Physical chemistry of light OBJECTIVES l Calculate the

Physical chemistry of light OBJECTIVES: l Calculate the wavelength, frequency, or energy of light, given two of these values.

Physical chemistry of light OBJECTIVES: l Explain the origin of the atomic emission spectrum of an element.

Light l The study of light led to the development of the quantum mechanical model. l Light is a kind of electromagnetic radiation. l Electromagnetic radiation includes many kinds of waves l All move at 3. 00 x 108 m/s = c
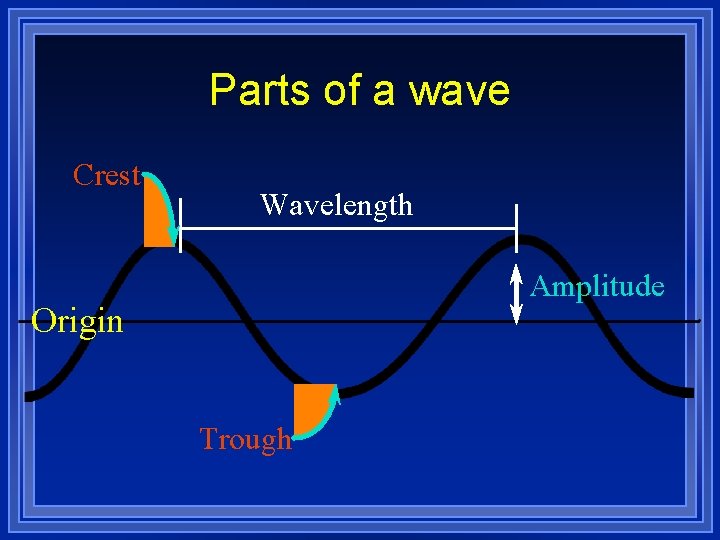
Parts of a wave Crest Wavelength Amplitude Origin Trough

Parts of Wave l Origin - the base line of the energy. l Crest - high point on a wave l Trough - Low point on a wave l Amplitude - distance from origin to crest l Wavelength - distance from crest to crest l Wavelength is abbreviated by the Greek letter lambda =

Frequency l The number of waves that pass a given point per second. l Units: cycles/sec or hertz (hz or sec-1) l Abbreviated by Greek letter nu = n c =

Frequency and wavelength l Are inversely related l As one goes up the other goes down. l Different frequencies of light are different colors of light. l There is a wide variety of frequencies l The whole range is called a spectrum,

Low energy High energy Radio Micro Infrared Ultra- XGamma waves. violet Rays Low High Frequency Long Short Wavelength Visible Light

Prism White light is made up of all the colors of the visible spectrum. l Passing it through a prism separates it. l

If the light is not white By heating a gas with electricity we can get it to give off colors. l Passing this light through a prism does something different. l

Atomic Spectrum Each element gives off its own characteristic colors. l Can be used to identify the atom. l How we know what stars are made of. l

• These are called discontinuous spectra, or line spectra • unique to each element. • These are emission spectra • The light is emitted given off

Light is a Particle l Energy is quantized. l Light is energy l Light must be quantized l These smallest pieces of light are called photons. l Photoelectric effect? l Energy & frequency: directly related.

Energy and frequency =hx l E is the energy of the photon l is the frequency l h is Planck’s constant l h = 6. 6262 x 10 -34 Joules x sec. l joule is the metric unit of Energy l. E
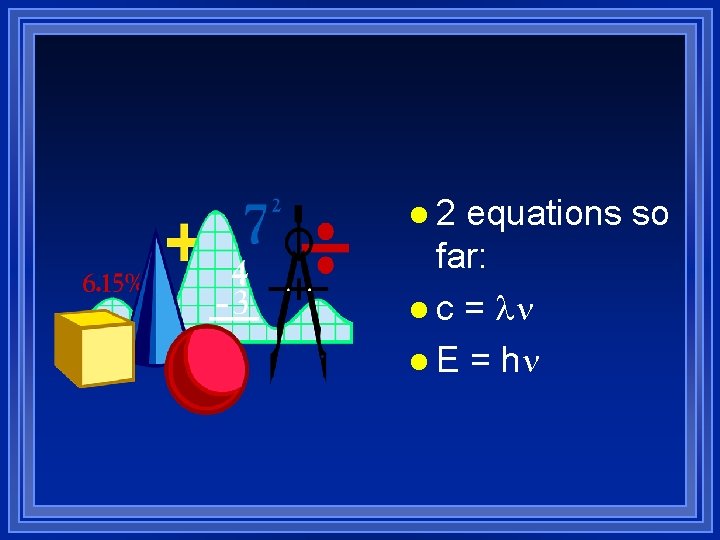
l 2 equations so far: l c = l E = h

