Physical and Chemical Properties A physical property describes

Physical and Chemical Properties • A physical property describes the look or feel of a substance. © 2013 Pearson Education, Inc.
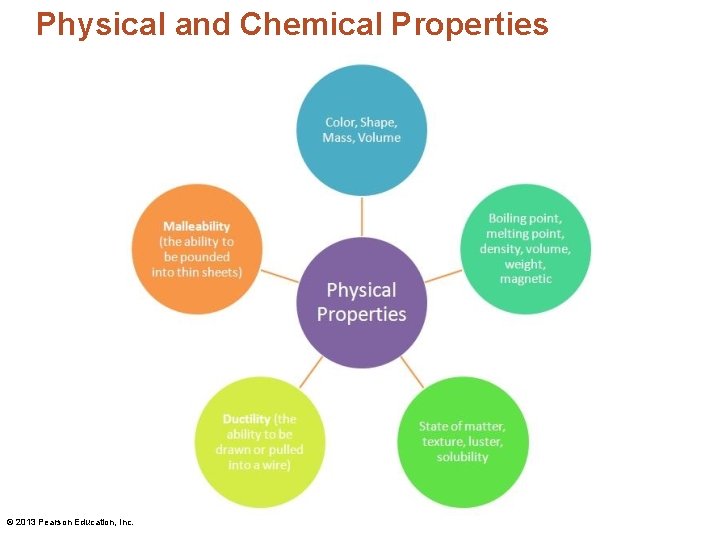
Physical and Chemical Properties © 2013 Pearson Education, Inc.

Physical and Chemical Properties • A chemical property describes the tendency of a substance to transform into a new substance. © 2013 Pearson Education, Inc.

Physical and Chemical Properties It is a chemical property of iron to transform into rust. © 2013 Pearson Education, Inc.

Physical and Chemical Properties © 2013 Pearson Education, Inc.

Physical and Chemical Properties • A physical change is a change in the physical properties of a substance. © 2013 Pearson Education, Inc.
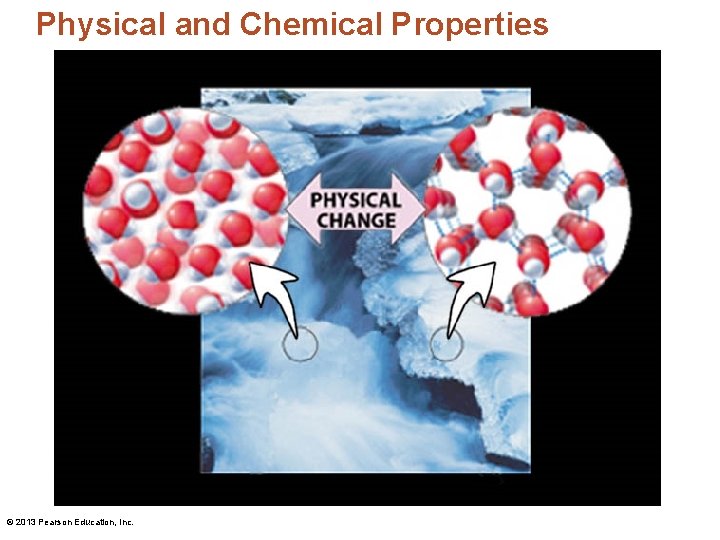
Physical and Chemical Properties © 2013 Pearson Education, Inc.

Physical and Chemical Properties © 2013 Pearson Education, Inc.

© 2013 Pearson Education, Inc.

