PHYSICAL BEHAVIOR OF MATTER PBOM UNIT 6 Phases

PHYSICAL BEHAVIOR OF MATTER (PBOM) UNIT 6
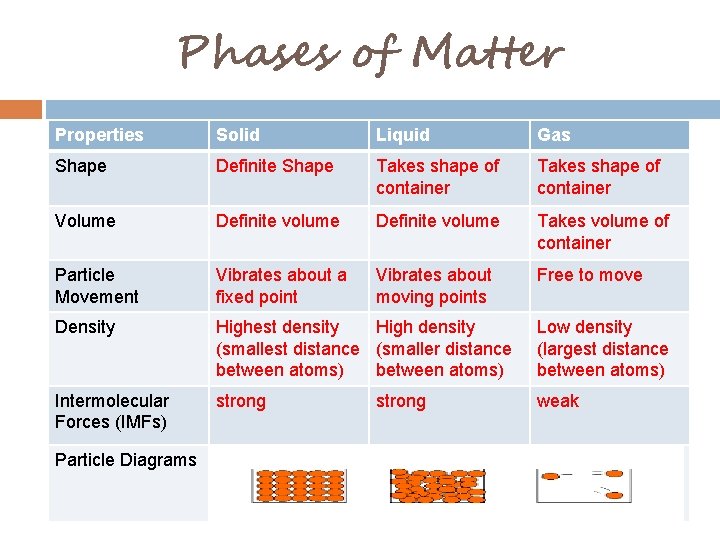
Phases of Matter Properties Solid Liquid Gas Shape Definite Shape Takes shape of container Volume Definite volume Takes volume of container Particle Movement Vibrates about a fixed point Vibrates about moving points Free to move Density Highest density High density (smallest distance (smaller distance between atoms) Low density (largest distance between atoms) Intermolecular Forces (IMFs) strong weak Particle Diagrams strong

Temperature: the measure of the AVERAGE KINETIC ENERGY of a substance’s particles v INDEPENDENT OF SAMPLE’S MASS

Temperature… Temperature is measured in: � Fahrenheit (ºF) � Celsius (ºC) Chemist use ºC and ºK � Kelvin (K) Absolute Zero -273ºC or 0 ºK; the temperature at which all PARTICLE MOTION ceases

Practice Different masses of aluminum and iron are found to have the same temperature. Compare the average kinetic energy of the aluminum atoms to the iron atoms. The aluminum and the iron have the same average kinetic energy because both samples are at the same temperature.

Practice Indicate whether the average kinetic energy of the water molecules is increasing, decreasing, or remaining the same. v H 2 O(s) changes to H 2 O(l) at 0 o. C: remains the same v H 2 O(l) changes to H 2 O(s) at 0 o. C: remains the same v H 2 O(l) at 10 o. C changes to H 2 O(l) at 20 o. C: increasing v H 2 O(l) at 20 o. C changes to H 2 O(l) at 10 o. C: decreasing