Examples l What is the wavelength of blue light with a frequency of 8. 3 x 1015 Hz? l What is the frequency of red light -5 with a wavelength of 4. 2 x 10 m? l What is the energy of a photon of each of the above?

Explanation of atomic spectra l When we write electron configurations, we are writing the lowest energy. l The energy level, and where the electron starts from, is called it’s ground state- the lowest energy level.
Changing the energy l Let’s look at a hydrogen atom
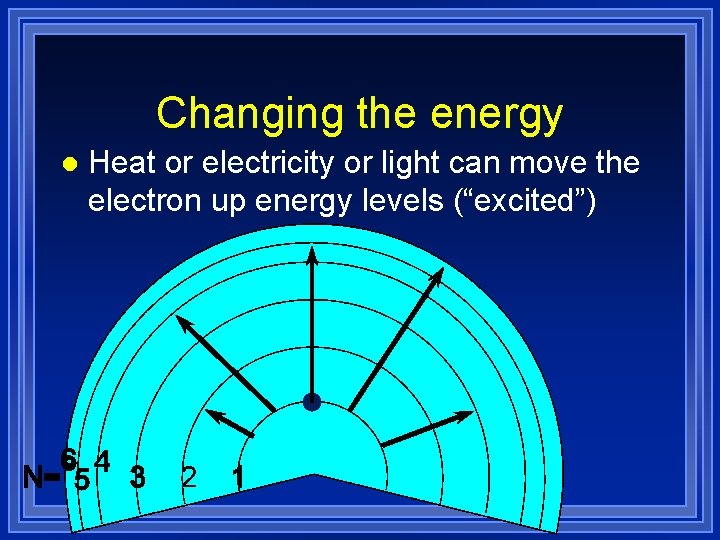
Changing the energy l Heat or electricity or light can move the electron up energy levels (“excited”)
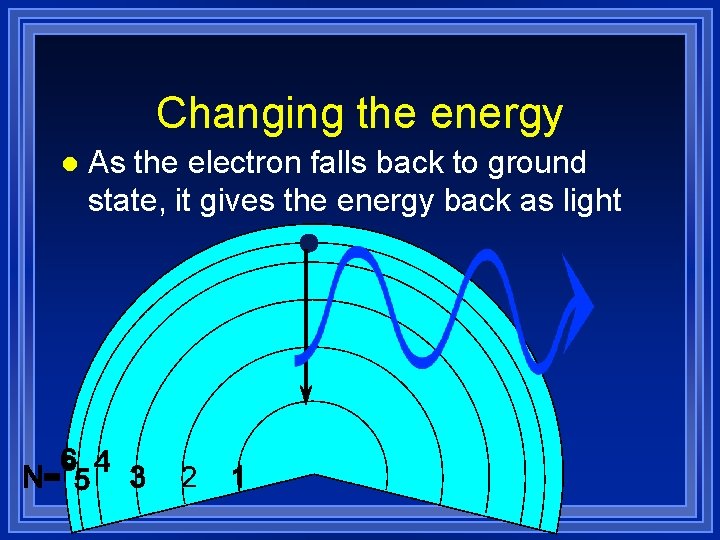
Changing the energy l As the electron falls back to ground state, it gives the energy back as light
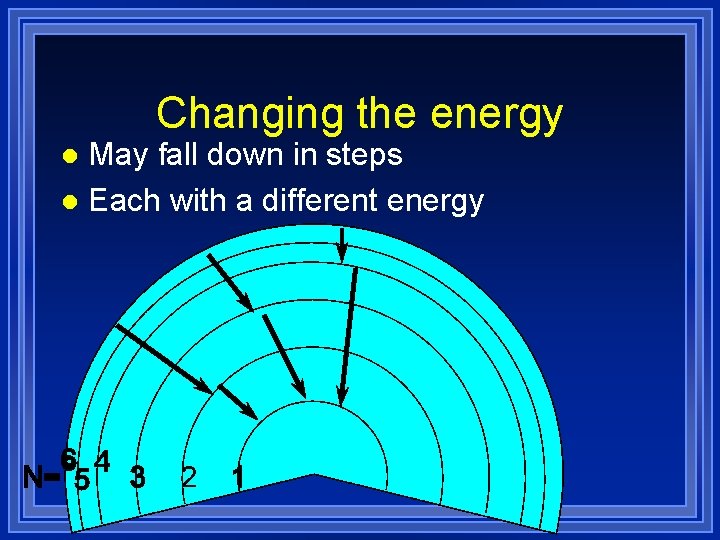
Changing the energy May fall down in steps l Each with a different energy l
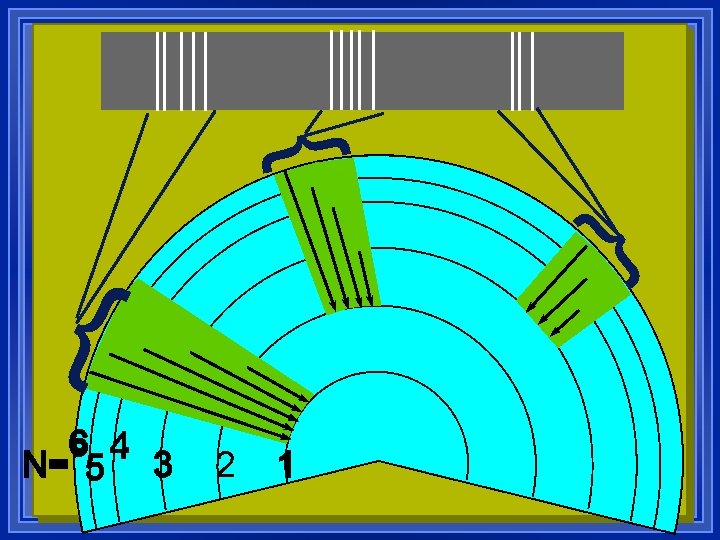

