Physical Behavior of Matter Physical Behavior of Matter

Physical Behavior of Matter

Physical Behavior of Matter

Physical Behavior of Matter

Physical Behavior of Matter
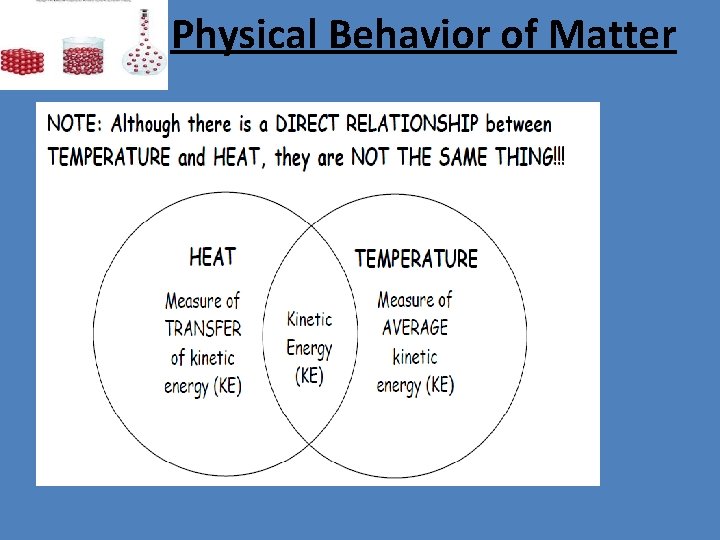
Physical Behavior of Matter

How does heat flow? Ex. A hot brick is placed in a cold bucket of water. Describe how heat flows and what happens to temperature.

Physical Behavior of Matter

Physical Behavior of Matter

Physical Behavior of Matter

Physical Behavior of Matter
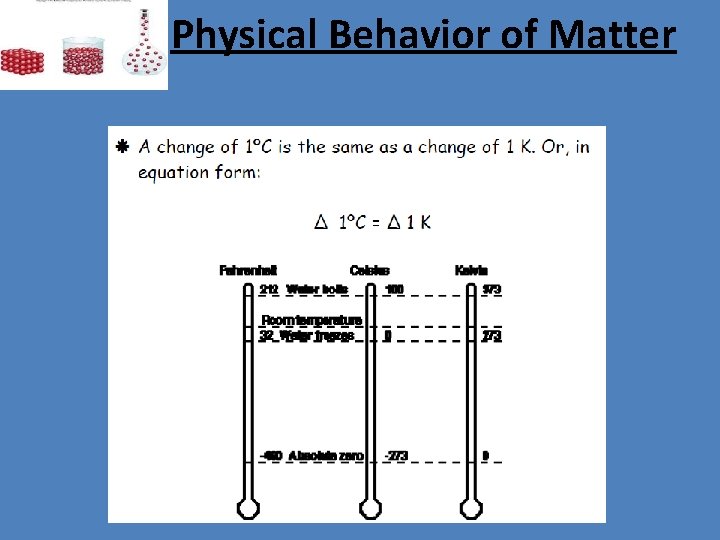
Physical Behavior of Matter

