Physical Behavior of Matter Phases of Matter 2

Physical Behavior of Matter Phases of Matter

2 Forms of Energy Kinetic Energy Potential Energy �Energy of motion �Temperature is the measurement of the average K. E. �Higher Temp = Higher K. E. �Heat is a form of energy �Stored Energy
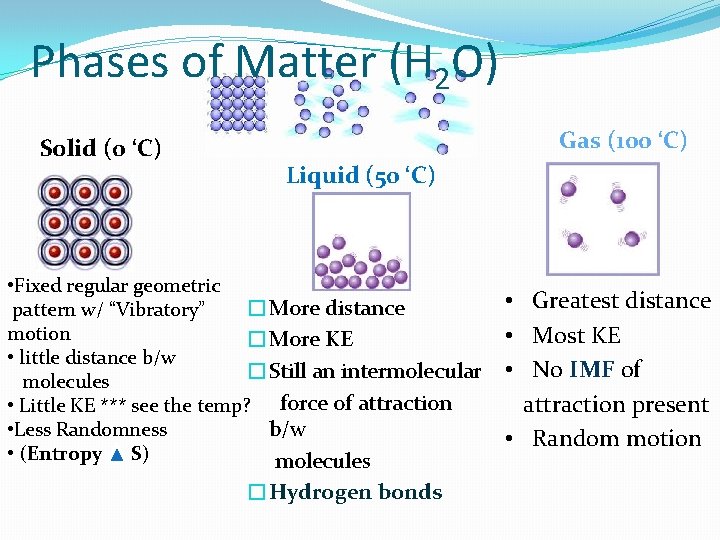
Phases of Matter (H 2 O) Solid (0 ‘C) Gas (100 ‘C) Liquid (50 ‘C) • Fixed regular geometric �More distance pattern w/ “Vibratory” motion �More KE • little distance b/w �Still an intermolecular molecules • Little KE *** see the temp? force of attraction b/w • Less Randomness • (Entropy S) molecules �Hydrogen bonds • Greatest distance • Most KE • No IMF of attraction present • Random motion
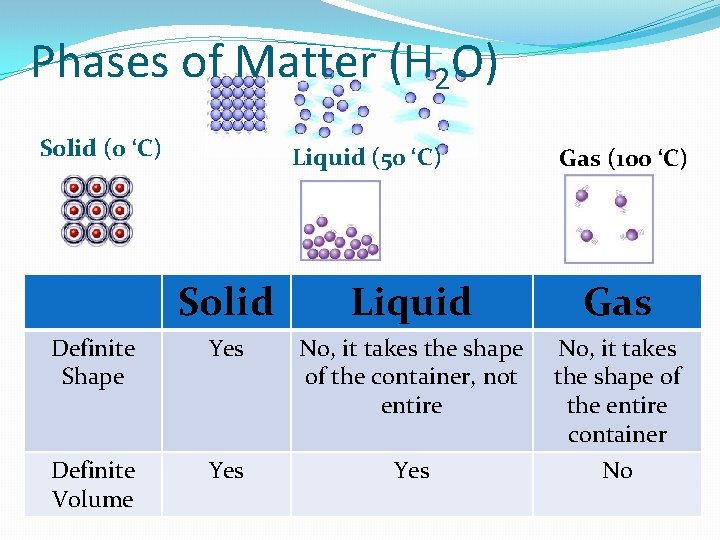
Phases of Matter (H 2 O) Solid (0 ‘C) Liquid (50 ‘C) Gas (100 ‘C) Solid Liquid Gas Definite Shape Yes No, it takes the shape of the container, not entire Definite Volume Yes No, it takes the shape of the entire container No

If you have Gas, that’s a no no !

Calibration of a Thermometer 2 Fixed Points Boiling (Condensation) Point 100 C and 373 K (AS MEASURED BY THE WATER / VAPOR EQUILIBRIUM) Melting (freezing) Point 0 C and 273 K (AS MEASURED BY THE ICE / WATER EQUILIBRIUM)
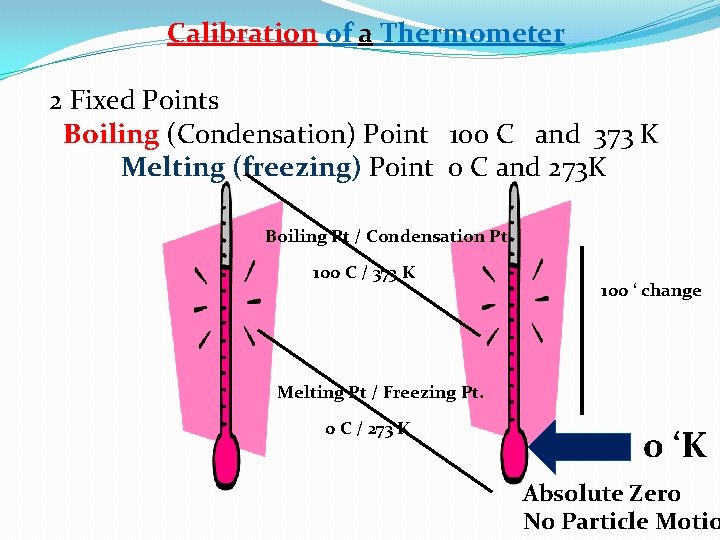
Calibration of a Thermometer 2 Fixed Points Boiling (Condensation) Point 100 C and 373 K Melting (freezing) Point 0 C and 273 K Boiling Pt / Condensation Pt 100 C / 373 K 100 ‘ change Melting Pt / Freezing Pt. 0 C / 273 K 0 ‘K Absolute Zero No Particle Motio
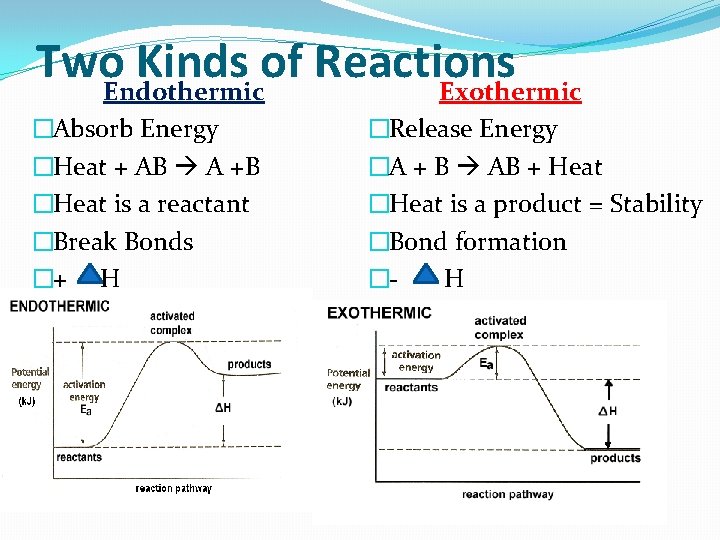
Two. Endothermic Kinds of Reactions Exothermic �Absorb Energy �Heat + AB A +B �Heat is a reactant �Break Bonds �+ H �Release Energy �A + B AB + Heat �Heat is a product = Stability �Bond formation �H
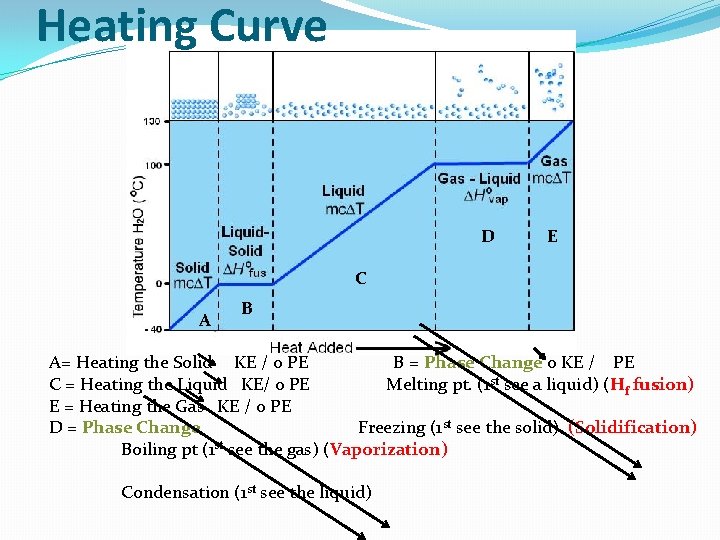
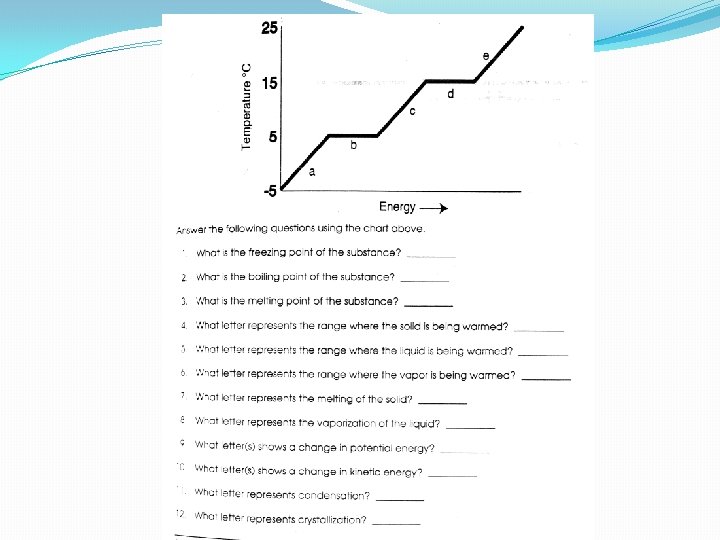
Heating Curve D E C A B A= Heating the Solid KE / 0 PE B = Phase Change 0 KE / PE C = Heating the Liquid KE/ 0 PE Melting pt. (1 st see a liquid) (Hf fusion) E = Heating the Gas KE / 0 PE D = Phase Change Freezing (1 st see the solid) (Solidification) Boiling pt (1 st see the gas) (Vaporization) Condensation (1 st see the liquid)