Physical and Chemical Properties • Chemical properties are properties that characterize the ability of a substance to react with other substances or to transform from one substance into another. © 2013 Pearson Education, Inc.
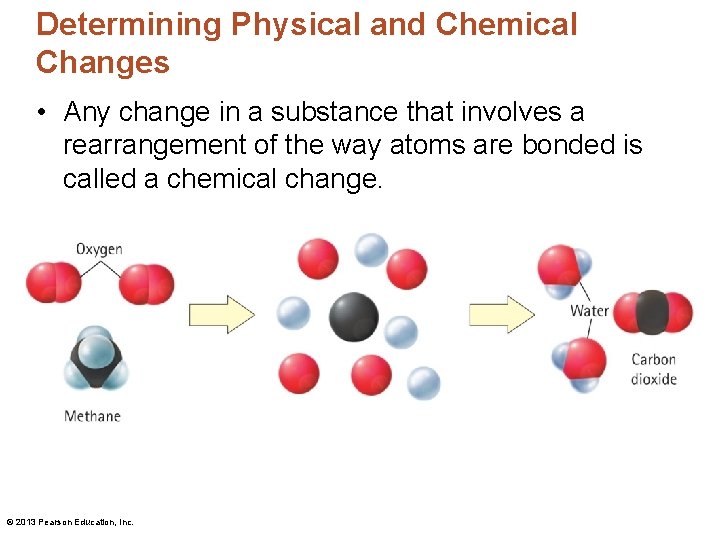
Determining Physical and Chemical Changes • Any change in a substance that involves a rearrangement of the way atoms are bonded is called a chemical change. © 2013 Pearson Education, Inc.
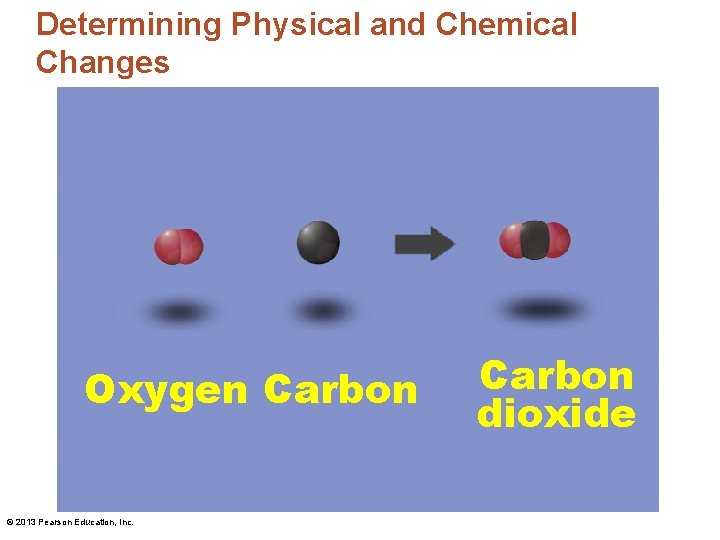
Determining Physical and Chemical Changes Oxygen Carbon © 2013 Pearson Education, Inc. Carbon dioxide

Determining Physical and Chemical Changes • A physical change is a change in the physical properties of a substance. • A chemical change is the transformation of one or more substances into others. – A substance is identified not only by the kinds of atoms it contains but also by how those atoms are connected to one another. – During a chemical change, a new substance is formed as atoms rearrange themselves into new configurations. © 2013 Pearson Education, Inc.