Temperature Conversion Kelvin Celsius Table T K = ºC + 273 1ºC = 1ºK It is possible to have a negative CELSIUS value, but never a negative KELVIN value!
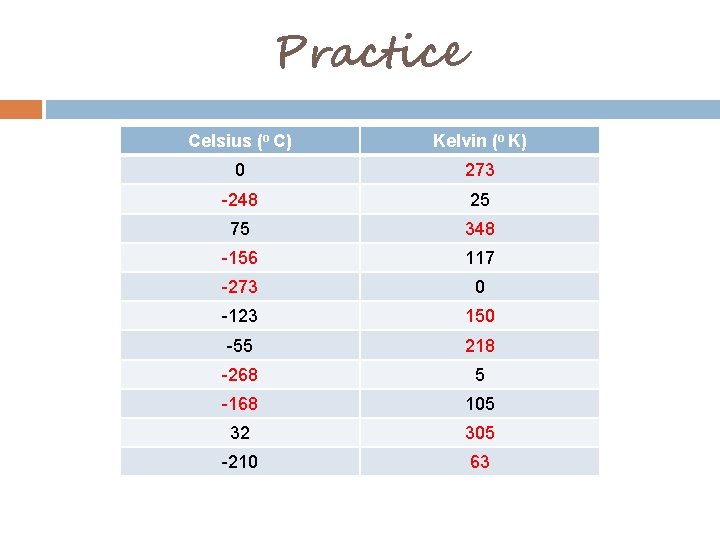
Practice Celsius (o C) Kelvin (o K) 0 273 -248 25 75 348 -156 117 -273 0 -123 150 -55 218 -268 5 -168 105 32 305 -210 63
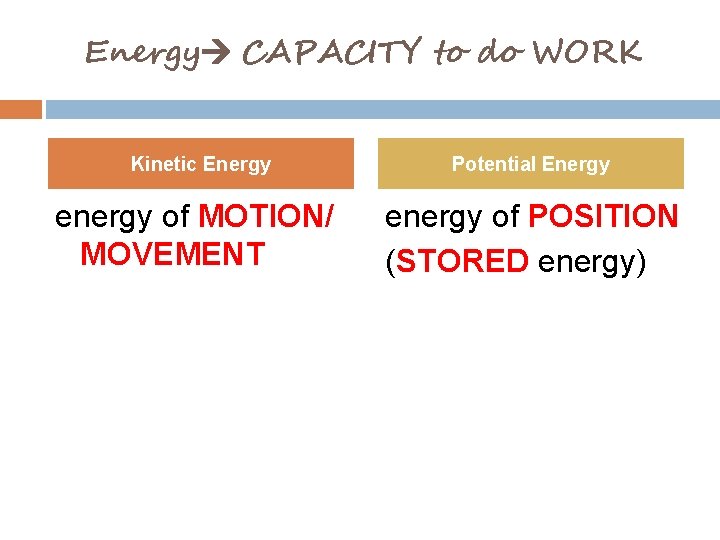
Energy CAPACITY to do WORK Kinetic Energy Potential Energy energy of MOTION/ MOVEMENT energy of POSITION (STORED energy)

Heat: quantity of ENERGY v Can ONLY be measured as it’s TRANSFERRED from a (HOTTER) object to a (COOLER) object v DEPENDENT on SAMPLE’S MASS vthe larger the sample’s mass, the more heat needed to bring it to the desired temp v Measured in JOULES (J), KILOJOULES (k. J) & CALORIES (Cal)

Practice A student is examining two samples of ice. Sample A has a mass of 10 g while sample B has a mass of 1 g. Both samples are at their freezing point. Compare the two samples in terms of heat energy. The 10 g sample of ice will require more heat.

Measurement of Heat The amount of heat LOST or GAINED in a physical or chemical reaction can be calculated using the following equation: Table T q = mc T q = heat (units = Joules or J) m = mass of sample C = specific heat of the sample (see Table B for H 2 O) T = change in temperature

Practice How many joules are absorbed when 50. 0 g of water are heated from 30. 2 o. C to 58. 6 o. C? q = mc T m=50. 0 g C=4. 18 J/g. K T =(58. 6 -30. 2) o. K=28. 4 o. K q=50. 0(4. 18)(28. 4) q=5935. 6 J or 5. 9 k. J

Practice If 100. 0 J are added to 20. 0 g of water at 30. 0 o. C, what will be the final temperature of water? q = mc T q=100. 0 J m=20. 0 g C=4. 18 J/g. K Ti=30. 0 o. C 303 o. K Tf= 100. 0=20. 0(4. 180)(Tf-303) 100. 0=83. 6(Tf-303) 1. 19=Tf-303 Tf=304. 2 o. K

Heat of Fusion (Hf): the amount of heat (or PE) required to change a substance from a solid to a liquid Table T q = m. Hf q = heat (J, k. J) m = mass of sample (g) Hf=heat of fusion

Practice What is the total number of kilojoules of heat needed to change 15. 0 g of ice to water at 0 o. C? q = m. Hf m=15. 0 g Hf=334 J/g q=15. 0(334) q=5010 J q=5. 0 k. J

Heat of Vaporization Heat of Vaporization: the amount of heat required to change a substance from a liquid to a gas Table T q = m. Hv q = heat (J, k. J) m = mass of sample (g) Hv=heat of vaporization