S Hf Heat of Fusion Solidification L HV Heat of Vaporization Condensation G Sublimation Deposition Remember, there is no increase in KE because all energy is being used for the Phase change!
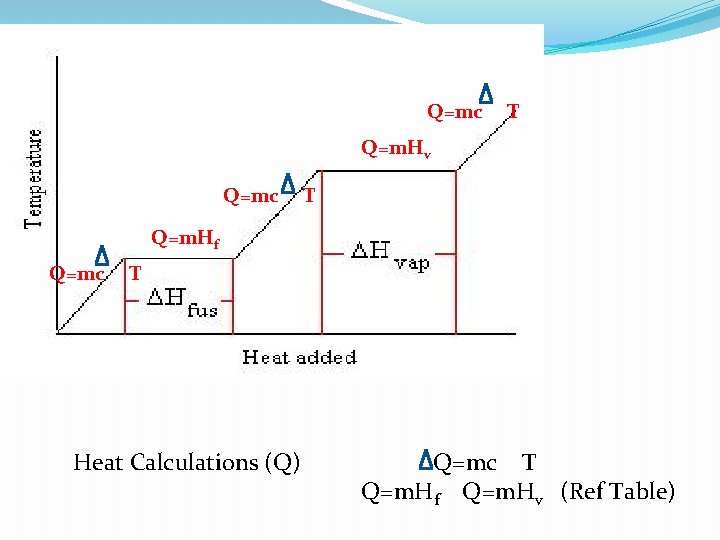
Q=mc T Q=m. Hv Q=mc T Q=m. Hf Q=mc T Heat Calculations (Q) Q=mc T Q=m. Hf Q=m. Hv (Ref Table)

C = Specific heat capacity This is the amount of heat needed to raise the temperature of 1 g of water by 1 ‘C
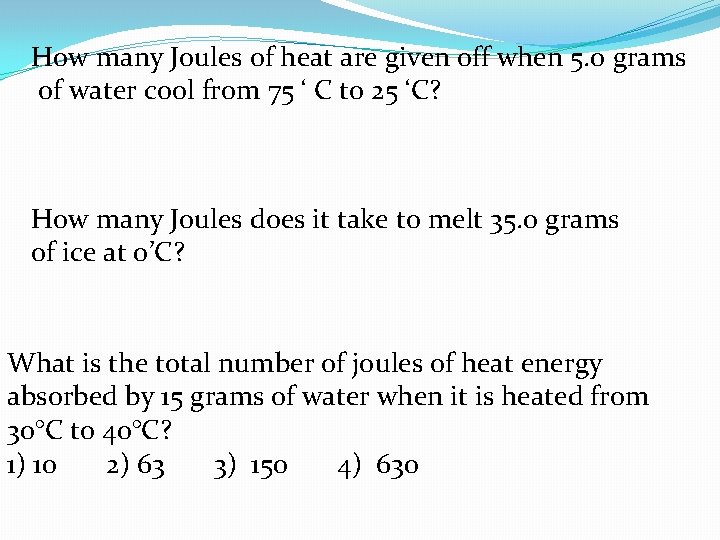
How many Joules of heat are given off when 5. 0 grams of water cool from 75 ‘ C to 25 ‘C? How many Joules does it take to melt 35. 0 grams of ice at 0’C? What is the total number of joules of heat energy absorbed by 15 grams of water when it is heated from 30°C to 40°C? 1) 10 2) 63 3) 150 4) 630

Vapor Pressure Is the amount of pressure that a vapor Exerts on the walls of a sealed container. STP = Standard Temperature Pressure 0 ‘C 1 atm 273 ‘K 101. 3 kpa When Vapor Pressure = atmospheric pressure BOILING OCCURS ****The LOWER the boiling point, the HIGHER the vapor pressure!!!
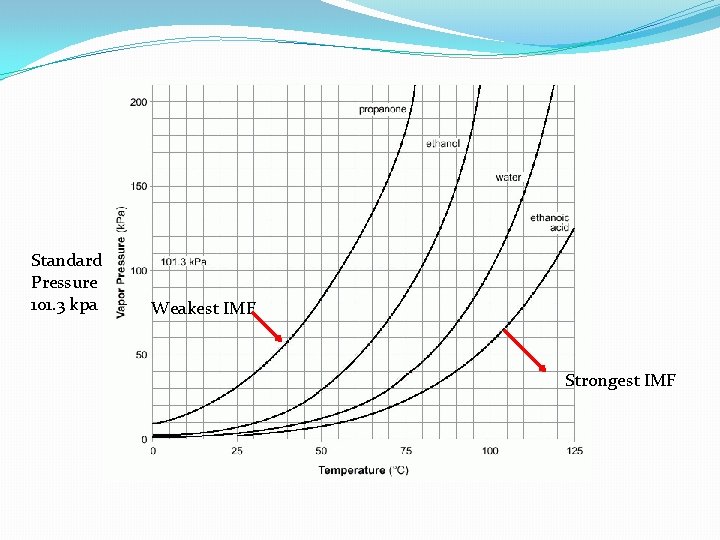
Standard Pressure 101. 3 kpa Weakest IMF Strongest IMF

Gases: Ideal vs Real Kinetic Molecular Theory �Gases travel in straight line, random motion �When they collide, transfer energy between particles �Collisions are elastic �Actual Volume of a gas is negligible (small) as compared to the volume they occupy �There is no attraction between gas particles �These describe the characteristics of an IDEAL GAS Real gas �All characteristics are the same except �No Elastic Collisions �There is a slight IMF of attraction between gas particles (VDW)

So, How can we make: 1. a real gas act like an ideal gas 2. an ideal gas act like a real gas What conditions of temperature and pressure favor these gases? What is your IDEAL vacation? Watch this!

CO 2 CO 2

Gas Laws

Boyles Law (BLT) 1. At constant Temperature 2. An increase in Pressure will decrease the volume 3. Inverse relationship 4. P 1 P 2 = V 2 V 1 5. PV = K (constant)

Charle’s Law (CLP) 1. At constant Pressure 2. If you inc. T, you inc. V 3. temp is in degrees KELVIN 4. Direct relationship V 1 = T 1 V 2 T 2 0 K = 0 C + 273

Guy Lussac’s Law (GLV) 1. Volume is Constant 2. Inc in Temperature will Inc Pressure 3. Direct Relationship 4. T 1 = P 1 T 2 P 2

COMBINED GAS LAW 1. No Constants 2. Temperature in Kelvin

Can I make a suggestion? V 1 P 1 T 2 = V 2 P 2 T 1 Now, you are always just one step away from solving any gas law problem!
Periodic table

History and facts �First developed by Mendeleev �Later developed by Henry Mosley �Arranged the periodic table based on Atomic Number
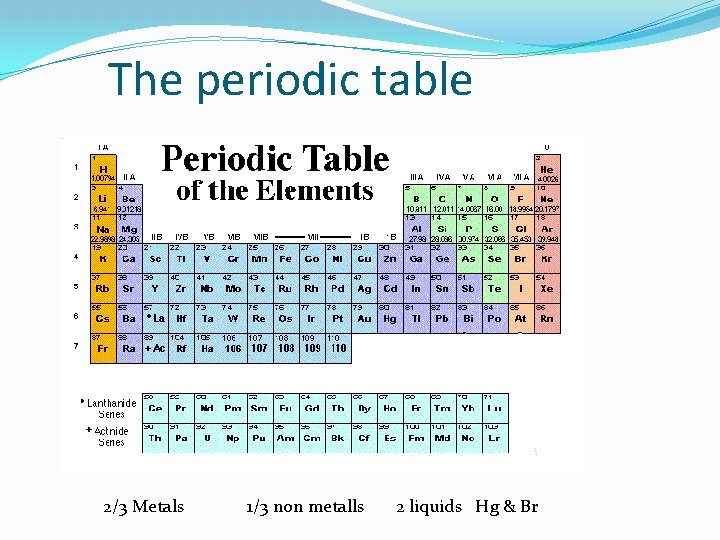
The periodic table 2/3 Metals 1/3 non metalls 2 liquids Hg & Br
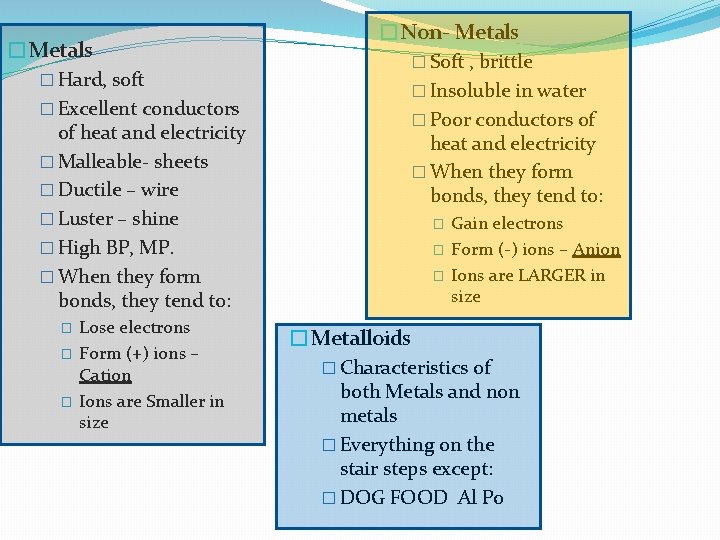
�Metals � Hard, soft � Excellent conductors of heat and electricity � Malleable- sheets � Ductile – wire � Luster – shine � High BP, MP. � When they form bonds, they tend to: � � � Lose electrons Form (+) ions – Cation Ions are Smaller in size �Non- Metals � Soft , brittle � Insoluble in water � Poor conductors of heat and electricity � When they form bonds, they tend to: � � � Gain electrons Form (-) ions – Anion Ions are LARGER in size �Metalloids � Characteristics of both Metals and non metals � Everything on the stair steps except: � DOG FOOD Al Po