Determining Physical and Chemical Changes © 2013 Pearson Education, Inc.
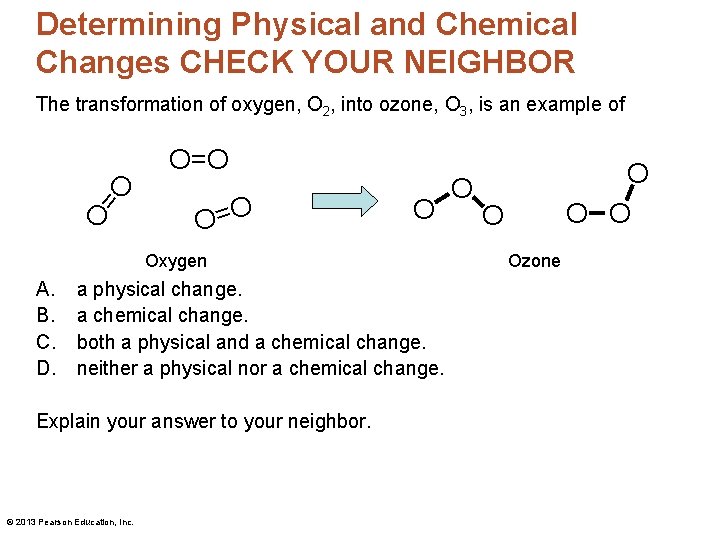
Determining Physical and Chemical Changes CHECK YOUR NEIGHBOR The transformation of oxygen, O 2, into ozone, O 3, is an example of O= O O=O O = O O Oxygen A. B. C. D. a physical change. a chemical change. both a physical and a chemical change. neither a physical nor a chemical change. Explain your answer to your neighbor. © 2013 Pearson Education, Inc. O O Ozone
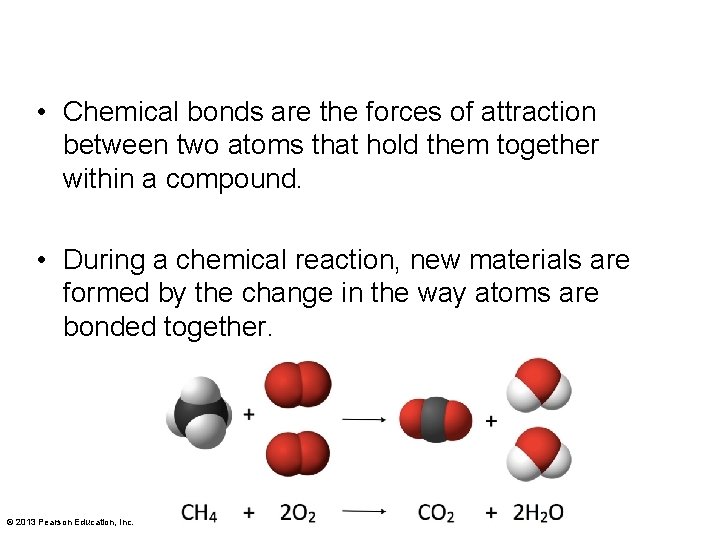
• Chemical bonds are the forces of attraction between two atoms that hold them together within a compound. • During a chemical reaction, new materials are formed by the change in the way atoms are bonded together. © 2013 Pearson Education, Inc.

Determining Physical and Chemical Changes • A physical change imposes a new set of conditions on the same material. • A chemical change forms a new material that has its own unique set of physical properties. • Both physical and chemical changes result in a change in physical appearance. © 2013 Pearson Education, Inc.
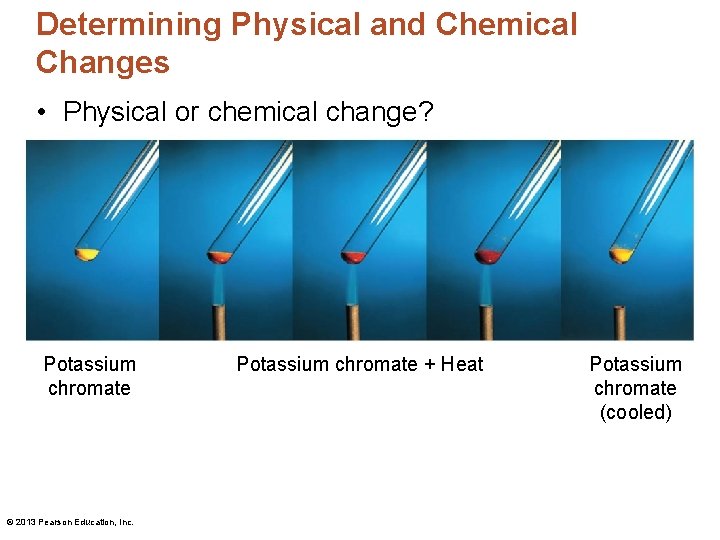
Determining Physical and Chemical Changes • Physical or chemical change? Potassium chromate © 2013 Pearson Education, Inc. Potassium chromate + Heat Potassium chromate (cooled)

Determining Physical and Chemical Changes • Is growing up a physical or chemical change? • Hint: Does this boy look like the peanut butter sandwich he ate in order to grow? © 2013 Pearson Education, Inc.