Practice What is the total number of kilojoules required to completely boil 100. 0 g of water at 100 o. C. q = m. Hv m=100. 0 g Hv= 2260 J /g q=100. 0(2260) q=226000 J or 226 k. J
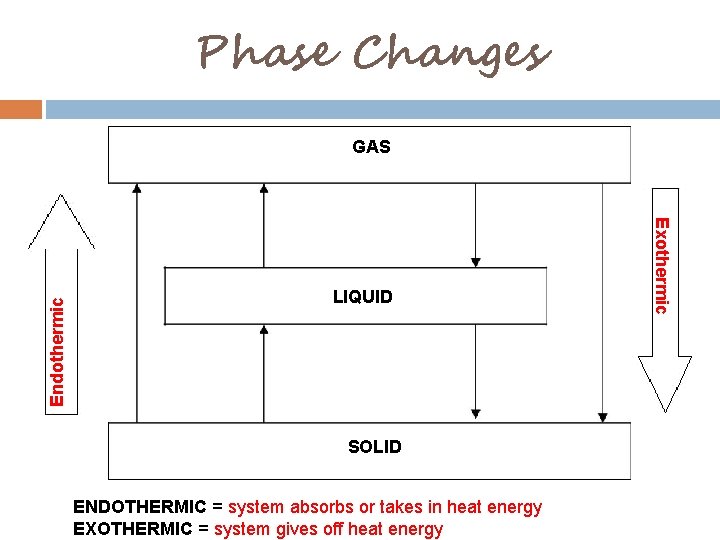
Phase Changes LIQUID SOLID ENDOTHERMIC = system absorbs or takes in heat energy EXOTHERMIC = system gives off heat energy Exothermic Endothermic GAS
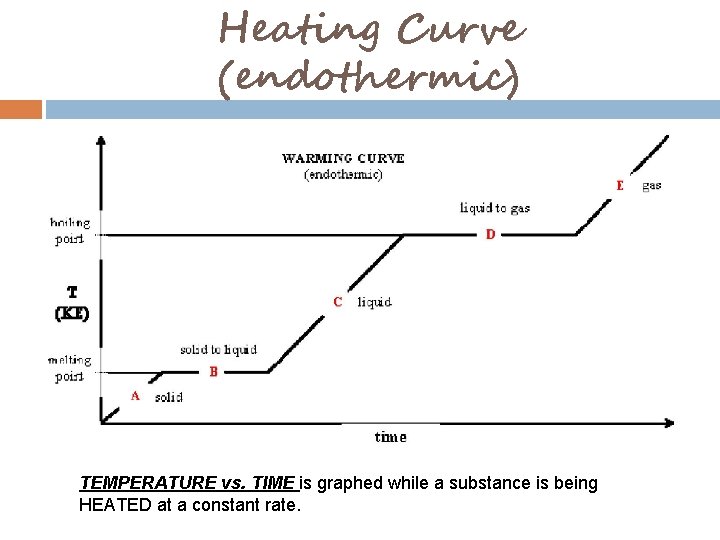
Heating Curve (endothermic) TEMPERATURE vs. TIME is graphed while a substance is being HEATED at a constant rate.
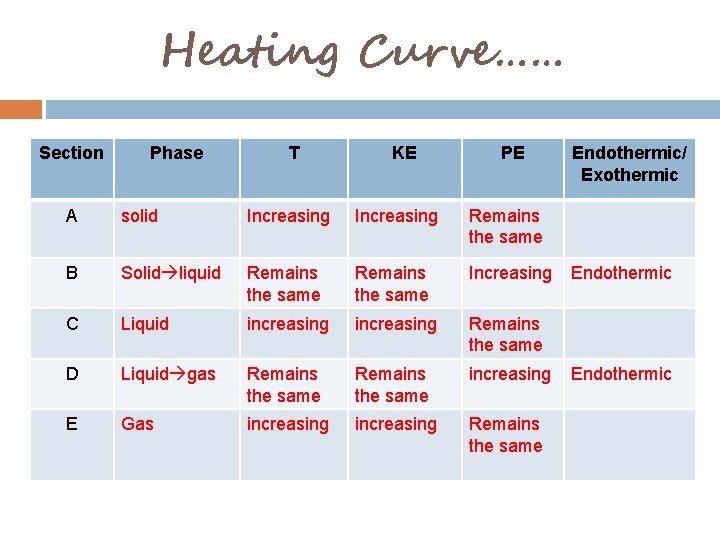
Heating Curve…… Section Phase T KE PE A solid Increasing Remains the same B Solid liquid Remains the same Increasing C Liquid increasing Remains the same D Liquid gas Remains the same increasing E Gas increasing Remains the same Endothermic/ Exothermic Endothermic
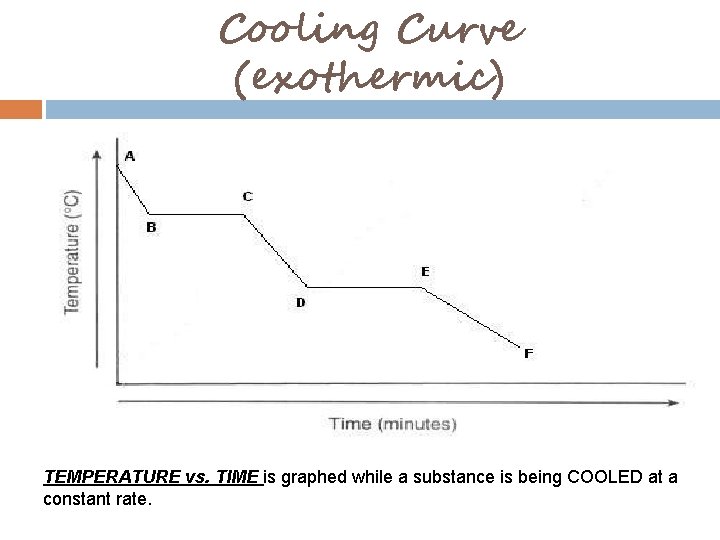
Cooling Curve (exothermic) TEMPERATURE vs. TIME is graphed while a substance is being COOLED at a constant rate.