Periods Groups • Run up and down • Run from left to right across the p. t. • Periods = energy levels • You see the greatest differences among the elements as you move across the periods • Group numbers = # of val e • (true for grps 1, 2, 13 -18) • **You see the greatest similarities within a group (due to the same of Val e • Also see the fastest change from NM-M (grps 13 -16)

groups 1. Alkali Metals a) Most reactive metals b) +1 oxidation number c) Form oxides with the general formula of M 2 O 2. Alkaline Earth Metal a) Reactive, not like Group 1 b) +2 oxidation number c) Form oxides with the general formula MO 3 – 12. Transitional Elements a) Fill multiple energy levels at a time b) *** Always yield pretty colors 13 -16. Metalloids a) Characteristics of metals and non- metals b) Form oxides with the general formula M 2 O 3
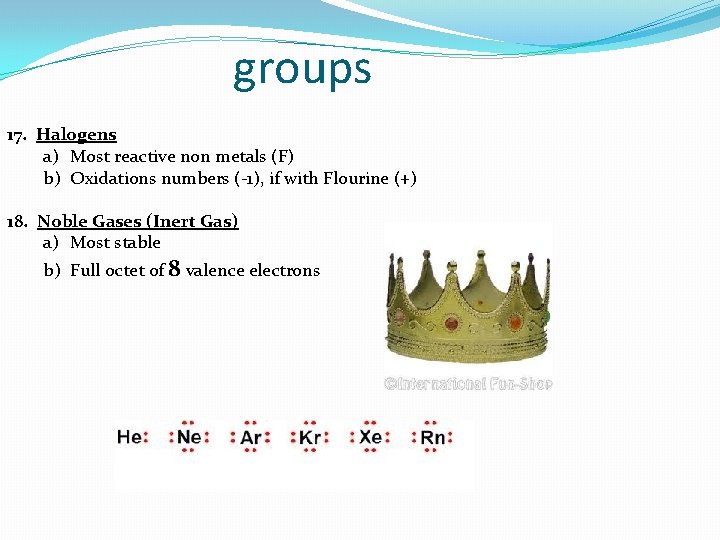
groups 17. Halogens a) Most reactive non metals (F) b) Oxidations numbers (-1), if with Flourine (+) 18. Noble Gases (Inert Gas) a) Most stable b) Full octet of 8 valence electrons

Trends of the periodic table *** remember this !!! ( M E R I ) M = Metallic Character E = Electronegativity R = Atomic Radii I = Ionization Energy

Trends of the periodic table *** remember this !!! ( M E R I )
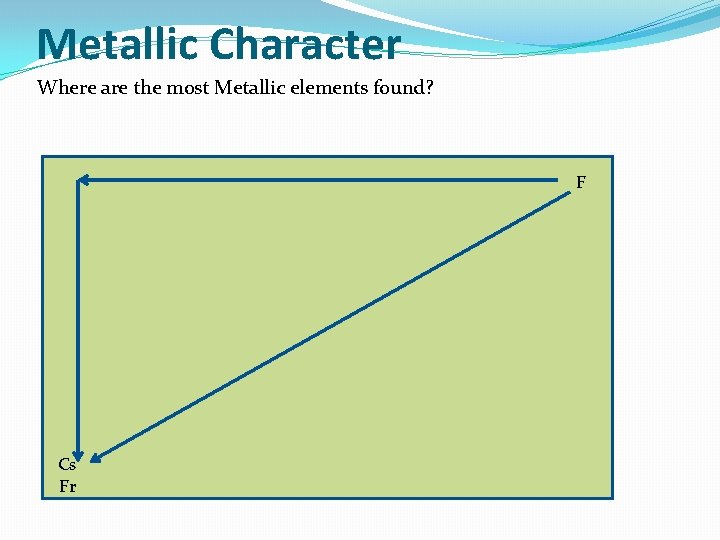
Metallic Character Where are the most Metallic elements found? F Cs Fr

Electronegativity 1. 2. 3. Where are the most Non-Metallic elements found? Def. The love or affinity for electrons (attraction of electrons to a nucleus) Based on a scale (Fr) 0. 9 – 4. 0 (F) F Cs Fr
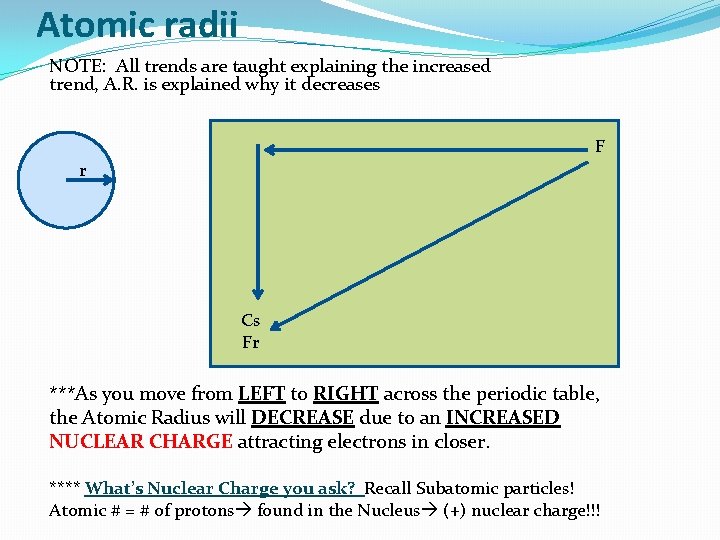
Atomic radii NOTE: All trends are taught explaining the increased trend, A. R. is explained why it decreases F r Cs Fr ***As you move from LEFT to RIGHT across the periodic table, the Atomic Radius will DECREASE due to an INCREASED NUCLEAR CHARGE attracting electrons in closer. **** What’s Nuclear Charge you ask? Recall Subatomic particles! Atomic # = # of protons found in the Nucleus (+) nuclear charge!!!
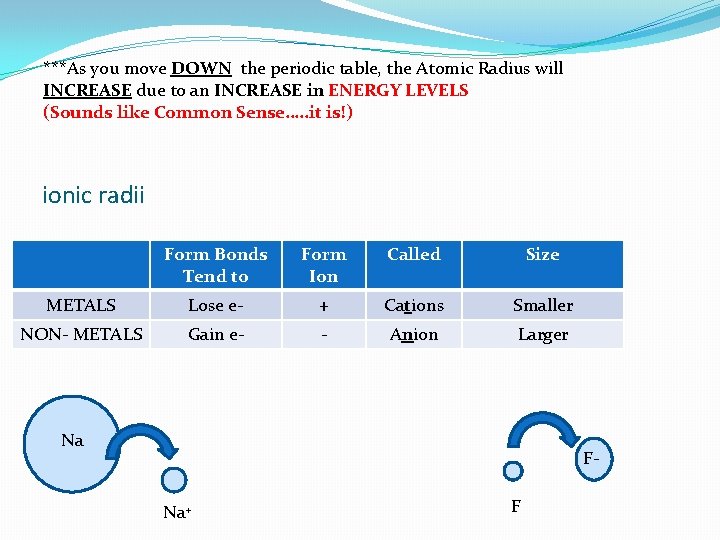
***As you move DOWN the periodic table, the Atomic Radius will INCREASE due to an INCREASE in ENERGY LEVELS (Sounds like Common Sense…. . it is!) ionic radii Form Bonds Tend to Form Ion Called Size METALS Lose e- + Cations Smaller NON- METALS Gain e- - Anion Larger Na FNa+ F
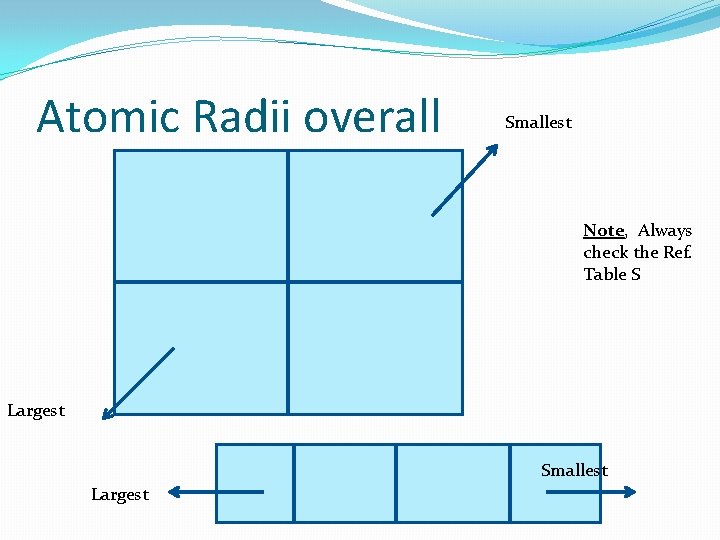
Atomic Radii overall Smallest Note, Always check the Ref. Table S Largest Smallest Largest