Ultraviolet Visible Infrared l Further they fall, more energy, higher frequency. l This is simplified l the orbitals also have different energies inside energy levels l All the electrons can move around.

What is light? Light is a particle - it comes in chunks. l Light is a wave- we can measure its wavelength and it behaves as a wave 2 l If we combine E=mc , c= , E = 1/2 mv 2 and E = h l We can get: = h/mv l called de Broglie’s equation l Calculates the wavelength of a particle. l

Sample problem l What is the approximate mass of a particle having a wavelength of 10 -7 meters, and a speed of 1 m/s? -Use = h/mv = 6. 6 x 10 -27 (Note: 1 J = N x m; 1 N = 1 kg x m/s 2

Matter is a Wave l Does not apply to large objects l Things bigger than an atom l. A baseball has a wavelength of about 10 -32 m when moving 30 m/s l An electron at the same speed has a wavelength of 10 -3 cm l Big enough to measure.

The physics of the very small l Quantum mechanics explains how the very small behaves. l Classic physics is what you get when you add up the effects of millions of packages. l Quantum mechanics is based on probability

Heisenberg Uncertainty Principle l -It is impossible to know exactly the location and velocity of a particle. l The better we know one, the less we know the other. l Measuring changes the properties. l Instead, analyze interactions with other particles

More obvious with the very small l To measure where a electron is, we use light. l But the light moves the electron l And hitting the electron changes the frequency of the light.

Before Photon Moving Electron After Photon changes wavelength Electron changes velocity
- Slides: 30