Determining Physical and Chemical Changes CHECK YOUR NEIGHBOR Melting a piece of solid gold is A. B. C. D. a physical change. a chemical change. both a physical and a chemical change. neither a physical nor a chemical change. Explain your answer to your neighbor. © 2013 Pearson Education, Inc.

Determining Physical and Chemical Changes CHECK YOUR NEIGHBOR Tarnishing a piece of silver is A. B. C. D. a physical change. a chemical change. both a physical and a chemical change. neither a physical nor a chemical change. Explain your answer to your neighbor. © 2013 Pearson Education, Inc.

Elements to Compounds • An element is a material made of only one kind of atom. Pure gold is an element because it is made of only gold atoms. • An atom is the fundamental unit of an element. The term "element" is used when referring to macroscopic quantities. The term "atom" is used when discussing the submicroscopic. © 2013 Pearson Education, Inc.

Elements to Compounds • The elemental formula is used to show the proportion by which atoms combine to form an element. Compound Formula Oxygen O 2 Ozone O 3 Sulfur S 8 Gold Au © 2013 Pearson Education, Inc.
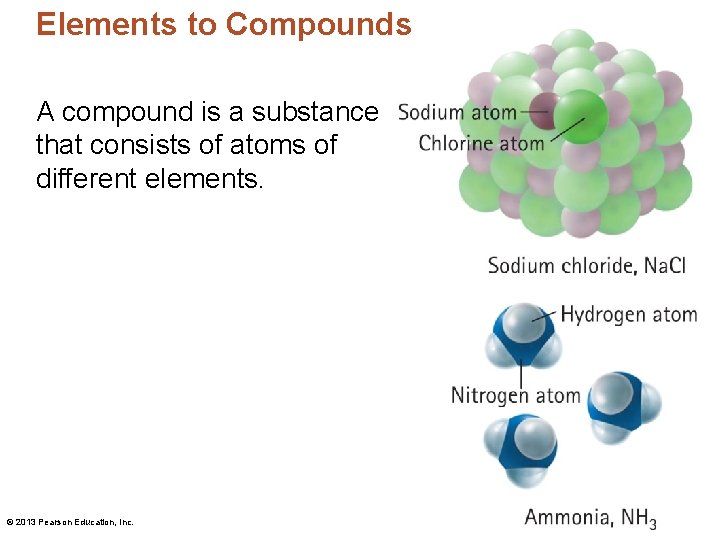
Elements to Compounds A compound is a substance that consists of atoms of different elements. © 2013 Pearson Education, Inc.

Elements to Compounds • Compounds have properties uniquely different from the elements from which they are made. © 2013 Pearson Education, Inc.

Elements to Compounds • A chemical formula is used to show the proportion by which elements combine to form a compound. Compound Formula Sodium chloride Na. Cl Ammonia NH 3 Water H 2 O © 2013 Pearson Education, Inc.

Chemical Symbols • The chemical symbols of the elements are shorthand representations of the elements • J. J. Berzelius, a Swedish chemist is given credit for creating the modern symbols for elements • He proposed that all elements be given corresponding to the first letter of their names.

Chemical Symbols • In case of two elements that began with the same letter, the second letter or an important letter in the name was added. • In some cases the Latin name was used. • So the symbols for Sulfur is S; Selenium is Se; Strontium is Sr; and Sodium is Na (Latin for Natrium)

Chemical Symbols • All the symbols contain the first letter as a CAPITAL letter and the second letter if any is always lower case • Elements 104 -109 have 3 letters • That is because of the international union of pure and applied chemistry. – In this system the Latin and Greek roots are used to name the elements