Ionization energy The amount of energy required to remove the outer most (valence) electron, results in the formation of an ion. Where are the highest I. E. ’s Found? Which is the smallest? Most Non-Metallic? Na 0 + Energy Na+ + e. F ionization energy Non-Metals: Do not like to lose electrons Smallest/ Tighter hold of electrons Cs Fr
Bonding
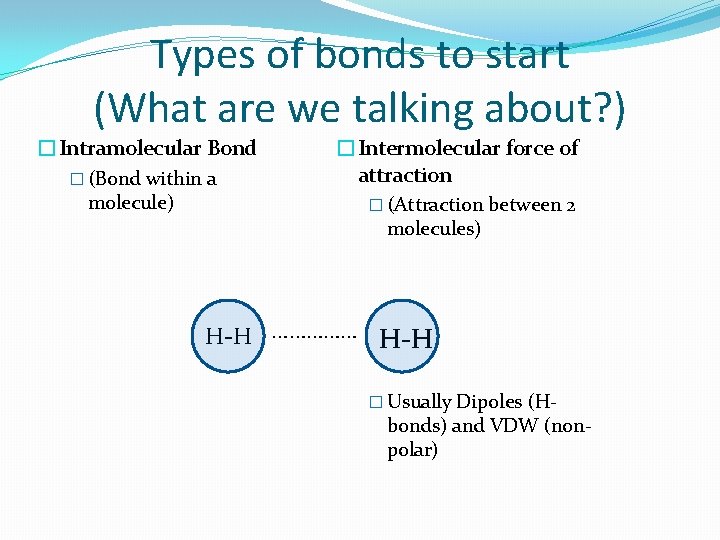
Types of bonds to start (What are we talking about? ) �Intramolecular Bond � (Bond within a molecule) �Intermolecular force of attraction � (Attraction between 2 molecules) H-H …………… H-H � Usually Dipoles (H- bonds) and VDW (nonpolar)

2 types of Bonds Covalent � A bond between 2 elements (Nm-Nm) �Involves the sharing of electrons �E. N. difference < 1. 7 Ionic �Bond between 2 elements (M-Nm) �Involves the transfer of electrons �E. N. Difference 1. 7 <

Covalent Bond characteristics Characteristics �Weaker �Low B. P. , High Vapor Pressure �Soft, Brittle �Insoluble in water Ionic �Stronger �High B. P. , Low Vapor Pressure �Hard �Crystals �Soluble

