Physical Characteristics of the Ocean Environment SOL 5





















- Slides: 22

Physical Characteristics of the Ocean Environment SOL 5. 6

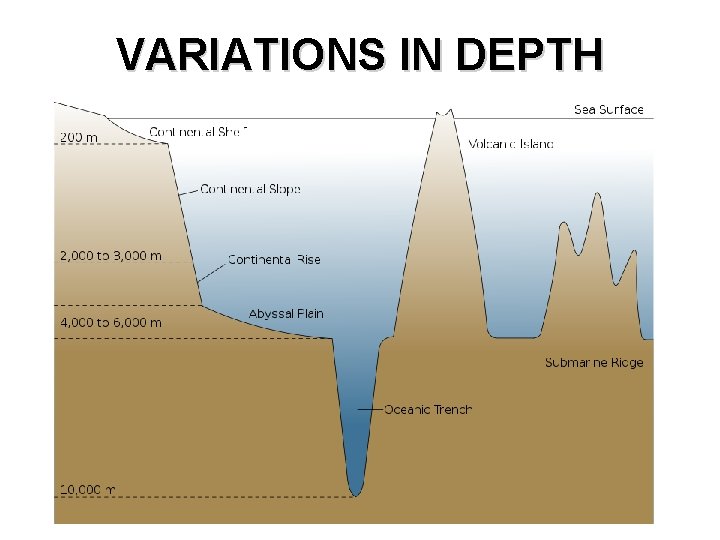
OBJECTIVES • TSW – Describe the variation in depths associated with ocean features (shelf, slope, rise, abyssal plain, and trench) – Explain the cause of salinity in ocean water – Compare and contrast various salinity levels of ocean water – Explain what causes currents and tides – Locate and describe the Gulf Stream – Describe how scientists have explored the oceans

VARIATIONS IN DEPTH

Why is ocean water salty?

SALINITY • All water is made of hydrogen and oxygen. • Ocean water is not just pure water. • It has dissolved gases, like air, and dissolved solids, especially salt. • The salinity measures how much salt is present in water. (“Salinity = saltiness”) • Common salt (sodium chloride) is the main salt found in the ocean.

SALINITY • Different bodies of water can have different amounts of salt in solution (different salinities) • Ocean water is about 220 times saltier than fresh lake water. • Measure salinity by finding how much total salt is dissolved in 1, 000 grams of ocean water (1 ppt) • On average, 35 grams of salt are dissolved in every 1, 000 grams of ocean water (35 ppt) • The salinity of fresh water is usually less than 0. 5 ppt (0. 5 ppt – 17 ppt is called brackish)

How did the salt get into the ocean?

SALINITY • Most of the salt in the ocean was carried there by rivers. Rivers pick up pieces of rock, sediment, and soil as they flow. All rivers meet at an estuary, where the runoff containing mineral salts, is deposited into the ocean. Eventually, the salts dissolve into the ocean. • Evaporation is another reason. The sun’s heat causes some water to evaporate. Salts do not evaporate. Over time, the salts that remain increase the salinity of the ocean. • Some of the salt in the oceans comes from undersea volcanoes and hydrothermal vents.

Why does ocean salinity vary?

SALINITY • The ocean’s salinity varies from place to place. • In hot tropical/warm, dry regions, ocean water evaporates and little precipitation cause ocean water to have a high salinity. • In cold regions, salinity is lower because less evaporation takes place and melting ice and snow add fresh water. Areas of lower salinity also occur where freshwater rivers enter the ocean.

SALINITY • Depth affects salinity, too. • Salt water has a greater density (“is heavier”) than fresh water. • Saltier water sinks deeper. • Therefore, salinity increases with ocean depth.

SALINITY SOL 5. 6 Video Clip – 2 mins

Physical Characteristics CHECKPOINT 1. What does salinity measure? a) b) c) d) How much ocean water evaporates How much salt is present in ocean water How deep the ocean water is How much pressure is applied

Physical Characteristics CHECKPOINT 2. What are two reasons that the ocean is saltwater? No multiple choice. . .

Physical Characteristics CHECKPOINT 3. Explain why salinity increases as you go deeper in the ocean. No multiple choice. . .

Physical Characteristics CHECKPOINT 4. How does most salt get into the ocean? a) b) c) d) rainfall rivers animals volcanoes

Physical Characteristics CHECKPOINT 5. What makes the ocean in hot tropical regions have a high salinity? a) b) c) d) Melting polar ice caps Wind patterns High evaporation rate None of the above

Physical Characteristics CHECKPOINT BIG BUCKS BONUS: Which type of ocean water would have the highest salinity? a) b) c) d) Deep water in the tropics Shallow water in the tropics Deep water near the poles Shallow water near the poles

Investigating the Ocean Environment - Salinity SOL 5. 6 Lab

SOL 5. 6 Lab • Salt • Fresh water • Large, clear plastic cups • Food coloring • Spoons • Potato slices • ocean water • Graduated cylinders • Hand lenses or microscope • Droppers • Clean microscope slides • Lab sheet (each) • One lab partner (jigsaw)

SOL 5. 6 PROCEDURE A PARTNER 1 • Fill 2 clear plastic cups less than half full with the same amount of fresh water • Mix a few drops of food coloring into one cup (fresh water) • Very slowly and gently lower a potato slice into the cup without stirring the water. PARTNER 2 • Mix 8 teaspoons of salt into the water of the other cup (salt water) • Hold the spoon directly over the salt water while PARTNER 1 pours the colored water very slowly into the spoon so that it slowly drips out into the salt water • Try to keep the spoon close to the salt water when pouring the colored water in order to reduce splashing and mixing of the waters

SOL 5. 6 PROCEDURE B PARTNER 1 PARTNER 2 • Get ½ cup of salt water • Get ½ cup of freshwater • Smell both samples and describe what you smell • Pour ¼ of freshwater into a graduated cylinder to determine the volume • Pour ¼ cup of salt water into a graduated cylinder to determine the volume • Put a drop of saltwater on a clean microscope slide and observe it with a magnifying glass (or a microscope) • Put a drop of freshwater on a clean microscope slide and observe it with a magnifying glass (or a microscope) • Fan the two drops of water until the water evaporates