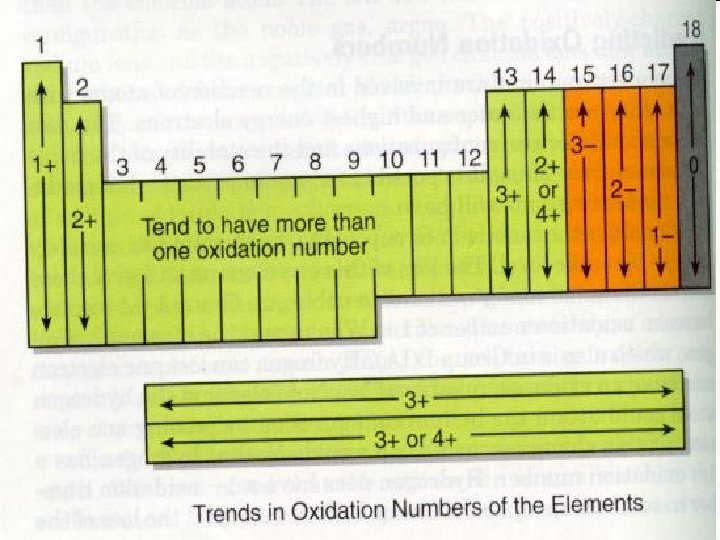
Chemical Symbols • If an atom gains or loses electrons, it must, then, become electrically charged • An atom that has become charged is called an ion • Chemists have a short hand version for writing the charges of an ion. – It is usually placed on the upper right hand corner of the element symbol – The lower left hand corner is the atomic number – The upper left hand corner is the mass number – The lower right hand corner shows the number of atoms

Chemical Symbols • Chemists combine symbols in chemical formulas to represent compounds. • A chemical formula is a combination of symbols that represents the composition of a compound

Chemical Symbols • A formula shows two things: – 1. It indicates the elements present in the compound – 2. It shows the relative number of atoms of each element in the compound • They often contain numbers to indicate the ratio of elements in a compound – Example: water’s formula is H 2 O – The small number 2 means that there are 2 Hydrogen atoms present in water – If there is no subscript that means that there is only one atom of that element present