Lewis Dots 1. Show only the valence e-, (not Kernel e-) 2. The goal with bonding is to achieve an outer valence of 8 Val e- (like a noble gas) 3. ***To achieve a noble gas configuration*** a) (Rule of Octet) Metals (Grp 13) (Grp 14) NM Lose e. Gain e. Take on Noble Gas Configuration of the Noble Gas Before it! After it!
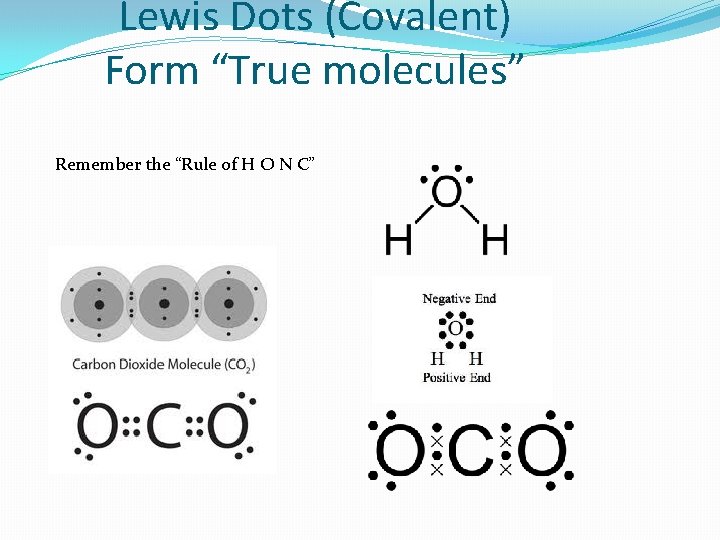
Lewis Dots (Covalent) Form “True molecules” Remember the “Rule of H O N C”
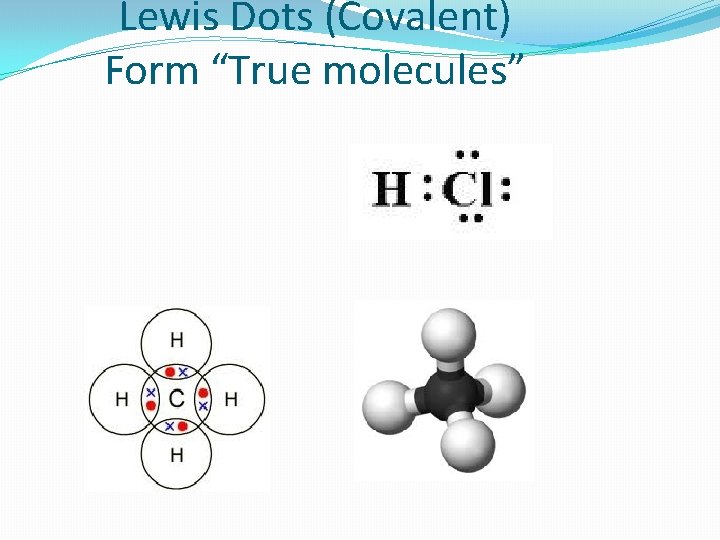
Lewis Dots (Covalent) Form “True molecules”
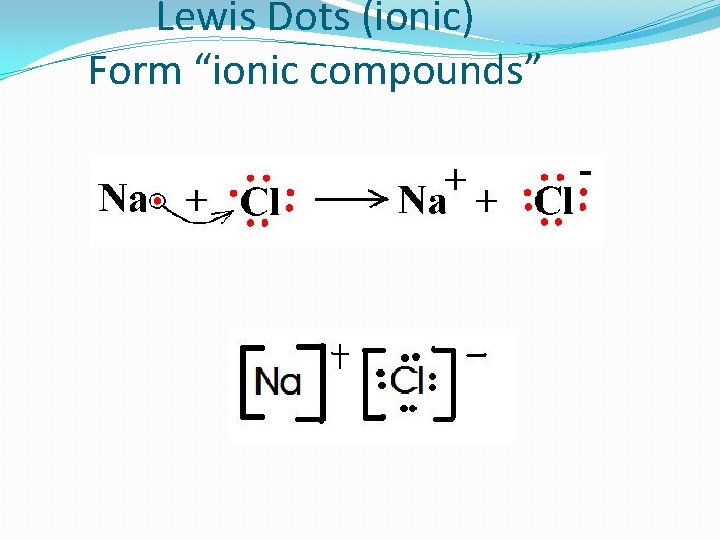
Lewis Dots (ionic) Form “ionic compounds”
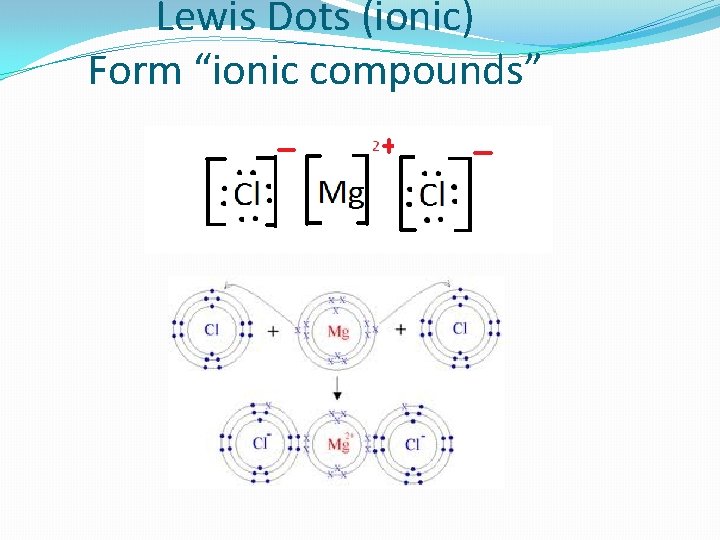
Lewis Dots (ionic) Form “ionic compounds”
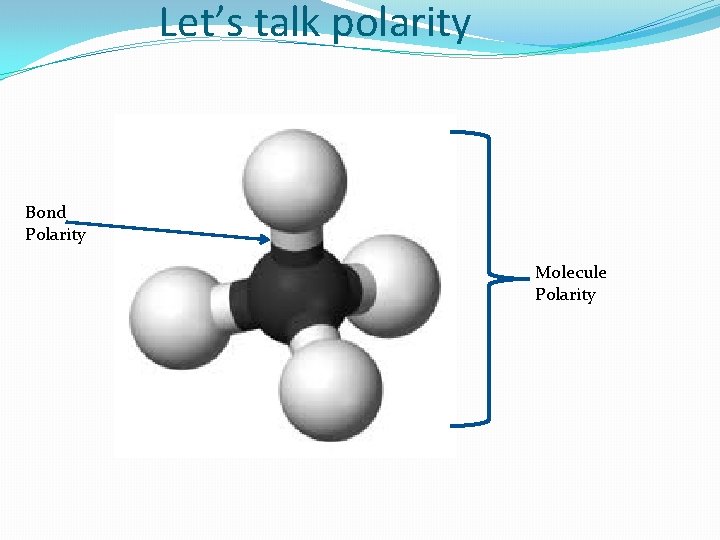
Let’s talk polarity Bond Polarity Molecule Polarity
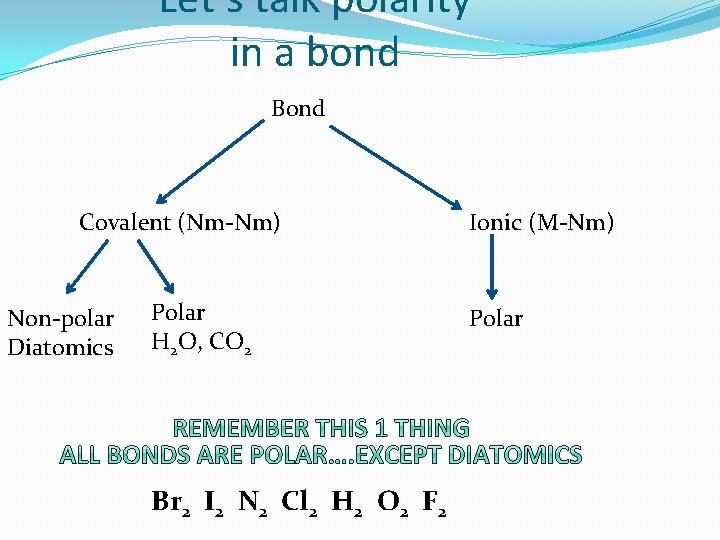
Let’s talk polarity in a bond Bond Covalent (Nm-Nm) Non-polar Diatomics Polar H 2 O, CO 2 Br 2 I 2 N 2 Cl 2 H 2 O 2 F 2 Ionic (M-Nm) Polar
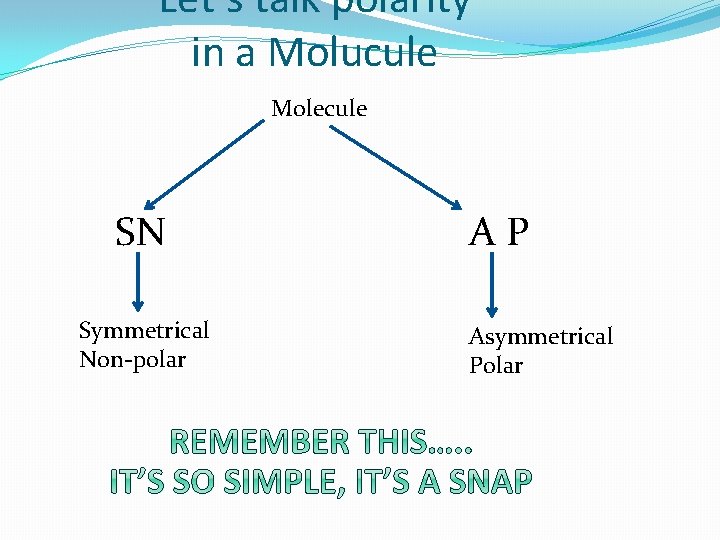
Let’s talk polarity in a Molucule Molecule SN Symmetrical Non-polar AP Asymmetrical Polar
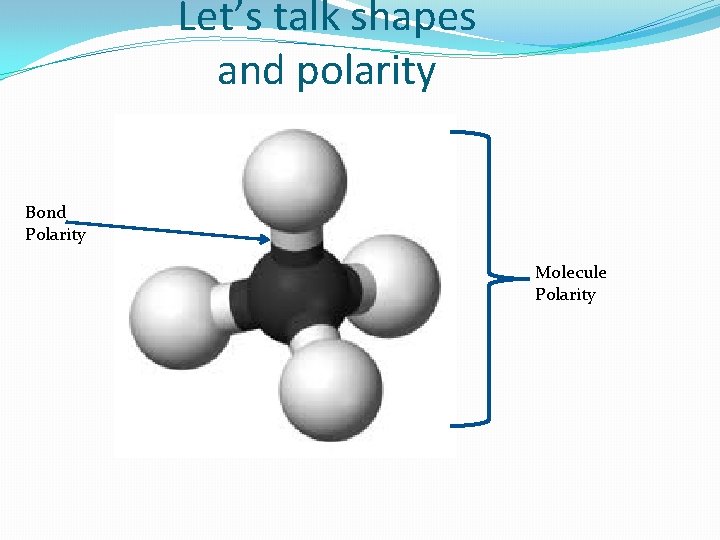
Let’s talk shapes and polarity Bond Polarity Molecule Polarity
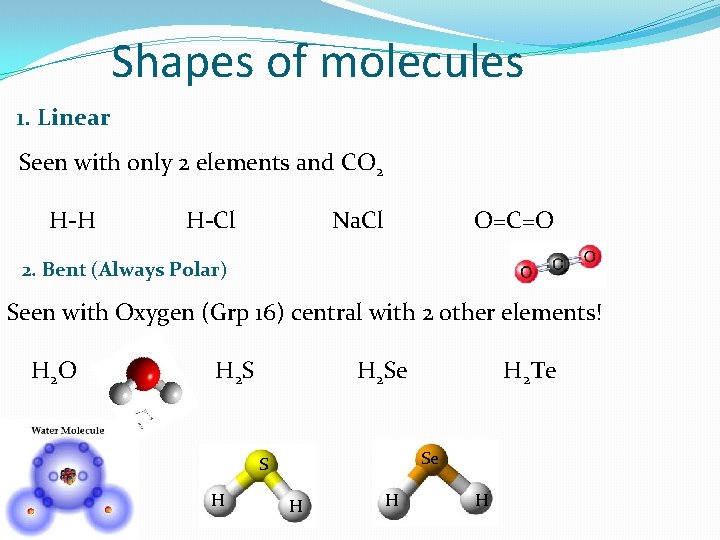
Shapes of molecules 1. Linear Seen with only 2 elements and CO 2 H-H H-Cl Na. Cl O=C=O 2. Bent (Always Polar) Seen with Oxygen (Grp 16) central with 2 other elements! H 2 O H 2 Se Se S H H 2 Te H H H
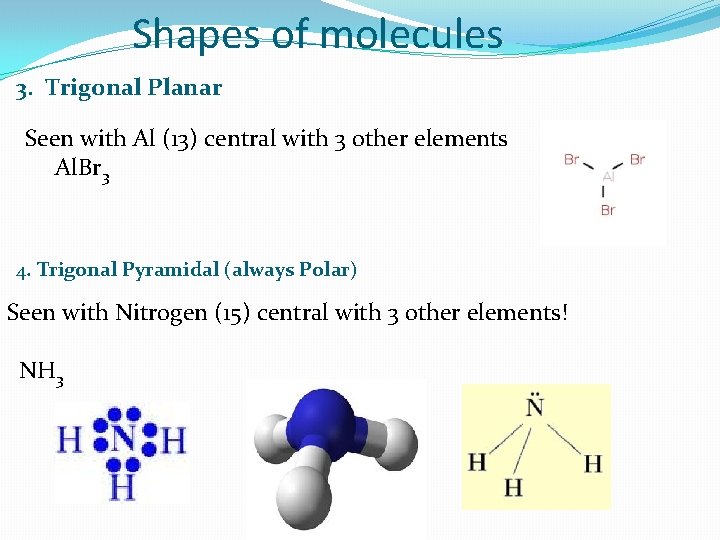
Shapes of molecules 3. Trigonal Planar Seen with Al (13) central with 3 other elements Al. Br 3 4. Trigonal Pyramidal (always Polar) Seen with Nitrogen (15) central with 3 other elements! NH 3
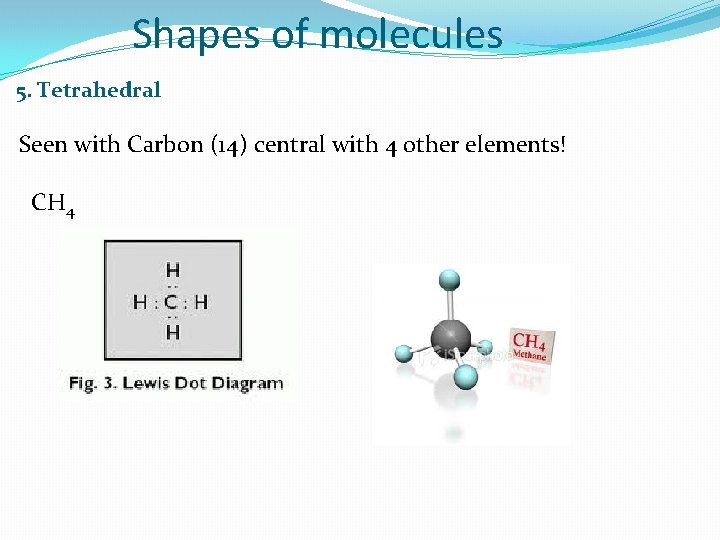
Shapes of molecules 5. Tetrahedral Seen with Carbon (14) central with 4 other elements! CH 4
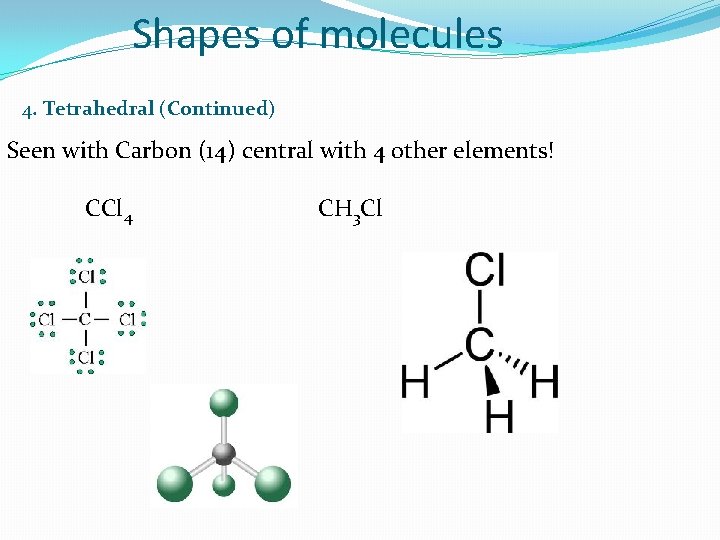
Shapes of molecules 4. Tetrahedral (Continued) Seen with Carbon (14) central with 4 other elements! CCl 4 CH 3 Cl