Physical Behavior of Matter

Physical Behavior of Matter
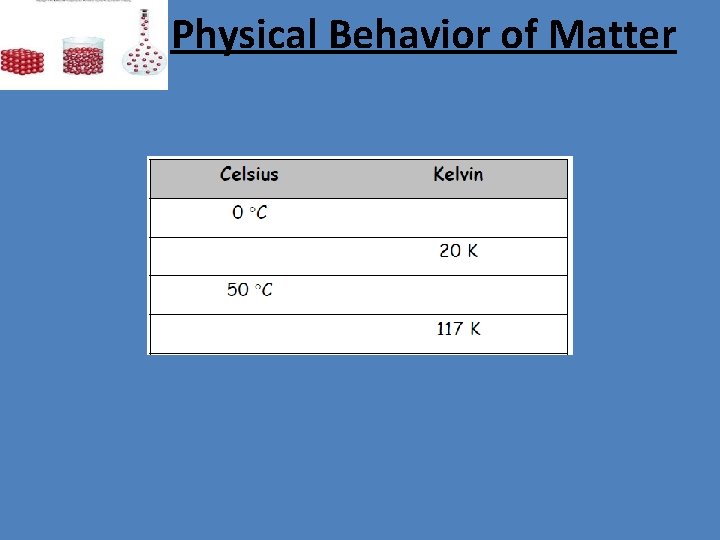
Physical Behavior of Matter
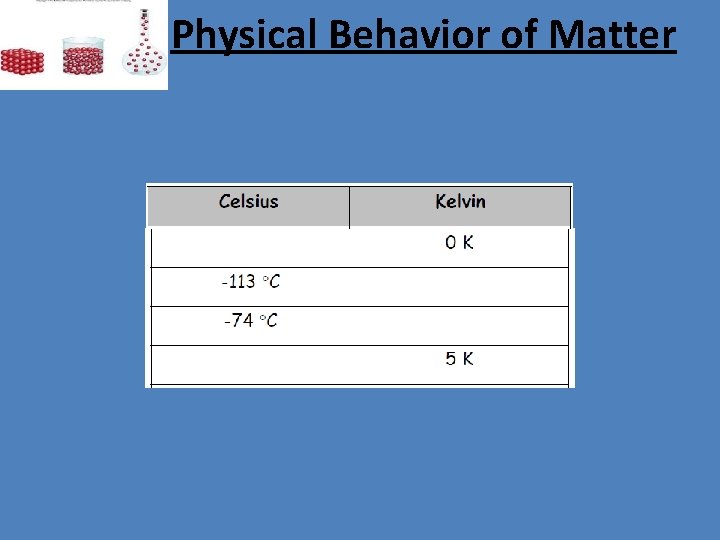
Physical Behavior of Matter

Physical Behavior of Matter

Review from Wednesday’s class…. . 1. Energy of motion/movement is known as Kinetic energy __________. Potential energy 2. Energy that is stored is known as ______. of the average KE of a substance’s 3. Define temperature. Measure particles. 4. True or False? Heat transferred depends on The larger the sample size, the more heat needed sample size. True. to bring to desired temperature. Joules, Kilojoules, calories 5. Units of measurement for heat are _____. 6. What is the one thing that heat and temperature have in common? Kinetic energy