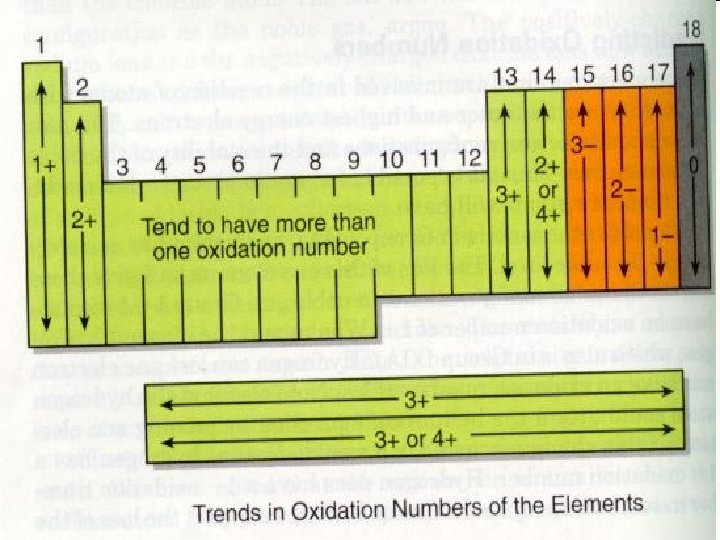
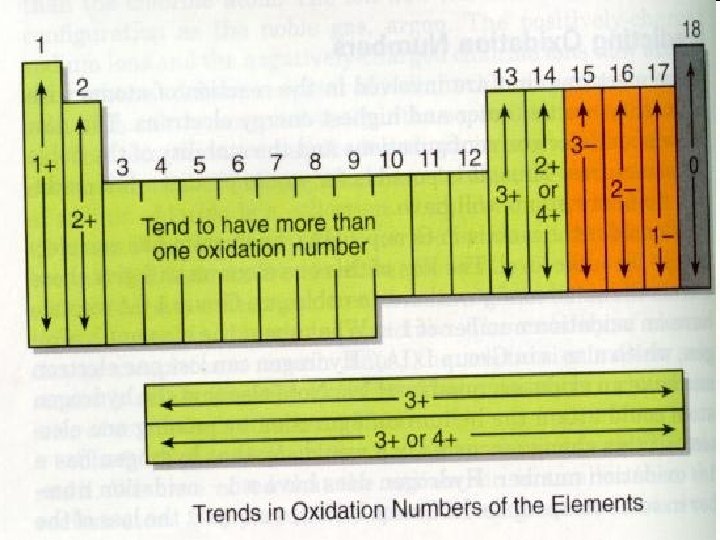
Naming Compounds • Guideline 1 – Start with the element farthest to the left in the periodic table. • Because they are positively charged. – For the element to the right, add the suffix -ide. • They are negatively charged. Example: Na. Cl © 2013 Pearson Education, Inc.
© 2013 Pearson Education, Inc.
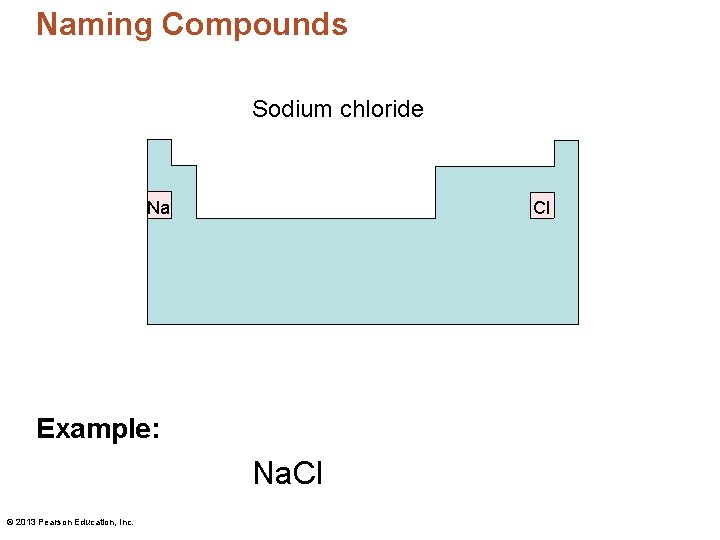
Naming Compounds Sodium chloride Na Cl Example: Na. Cl © 2013 Pearson Education, Inc.

Naming Compounds • Guideline 2 – With different possible combinations of elements, use prefixes to remove ambiguity. © 2013 Pearson Education, Inc.

Naming Compounds monoditritetra- © 2013 Pearson Education, Inc. 1 2 3 4

Naming Compounds Examples: CO carbon monoxide CO 2 carbon dioxide © 2013 Pearson Education, Inc.

Naming Compounds Examples: H 2 O dihydrogen monoxide H 2 O 2 © 2013 Pearson Education, Inc. dihydrogen dioxide

Naming Compounds Examples: H 2 O water H 2 O 2 © 2013 Pearson Education, Inc. hydrogen peroxide

Naming Chemical Compounds • Compounds are named according to the atoms and bonds that compose it. • Based on a set of rules

Naming Chemical Compounds • The rules for naming compound is called the nomenclature of chemistry • For example, Na. HCO 3 is commonly known as baking soda • However, its chemical name is sodium hydrogen carbonate

Naming Chemical Compounds • Compounds containing only 2 elements are called binary compounds. • To name these binary compounds: – First write the name of the element having a positive charge – Second add the name of the negative element • The name of the negative element must be modified to end in –ide.
© 2013 Pearson Education, Inc.

Naming Chemical Compounds • For example Aluminum Al and Nitrogen N combine to form Al. N, this is name aluminum nitride.