Cooling Curve…… Section Phase T KE PE AB Gas Decreasing Remains the same BC Gas liquid Remains the same Decreasing CD Liquid Decreasing DE Liquid solid Remains the same EF solid Remains the same Decreasing Endothermic /Exothermic Remains the same Decreasing Remains the same Exothermic
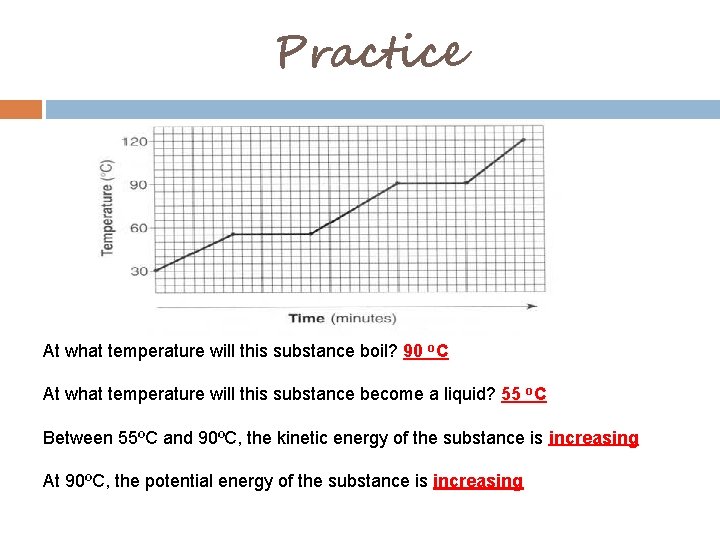
Practice At what temperature will this substance boil? 90 o. C At what temperature will this substance become a liquid? 55 o. C Between 55ºC and 90ºC, the kinetic energy of the substance is increasing At 90ºC, the potential energy of the substance is increasing

Liquid the phase of matter characterized by its constituent particles appearing to VIBRATE about MOVING POINTS.

Pressure Measured in: � Atm atmospheres � k. Pa kilopascals � Torr torres � Psi pounds per square inch � mm. Hg mm of Hg

Gas Pressure Force of the molecule/atom collisions per unit area

Vapor Pressure (Table H): Vapor pressure: the upward pressure exerted by a vapor in equilibrium with its liquid (Pressure a liquid “feels” pushing it to evaporate (turn to gas) • High vapor pressure = Evaporates easily (high volitility) • Example: Ethanol has higher than H 2 O, so it evaporates more easily

Factors that Affect Vapor Pressure Strength of the intermolecular forces: molecules held together by dipole-dipole (polar) have a lower vapor pressure than molecules held together by Van der Waals (nonpolar) 1. 2. example H 2 O does not evaporate as fast as methane (CH 4) Temperature: increase temperature, vapor pressure increases
vapor atmospheric