Physical Behavior of Matter http: //www. harcourtschool. com/activity/states_of_matter/
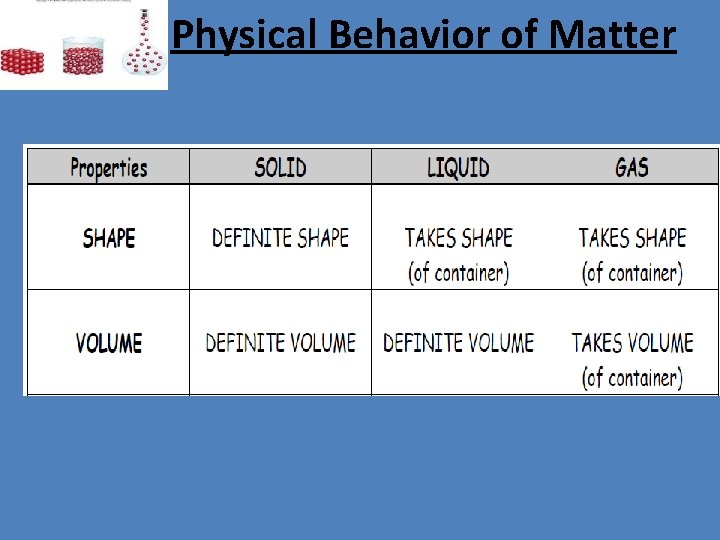
Physical Behavior of Matter
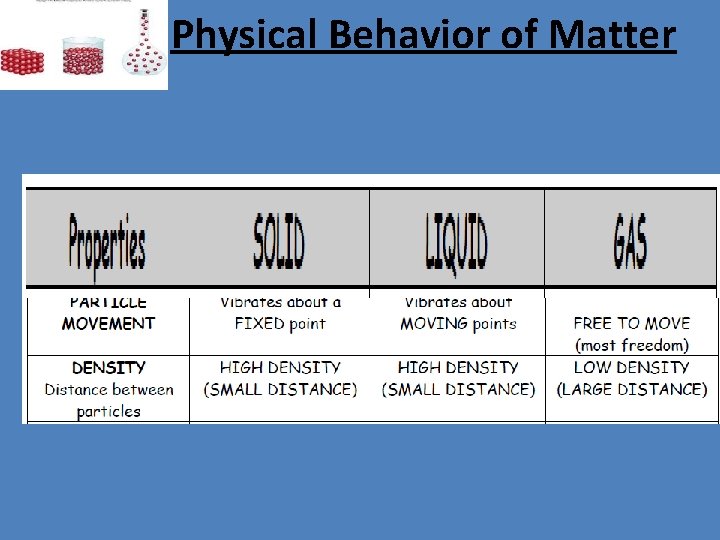
Physical Behavior of Matter
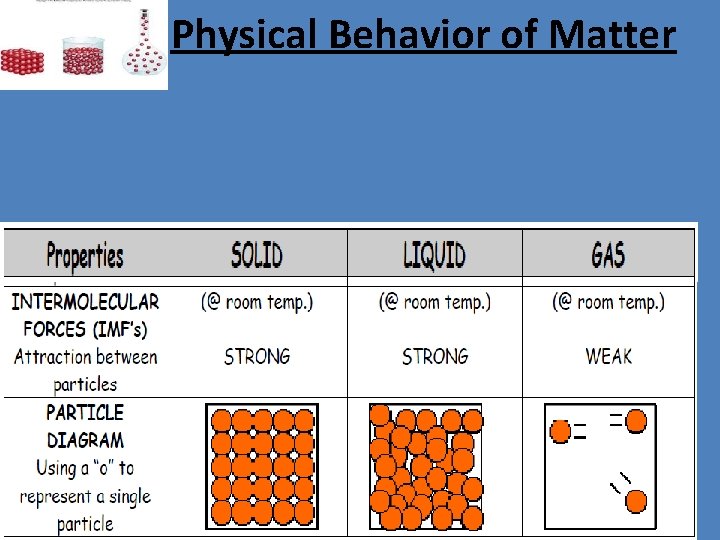
Physical Behavior of Matter