6 kinds of forces of attraction 4 - intramolecular (with-in) 2 - intermolecular (IMF) Name 1. Network Solids Examples Diamond, Graphite Si. C Silicon Carbide Si. O 2 Silicon Dioxide Characteristics Strongest, Hardest ^^^^^ BP, MP Insoluble in water Poor conductor of electricity
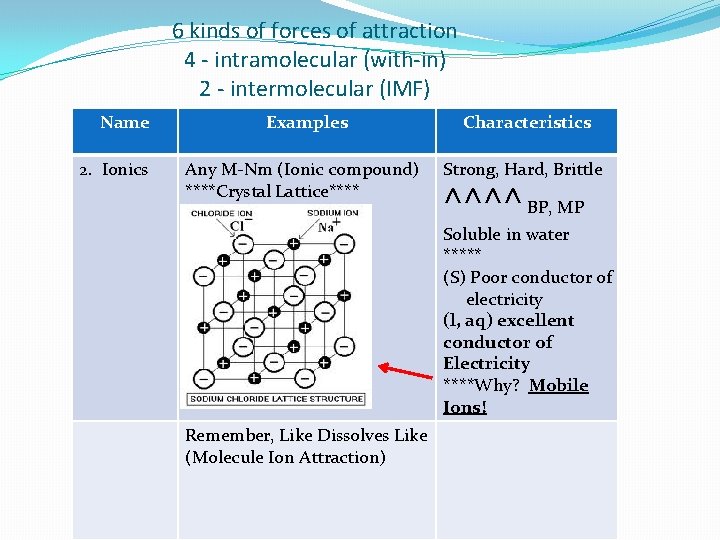
6 kinds of forces of attraction 4 - intramolecular (with-in) 2 - intermolecular (IMF) Name 2. Ionics Examples Characteristics Any M-Nm (Ionic compound) ****Crystal Lattice**** Strong, Hard, Brittle ^^^^ BP, MP Soluble in water ***** (S) Poor conductor of electricity (l, aq) excellent conductor of Electricity ****Why? Mobile Ions! Remember, Like Dissolves Like (Molecule Ion Attraction)
Name Example Characteristic 2. Ionic continued Remember, Like Dissolves Like (Molecule Ion Attraction) This is why salts (ionicpolar) dissolve in water (polar)
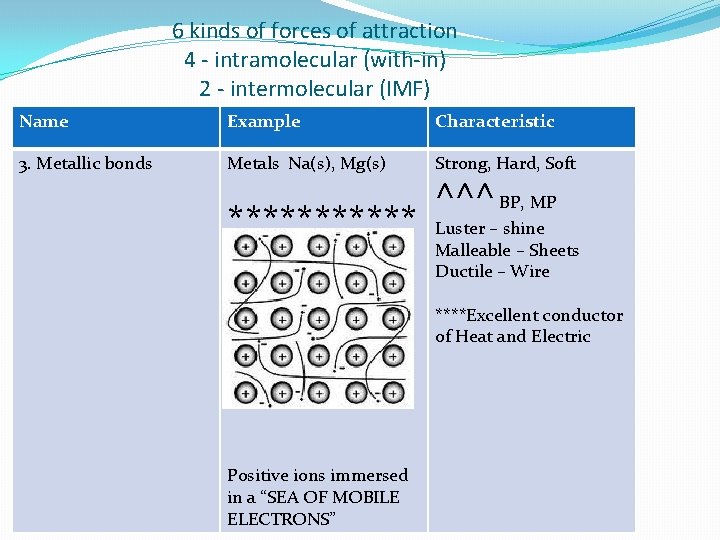
6 kinds of forces of attraction 4 - intramolecular (with-in) 2 - intermolecular (IMF) Name Example Characteristic 3. Metallic bonds Metals Na(s), Mg(s) Strong, Hard, Soft ****** ** Luster – shine Malleable – Sheets Ductile – Wire ^^^ BP, MP ****Excellent conductor of Heat and Electric Positive ions immersed in a “SEA OF MOBILE ELECTRONS”
6 kinds of forces of attraction 4 - intramolecular (with-in) 2 - intermolecular (IMF) Name Example Characteristic 4. Molecular Compounds All covalent Molecules Soft, Brittle ^^ B. P. , M. P. Insoluble in water Poor conductor of electricity
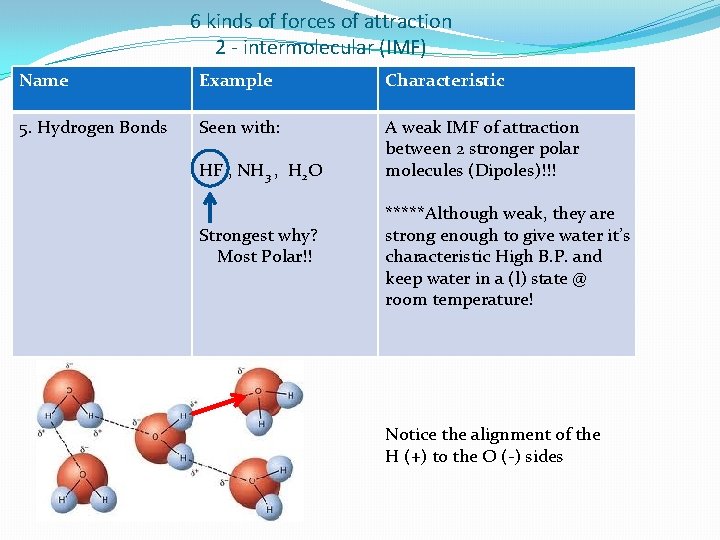
6 kinds of forces of attraction 2 - intermolecular (IMF) Name Example Characteristic 5. Hydrogen Bonds Seen with: A weak IMF of attraction between 2 stronger polar molecules (Dipoles)!!! HF , NH 3 , H 2 O Strongest why? Most Polar!! *****Although weak, they are strong enough to give water it’s characteristic High B. P. and keep water in a (l) state @ room temperature! Notice the alignment of the H (+) to the O (-) sides

6 kinds of forces of attraction 2 - intermolecular (IMF) Name Example Characteristic 5. Van der Waals “London disperssion forces” Seen with: (diatomics) Weakest IMF of attraction between 2 non-polar molecules or a very weak polar molecule (dipole-dipole) HCl. F 2(g), Cl 2 (g) , Br 2(l) I 2(s) , H 2(g), N 2(g) And CO 2 Therefore, usually seen with Diatomic gases only! Active during deposition (G S) strongest based on size (note Solid Broken during Sublimation therefore it is the (S G) strongest example)
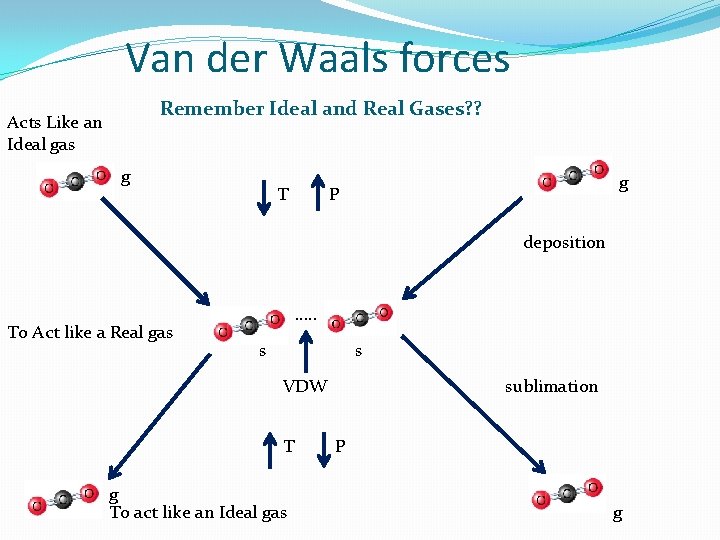
Van der Waals forces Remember Ideal and Real Gases? ? Acts Like an Ideal gas g T g P deposition To Act like a Real gas …. . s s VDW T g To act like an Ideal gas sublimation P g
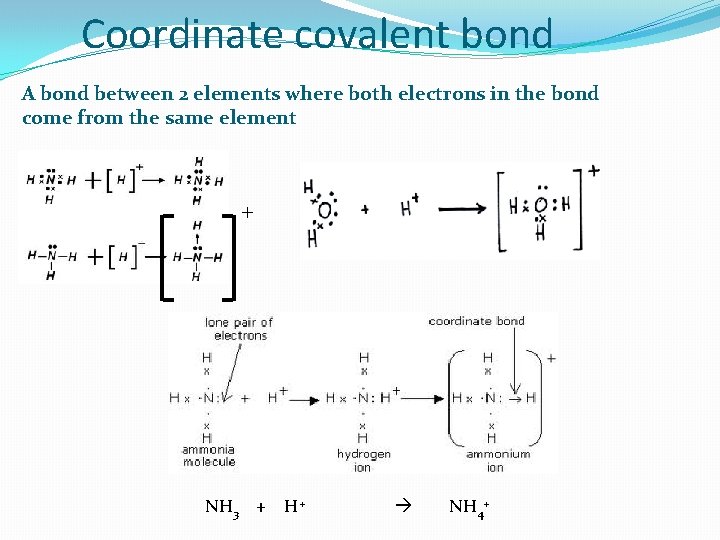
Coordinate covalent bond A bond between 2 elements where both electrons in the bond come from the same element + NH 3 + H+ NH 4+

Solutions

Mixtures (Varied Ratio) Homogeneous Heterogeneous � True Solutions (Soluble) � Solubility – Ability to dissolve in solution � (aq) See only 1 part � Separated by evaporation � Contains 2 Parts �See Multiple parts �Sand/ water, Dirt, Rocky road ice cream �Separated by filtration Solute – The substance that is being dissolved 2. Solvent – What is doing the dissolving (Water) 1.