Evaporation vs. Boiling Evaporation the process by which surface particles of liquid escape into the vapor state (l g) Dependent upon: Nature of the liquid Temperature of the liquid Surface area of the liquid Example: leaving clothing out to dry Boiling Occurs when the vapor pressure of liquid = atmospheric pressure Normal Boiling Point: Temperature when a liquid boils at standard pressure 101. 3 k. Pa or 1 atm (table A) Water Boiling at 23 C

Kinetic Molecular Theory (KMT): A model based on an the concept of an IDEAL GAS which is used to explain gas behavior in terms of the motion of their particles An IDEAL GAS is THEORETICAL and is used to PREDICT the behavior of REAL GASES (O 2, H 2, He, etc. )

Major Assumptions of the KMT q Gas particles have no volume (takes up no space) Gas particles travel in random, continuous, straight line motion Gas particles do not have attractive forces (NO IMF’s) Gas particles have elastic collisions; no energy is lost during a collision

Major Conclusions of the KMT The volume occupied by an ideal gas is the volume of the container The pressure exerted by an ideal gas is related to the number of collisions that the particles make with the walls of the container Temperature of the gas is proportional to the average kinetic energy of the gas

Ideal Gases vs. Real Gases Ideal Gases q Low pressure Real Gases High temperature (fast moving) Low IMF’s (NONPOLAR) Small molecules/atoms Large volume Slow movements are subject to more IMF’s Large molecules/atoms (large particles have high IMF’s) Small volume containers mean more collisions

Think about this Expanding Marshmallows 1. Why did this happen to the marshmallow? Decreased the pressure inside and the marshmallow expanded 2. What is the relationship between pressure and volume? As pressure decreases, volume increases 3. What remained constant in this video? temperature
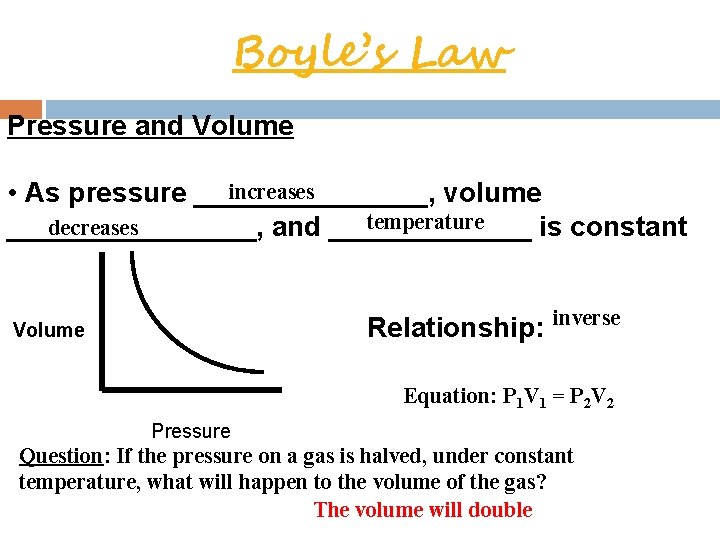
Boyle’s Law Pressure and Volume increases • As pressure ________, volume temperature decreases ________, and _______ is constant Volume Relationship: inverse Equation: P 1 V 1 = P 2 V 2 Pressure Question: If the pressure on a gas is halved, under constant temperature, what will happen to the volume of the gas? The volume will double