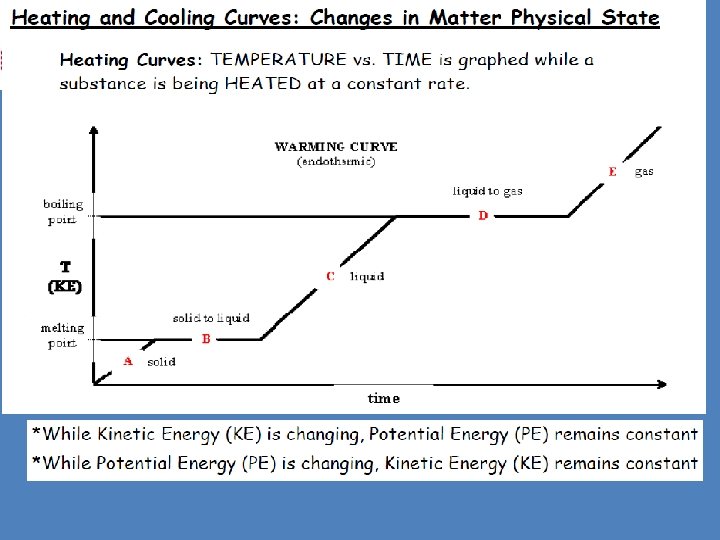
Physical Behavior of Matter
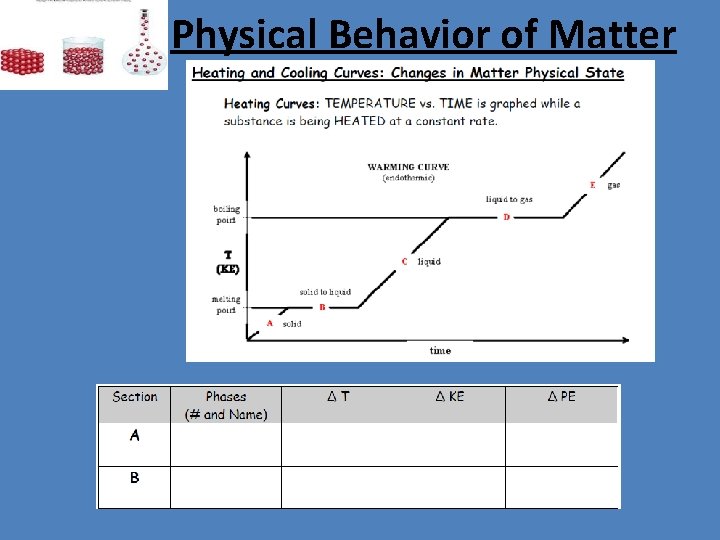
Physical Behavior of Matter
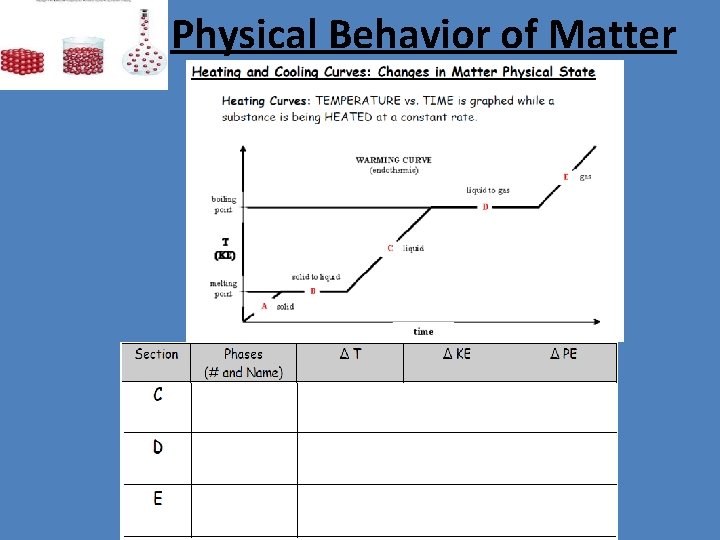
Physical Behavior of Matter
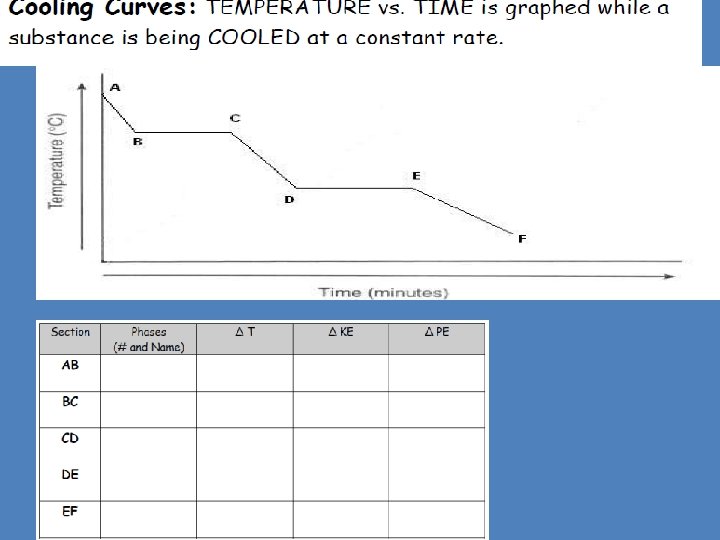
Physical Behavior of Matter
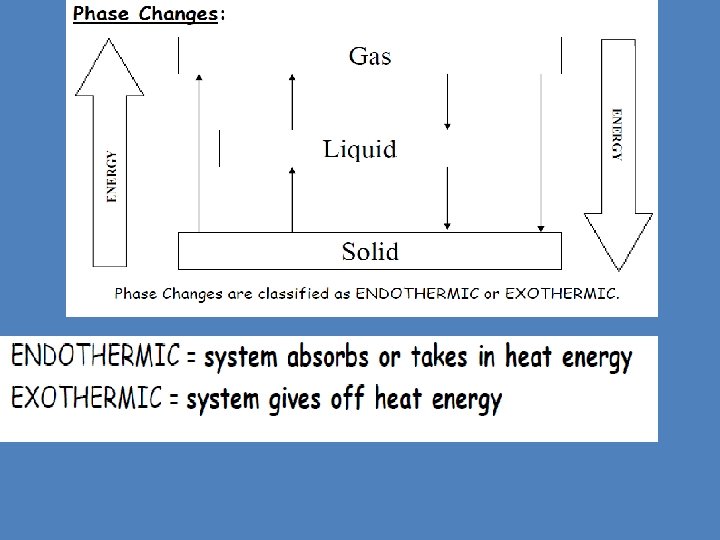