Factors that effect Solubility PANT 1. Pressure (g only) - P Solubility 2. Agitation – stirring (s) will Solubility 3. Nature of Solvent /solute 1. “Like dissolves like” 1. 2. Water (polar) will dissolve Salts (Ionicpolar) Water (Polar) does not dissolve fats –oils (non-polar) 4. Temperature - T T Solubility (solids only) Solubility (Gas…think soda)
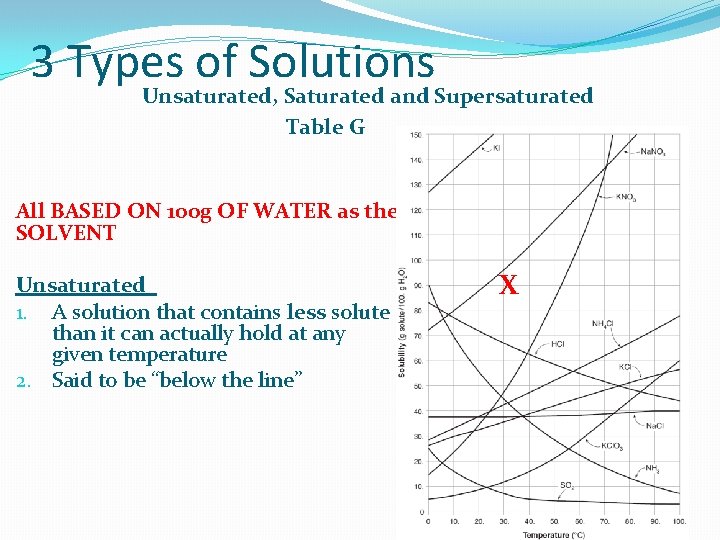
3 Types of Solutions Unsaturated, Saturated and Supersaturated Table G All BASED ON 100 g OF WATER as the SOLVENT Unsaturated 1. A solution that contains less solute than it can actually hold at any given temperature 2. Said to be “below the line” X
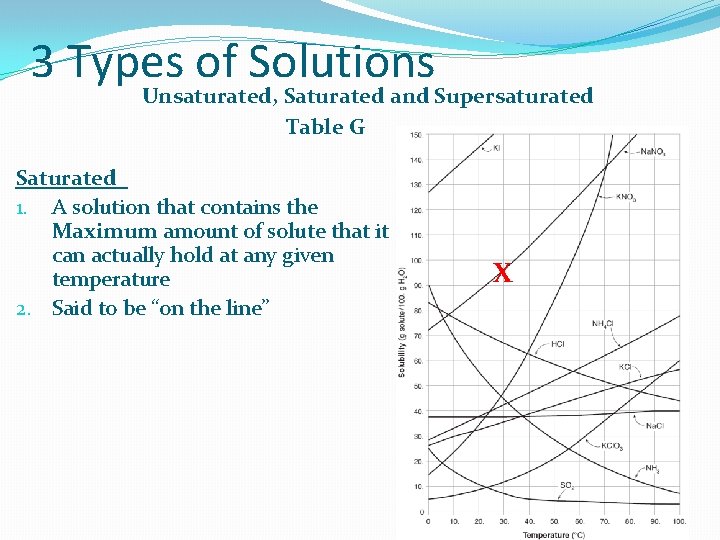
3 Types of Solutions Unsaturated, Saturated and Supersaturated Table G Saturated 1. A solution that contains the Maximum amount of solute that it can actually hold at any given temperature 2. Said to be “on the line” X
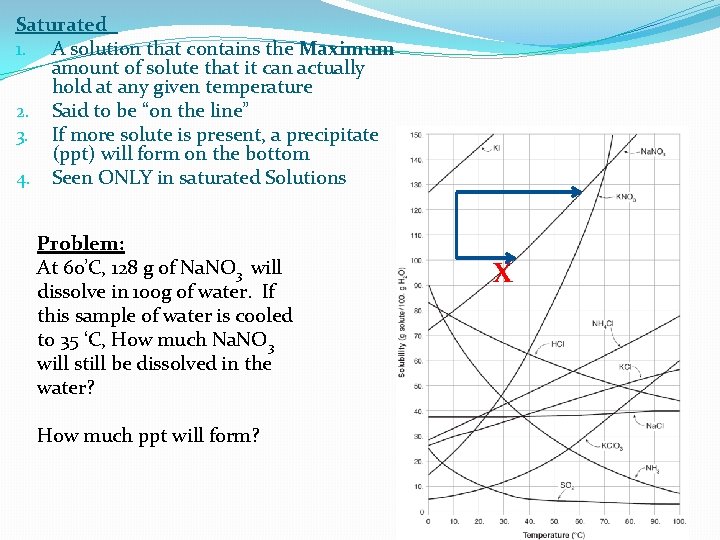
Saturated 1. A solution that contains the Maximum amount of solute that it can actually hold at any given temperature 2. Said to be “on the line” 3. If more solute is present, a precipitate (ppt) will form on the bottom 4. Seen ONLY in saturated Solutions Problem: At 60’C, 128 g of Na. NO 3 will dissolve in 100 g of water. If this sample of water is cooled to 35 ‘C, How much Na. NO 3 will still be dissolved in the water? How much ppt will form? X

ppt
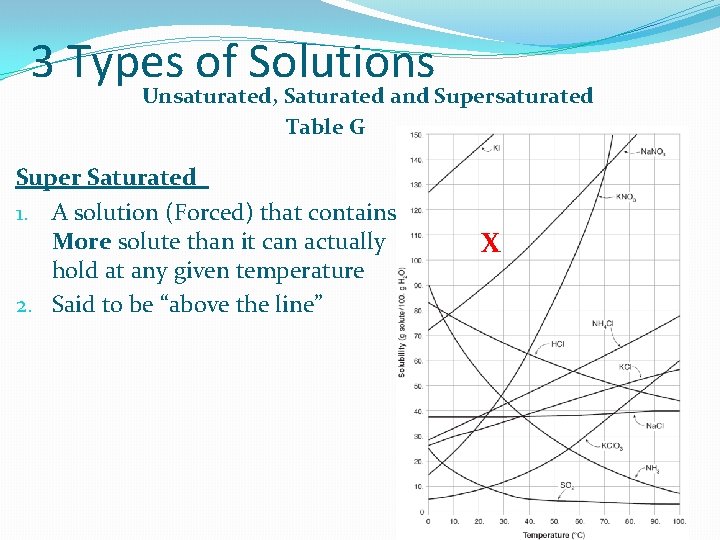
3 Types of Solutions Unsaturated, Saturated and Supersaturated Table G Super Saturated 1. A solution (Forced) that contains More solute than it can actually hold at any given temperature 2. Said to be “above the line” X

Making a Super Saturated Solution
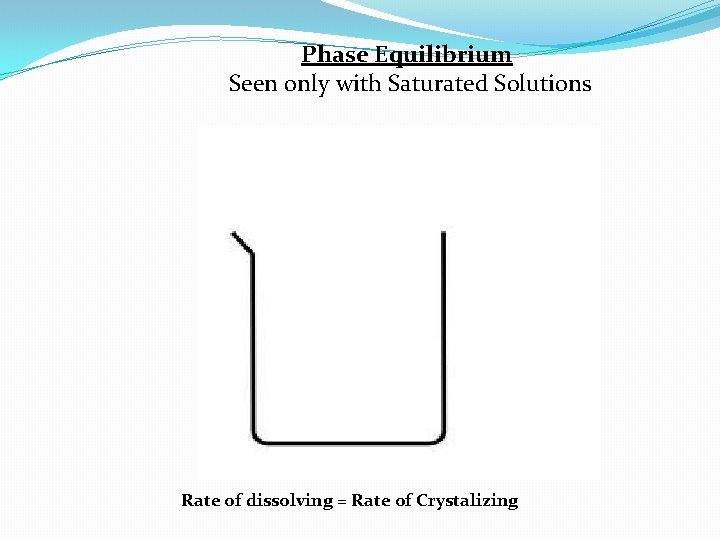
Phase Equilibrium Seen only with Saturated Solutions Rate of dissolving = Rate of Crystalizing