Boyle’s Law Practice Problem The volume occupied by a gas at STP is 250 L. At what pressure (in atm) will the gas occupy 1500 L, if the temperature is constant?
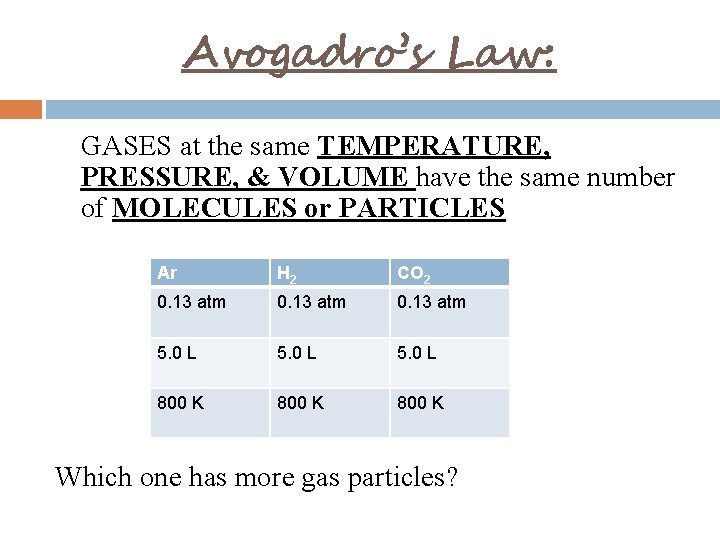
Avogadro’s Law: GASES at the same TEMPERATURE, PRESSURE, & VOLUME have the same number of MOLECULES or PARTICLES Ar H 2 CO 2 0. 13 atm 5. 0 L 800 K Which one has more gas particles?
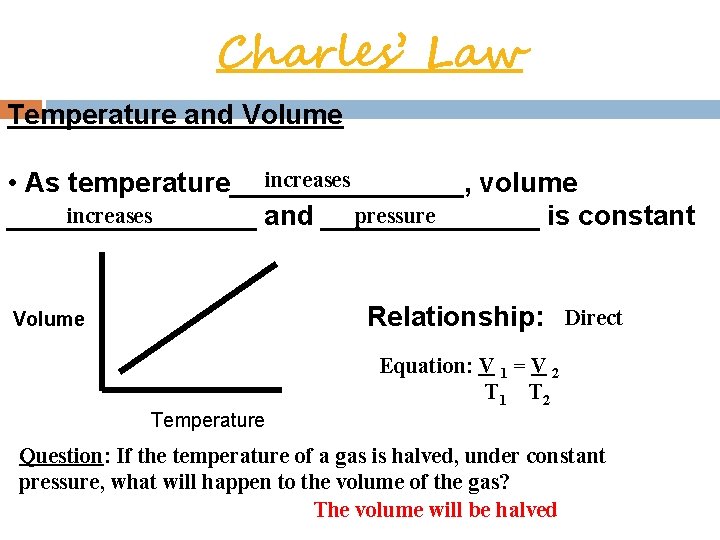
Charles’ Law Temperature and Volume increases • As temperature________, volume increases pressure ________ and _______ is constant Volume Relationship: Direct Equation: V = V 1 T 2 2 Temperature Question: If the temperature of a gas is halved, under constant pressure, what will happen to the volume of the gas? The volume will be halved

Charles Law Practice Problem: The volume of an ideally behaving gas is 300 L at 227°C. What volume will the gas occupy at 27°C when pressure remains constant?

Gay-Lussac’s Law D. Temperature and Pressure increases • As temperature________, increases volume pressure________ and _______ is constant Pressure Relationship: Direct Temperature Equation: P 1 = P 2 T 1 T 2 Question: If the temperature of a gas is tripled, under constant volume, what will happen to the pressure of the gas? The pressure will triple

Gay-Lussac’s Law Practice Problem The pressure exerted by an ideally behaving gas is 700 KPa at 200 K. What pressure does the gas exert at 500 K when volume remains constant?

Combined Gas Law The relationships among pressure, temperature, and volume can be mathematically represented by an equation known as the combined gas law. T • Equation (See Table _____) • When using this equation: P 1 V 1 = P 2 V 2 T 1 T 2 1. Temperature must be in Kelvin, NOT Celsius 2. For pressure and volume, make sure the units of the initial and final are the same 3. If a variable remains constant, you can take it out of the equation

Combined Gas Law 1. The pressure of a gas at 200. K is increased from 200. k. Pa to 305. k. Pa at a constant volume. What is the new temperature? 305 K 2. If a gas at 8. 00 atm is cooled from 600. K to 150. K in a rigid container, what is the final pressure? 2. 00 atm

Combined Gas Law 3. If I have 2. 90 L of gas at a pressure of 5. 00 atm and a temperature of 320. K, what will be the temperature of the gas if I decrease the volume of the gas to 2. 40 L and decrease the pressure to 3. 00 atm? 159 K 4. If I initially have a gas at a pressure of 12 atm, a volume of 23 liters, and a temperature of 250 K, and then I raise the pressure to 14 atm and increase the temperature to 350 K, what is the new volume of the gas? 28 L
- Slides: 46