Naming Chemical Compounds
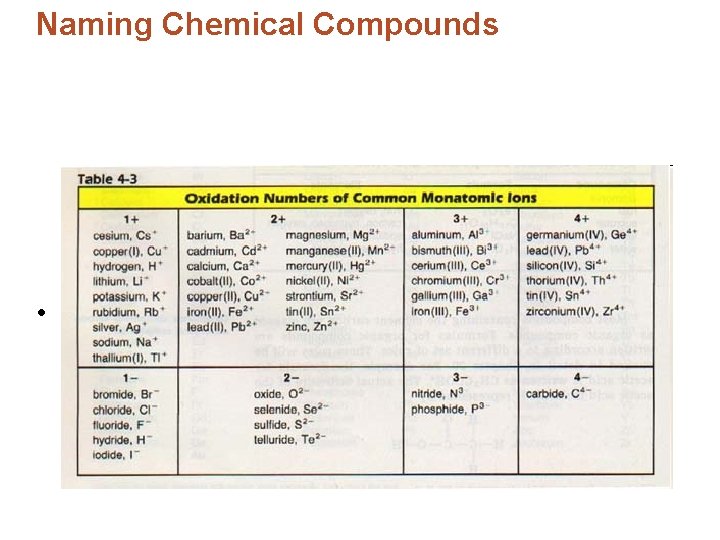
Naming Chemical Compounds • Some elements have more than one charge, therefore, they can form more than one compound with an element

Naming Chemical Compounds • By writing the roman numeral in parenthesis chemists are able to tell the difference between the names of the compounds and which charge is being assigned to that element.

Naming Chemical Compounds

Naming Chemical Compounds • There are many compounds that have been named by an older system in which prefixes indicate the number of atoms present.

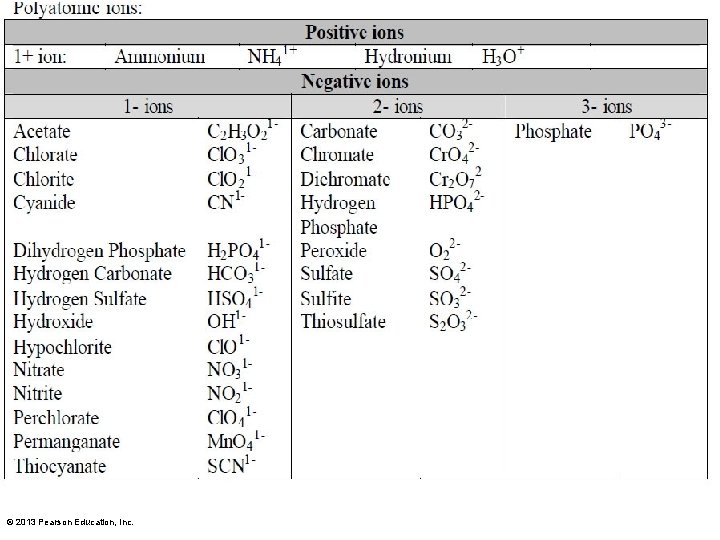
Naming Chemical Compounds • Not all compounds ending in –ide are binary. Look at the polyatomic ions that end in –ide: OH and CN

Naming Ionic Compounds • The name of an ionic compound is determined by the names of its component ions. • Example: – KI – potassium iodide – Mg. Cl 2 – magnesium chloride • The name of the cation comes first. • Cu(NO 3)2 – Copper can exist as 1+ or 2+ – Nitrate has a 1 - charge. Formula shows 2 nitrate ions. – Copper must be 2+ – Name is: Copper (II) Nitrate

• When naming compounds with more than 2 elements they are named the same way as binary compounds. • However, the ending of the name of the polyatomic ion is not changed
© 2013 Pearson Education, Inc.
© 2013 Pearson Education, Inc.

Naming Ionic Compounds • Name: – Cu. NO 3 – Fe. Cl 2 – KNO 3 – Ca(OH)2 – Cu 2 O

Naming Compounds • Guideline 3 – Common names are sometimes used for convenience. © 2013 Pearson Education, Inc.