Physical Behavior of Matter Review 1. Identify the following phase changes: EVAPORATION a. Liquid Gas b. Solid Liquid MELTING c. Solid Gas SUBLIMATION CONDENSATION d. Gas Liquid 2. Which of the TWO (endothermic or exothermic), ENDOTHERMIC absorbs energy? 3. Which of the TWO (endothermic or exothermic), EXOTHERMIC release energy? 4. Which of the TWO (endothermic or exothermic) happens spontaneously (unannounced)? EXOTHERMIC






• A 500. g sample of water had a temperature change from 10. c to 15 c. How much heat was produced?

#3 A sample is heated from 20 c to 100 c as 70, 000 J of heat is released. How many grams of water were heated?

#4 A 2, 000 g sample of water whose temp was 50 c lost 33, 600 J of heat over a ten minute period. What was the temperature of the water at the end?

6. Calculate the specific heat of a substance when 63 J of energy are transferred as heat to an 8. 0 gram sample to raise its temperature from 314 K to 340 K.
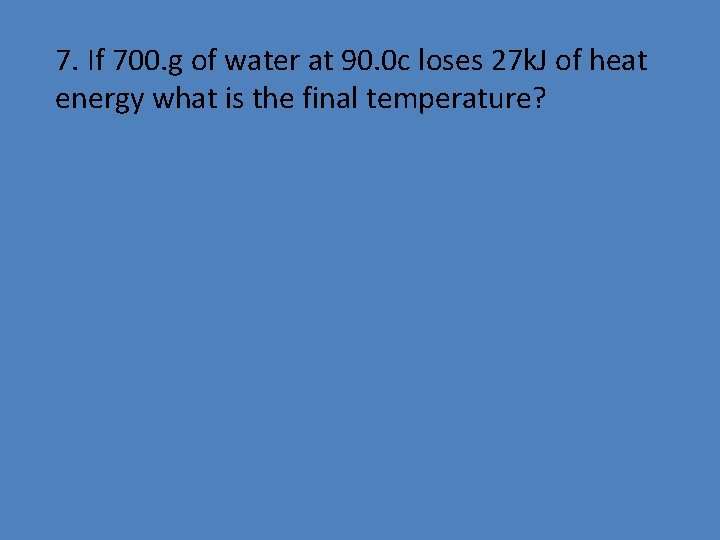
7. If 700. g of water at 90. 0 c loses 27 k. J of heat energy what is the final temperature?



3. q=mc^t Q=(22. 8)(. 14)(164) Q=52. 3488~52 J 4. q=mc^t 755=m(. 387)(25) m=78. 036~78 g


7. q=mc^t Q= (500. )(4. 18)(60) Q= 125, 400~100, 000 J




Physical Behavior of Matter

Physical Behavior of Matter









Physical Behavior of Matter
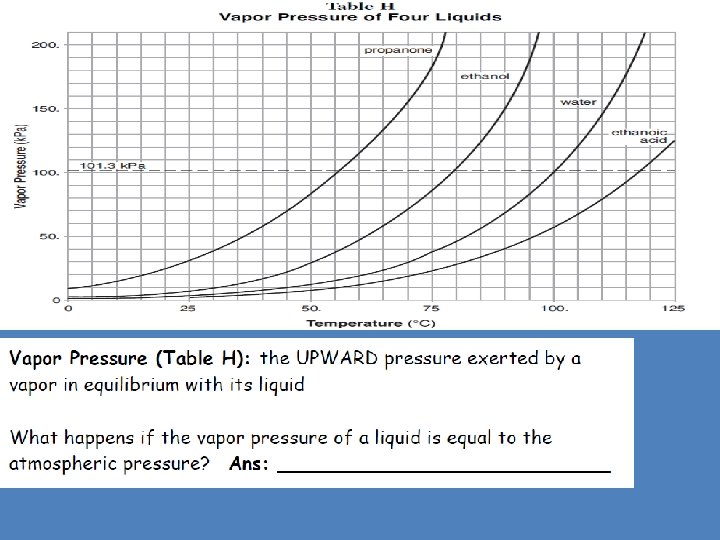
Physical Behavior of Matter

