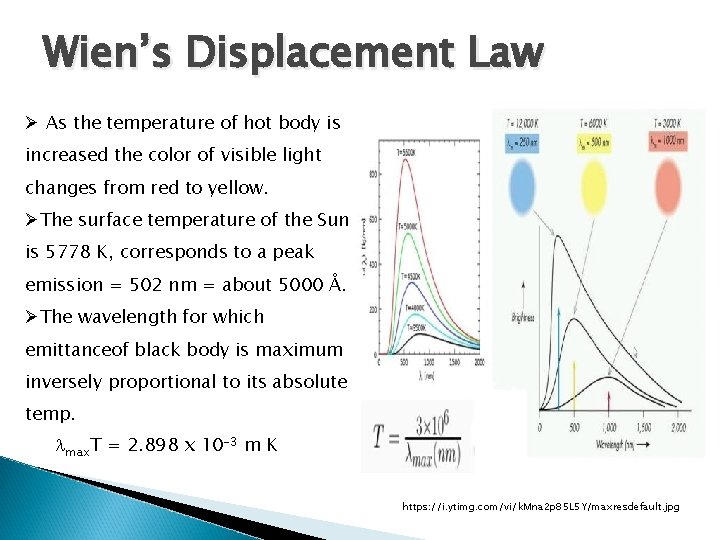
Wien’s Displacement Law Ø As the temperature of hot body is increased the color of visible light changes from red to yellow. ØThe surface temperature of the Sun is 5778 K, corresponds to a peak emission = 502 nm = about 5000 Å. ØThe wavelength for which emittanceof black body is maximum inversely proportional to its absolute temp. lmax. T = 2. 898 x 10 -3 m K https: //i. ytimg. com/vi/k. Mna 2 p 85 L 5 Y/maxresdefault. jpg

Planck’s Radiation Law Max Planck was a German physicist He introduced the concept of “quantum of action” He was awarded the Nobel Prize in 1918 for the discovery of the quantized nature of energy. In 1900 Planck developed a theory of blackbody radiation that leads to an equation for the intensity of the radiation. Max Planck https: //images. app. goo. gl/11 Xt. Ar. Uz. YYy 1 aa. VZ 8

Planck’s Radiation Law A body can emit or absorb energy in whole number multiples of hv. Energy in fraction of quantum cannot be absorbed or lost. This phenomena is called quantization of energy. Ø This expression is called Planck’s radiation law

Planck’s Radiation Law ØHe assumed the cavity radiation came from atomic oscillations in the cavity walls. ØAssumption of Planck’s theory ØAssumption 1# The energy of an oscillator can have only certain discrete values En. and correspond to different quantum state represented by quantum number n. ◦ En = n h ƒ ØAssumption 2# The oscillators emit or absorb energy when making a transition from one quantum state to another. ◦ The energy carried by the quantum of radiation is E = h ƒ.

Planck’s model graph https: //images. app. goo. gl/xpa 4 AFzy. PJhwwrb. N 6

References: https: //web. njit. edu www. google. com Chemistry for Degree Students by R. L. Madan publisher S. Chand Physical chemistry by B. R. Puri Sharma, L. R. Sharma & Pathania
- Slides: 13