Naming Compounds CHECK YOUR NEIGHBOR What is the name of the compound with the formula CBr 4? A. B. C. D. chrobrofor See. Ber 4 carbon bromide carbon tetrabromide Explain your answer to your neighbor. © 2013 Pearson Education, Inc.
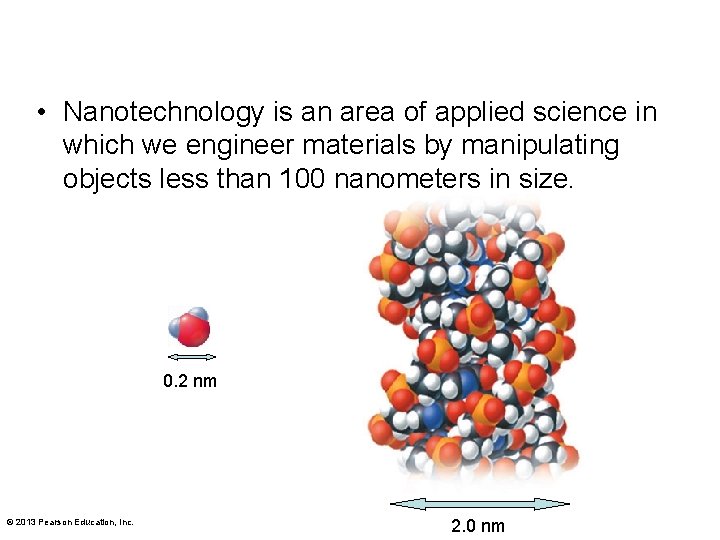
• Nanotechnology is an area of applied science in which we engineer materials by manipulating objects less than 100 nanometers in size. 0. 2 nm © 2013 Pearson Education, Inc. 2. 0 nm

Two main approaches in nanotechnology: 1. Top-down: This approach is an extension of microtechnology techniques to smaller and smaller scales. – A nanosized circuit board, might be carved from a larger block of material 2. Bottom-up: This approach involves building nano-sized objects atom by atom. © 2013 Pearson Education, Inc.
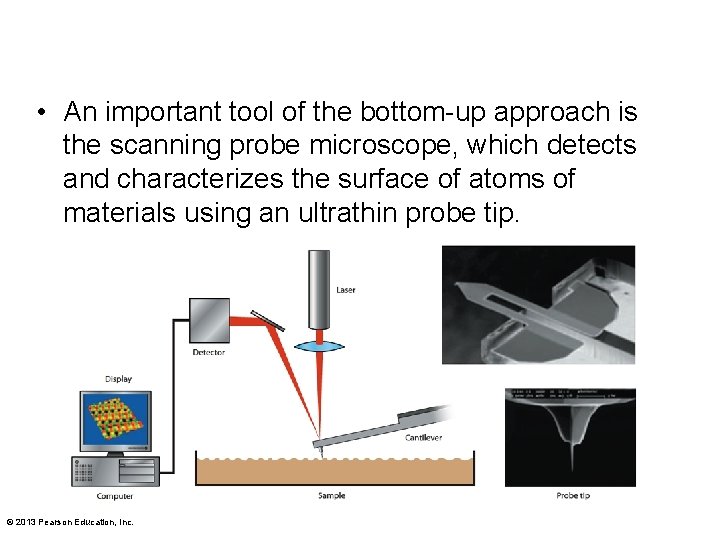
• An important tool of the bottom-up approach is the scanning probe microscope, which detects and characterizes the surface of atoms of materials using an ultrathin probe tip. © 2013 Pearson Education, Inc.

• The tip is mechanically dragged over the surface. • Interactions between the tip and the surface atoms cause movements in a cantilever attached to the tip. • A computer translates the movements into a topographical image © 2013 Pearson Education, Inc.

• Nanotechnology allows the continued miniaturization of integrated circuits need for smaller and more powerful computers. • New tech such as molecular computations (DNA) would have molecules reading, processing and writing information. © 2013 Pearson Education, Inc.

• Nanotechnology will and is opening up new possibilities that before didn’t seem possible. – Ex • • • Color changing paint Solar cells Smart dust Nano robots 3 D printers and photocopiers Medicine © 2013 Pearson Education, Inc.
- Slides: 64