Phase Equilibrium Seen only with Saturated Solutions
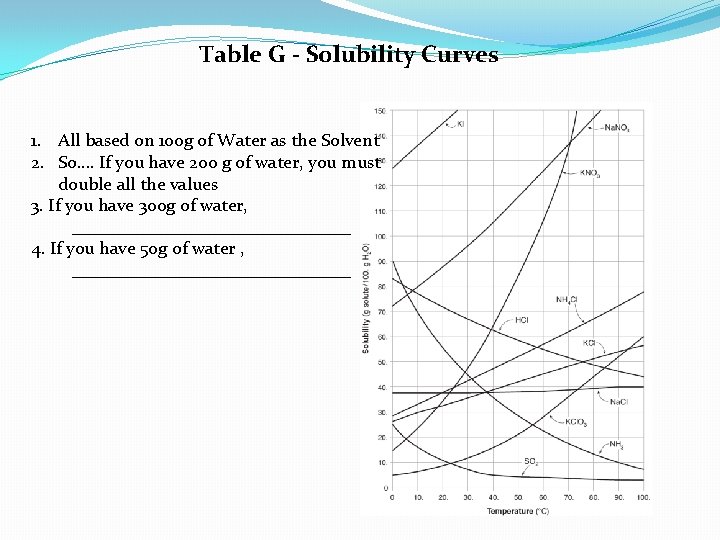
Table G - Solubility Curves 1. All based on 100 g of Water as the Solvent 2. So…. If you have 200 g of water, you must double all the values 3. If you have 300 g of water, ________________ 4. If you have 50 g of water , ________________
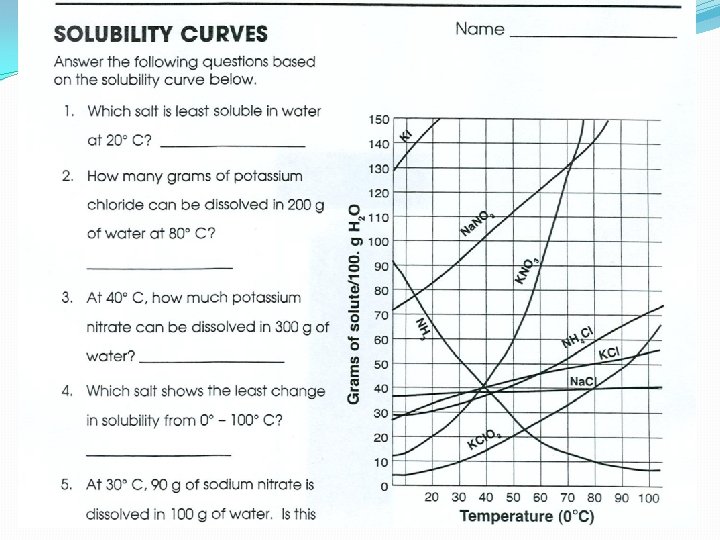


Concentration 1. Measured by Molarity 1 Gram of H 2 O = 1 ml 1 L = 1000 ml Parts Per Million (ppm) = Mass of solute Mass of Solution x 1, 000

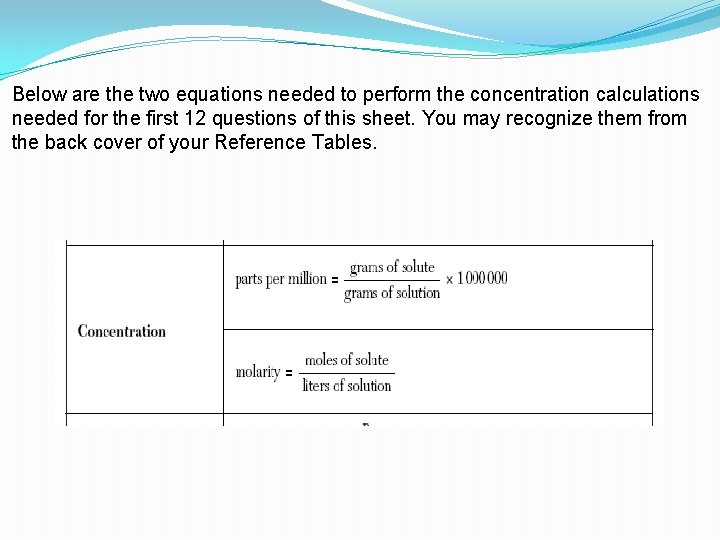
Below are the two equations needed to perform the concentration calculations needed for the first 12 questions of this sheet. You may recognize them from the back cover of your Reference Tables.

What is the molarity of a solution which has a total volume of 100 m. L and contains 3 moles of Na. Cl? What is the molarity of a solution that contains 1. 5 moles of Na. NO 3 in 2. 5 Liters of solution? A solution is made that contains 1. 25 moles of sugar in 175 m. L of solution. What is the molarity of the solution?

A 4 molar solution of ammonium chloride will contain how many moles of solute in 100 m. L of solution? Given a 250 m. L sample of a solution that is 0. 25 molar, how many moles of the solute do you possess?

How many moles of KNO 3 are needed to make 0. 750 L of a 3. 25 M solution? What volume of solution, measured in liters, is necessary to obtain 1 mole of solute if the concentration of the solution is 1. 25 M?

If a sample of solution contains 1. 5 moles of sodium hydroxide, and the concentration of the solution is 0. 8 M, then what volume of the solution do you possess? Given a 1. 4 M Na. Cl solution, 0. 90 moles of sodium chloride would be contained in how many milliliters of the solution? If you have made a 455 ppm solution, how many grams of sodium chloride are in 1000 grams of the solution?

When 0. 0043 grams of O 2 is dissolved in 100 m. L of water, what is the concentration of the solution, measured in ppm? A substance has a solubility of 350 ppm. How many grams of the substance are present in 1. 5 L of solution?

13. What is the percent by volume of ethanol if 50. 0 m. L of ethanol is diluted with water to form a total volume of 300. 0 m. L? 14. How many milliliters of a liquid were used to make a 13% solution with a total volume of 80 m. L? 15. What volume of a 35% solution of alcohol can be made using 20 m. L of the solute? 16) A rock contains 15 grams of calcium carbonate and 85 grams of other substances, what is the composition of the rock as measured in percent by mass?

Molarity by Dilution 1. Done using the Titration equation but make a change! M 1 V 1 = M 2 V 2



Electrolytes �Substances that when put into solution, will dissociate into their component ions �DISSOCIATION – the ability to break up into their component ions in solution �It is the “Mobile Ions” that give the solution the ability to conduct electricity �Ionics (Polar Substances) have this ability �Good Electrolytes (Salts, Acids and Bases) �Non-electrolytes (covalent compounds)
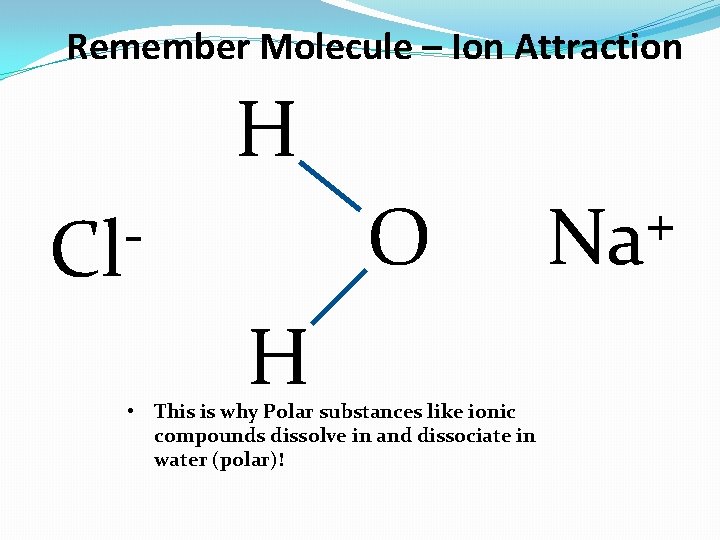
Remember Molecule – Ion Attraction H O Cl H • This is why Polar substances like ionic compounds dissolve in and dissociate in water (polar)! + Na

What other type of attraction do you see at work here?

Electrolytes (Reminder) �Substances that when put into solution, will dissociate in solution �DISSOCIATION – the ability to break up into their component ions in solution �It is the “Mobile Ions” that give the solution the ability to conduct electricity �Ionic salts (Polar Substances) have this ability along with Acids and bases �Good Electrolytes (Salts, Acids and Bases) �Non-electrolytes (covalent compounds)

Colligative Properties Are properties that are effected by the amount of dissolved particles in solution! Boiling Point Freezing (Melting) point �Always will be an elevation (raising) �Elevates by 0. 52’C per mole of dissolved ions (substance) �Always be a depression(lowering) �Lowers by 1. 86’C per mole of dissolved ions(substance) ****These are not so important. What is important is being able to identify which substance will have the greatest effect (or least) on the Colligative Properties

Acids, Bases and Salts �Acids �See H first! �HCl, H 2 SO 4 �Also �CH 3 COOH �Salts – Ionic Compounds (M-NM) �Bases �Any Metal – OH �Na. OH, Ca(OH)2 �Do NOT confuse with alcohols �CH 3 OH
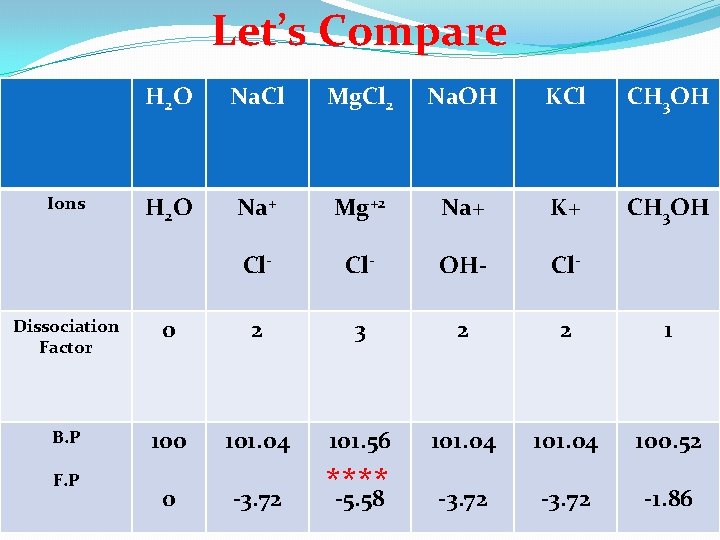
Let’s Compare Ions H 2 O Na. Cl Mg. Cl 2 Na. OH KCl CH 3 OH H 2 O Na+ Mg+2 Na+ K+ CH 3 OH Cl- OH- Cl- Dissociation Factor 0 2 3 2 2 1 B. P 100 101. 04 101. 56 101. 04 100. 52 0 -3. 72 **** -5. 58 -3. 72 -1. 86 F. P
- Slides: 99