Physical Behavior of Matter
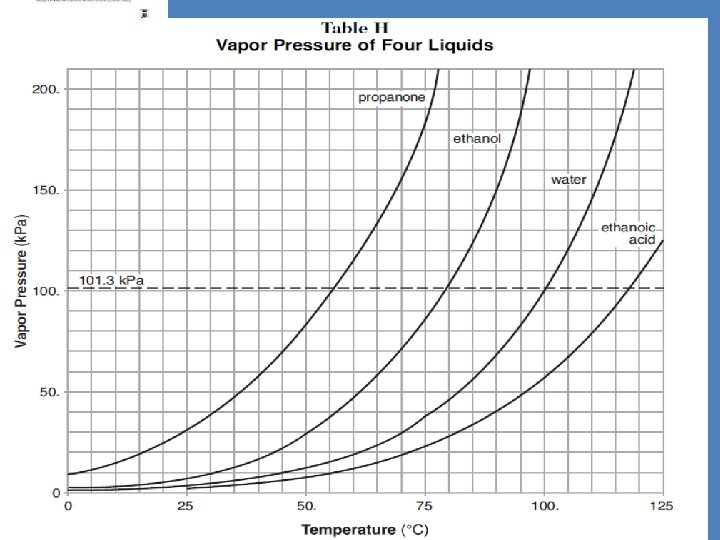
Physical Behavior of Matter
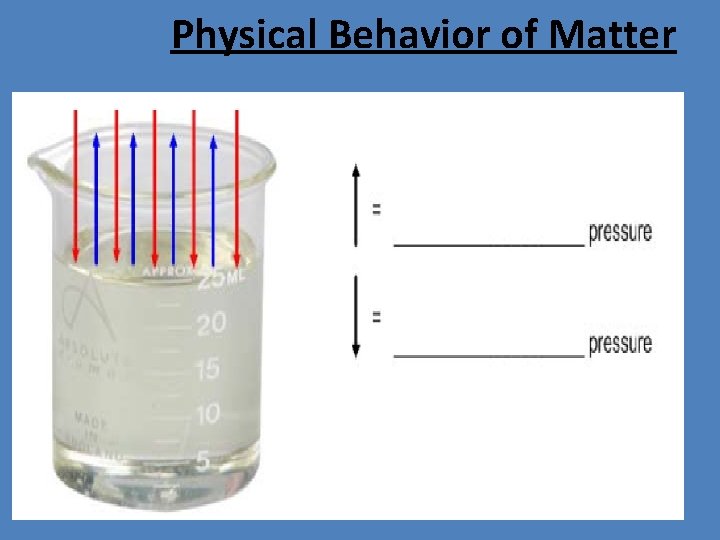
Physical Behavior of Matter

Convert Pressure: 1. 4. 67 atm=? k. Pa 2. 28. 7 k. Pa=? atm







Physical Behavior of Matter

Physical Behavior of Matter

Physical Behavior of Matter

Physical Behavior of Matter



1. A mixture of oxygen, nitrogen and hydrogen gases exert a total pressure of 74. 0 k. Pa at 0 c. The partial pressure of the oxygen is 20. 0 k. Pa and the partial pressure of nitrogen is 40. 0 k. Pa. What is the partial pressure of hydrogen in this mixture?









P 11 Boyles P 1 V 1= P 2 V 2 1. (100. 0)(780. 0)=760. V 2=102. 6315~103 m. L 2. (250. 0)(760. )=740. 0 V 2 V 2=256. 756~257 m. L

3. (25. 0)(760. )=600. 0 V 2 V 2=31. 66~31. 7 m. L 4. (750. 0)(700. 0)=800. 0 V 2 V 2=656. 25~656. 3 m. L

Charles V 1 = V 2 T 1 T 2 1. 125=V 2 298 273 2. 310. =V 2 331 288 V 2=114. 5134~115 m. L V 2=269. 72~270. m. L

3. 20. 0=V 2 306 330 V 2=21. 568~21. 6 m. L 4. 350. =V 2 243 273 V 2=393. 209~393 m. L


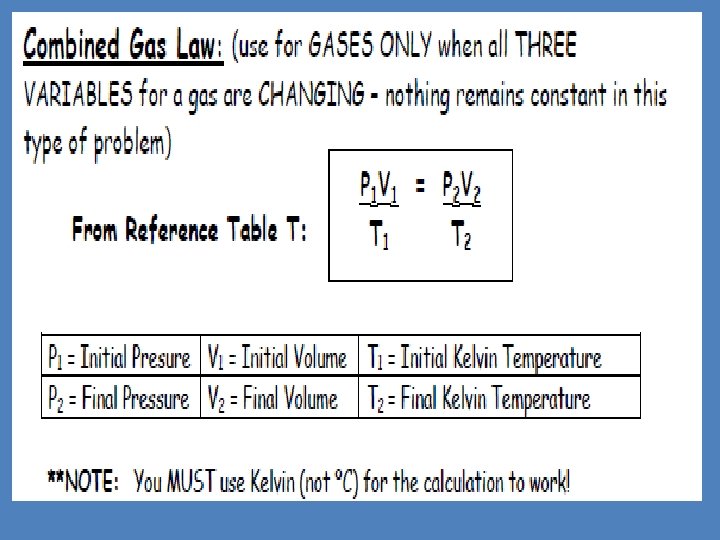


- Slides: 93