PHYSICAL AND CHEMICAL PHENOMENA Substances have two types

PHYSICAL AND CHEMICAL PHENOMENA Substances have two types of properties: physical and chemical properties. A physical property is any property that is measurable, whose value describes a state of a physical system, for example mass and volume. They are often referred to as observables. Physical properties are often characterized as intensive and extensive. Extensive properties depend on the quantity of the material considered (mass and volume); intensive properties don’t depend on the quantity of the material considered (colour and density).

Chemical properties A chemical property describes the interaction of one substance with another. According to science, a phenomenon is everything that modifies the state of a body. We have two types of phenomena: physical phenomena and chemical phenomena. Physical phenomena are transformations that occur without any changes in the composition of a substance. Chemical phenomena are characterized by internal structural rearrangements. Examples of chemical phenomena are the humidity in the air, or an apple going rotten.

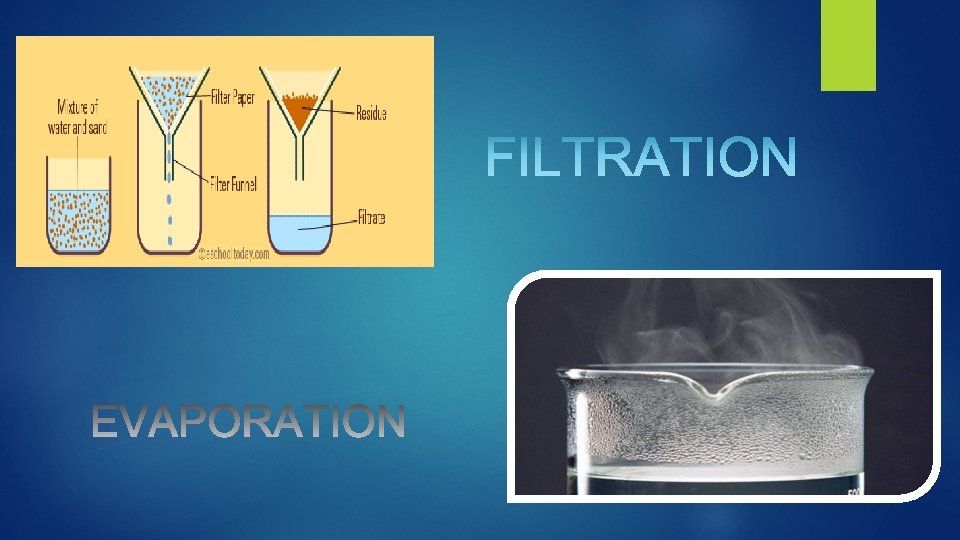
MIXTURES In chemistry, a mixture is a material made up of two or more different substances which are mixed but are not combined chemically. Mixtures have varying physical and chemical properties according to their composition. Ø The atmosphere (a mixture of gases) and sea water (a mixture of salts dissolved in water) are all examples of mixtures. Some mixtures can be separated into their components by using physical (mechanical or thermal) means. Some of the methods used to separate mixtures include:
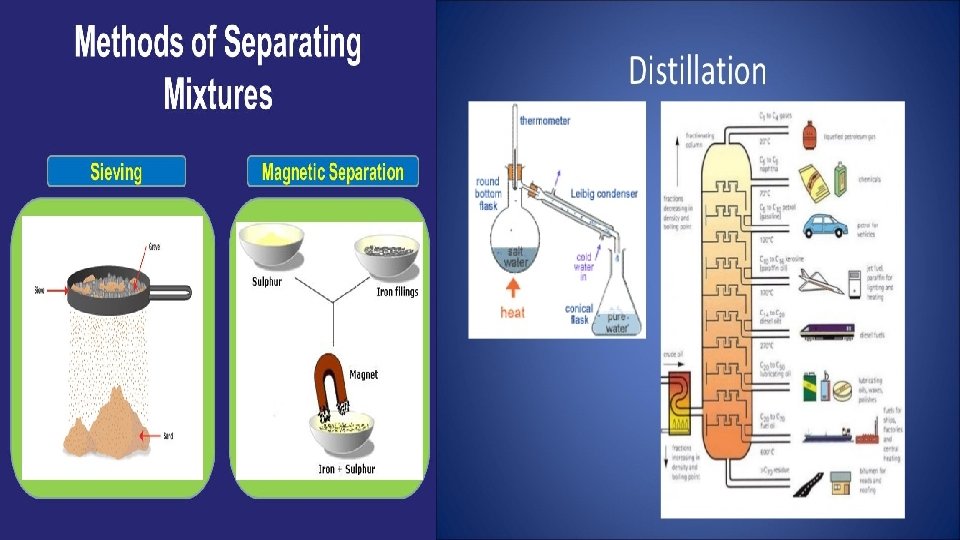
Some of the methods used to separate mixtures include:

HOMEGENEOUS AND HETEROGENEOUS MIXTURES Mixtures can be either homogeneous or heterogeneous. A homogeneous mixture is a type of mixture in which the composition is uniform and every part of the solution has the same properties. Examples of homogeneous mixtures include alloys and alcohol in water. A heterogeneous mixture is a type of mixture in which the components can be seen, as there are two or more phases present. One example of a mixture is air. Air is a homogeneous mixture of the gaseous substances nitrogen, oxygen, and smaller amounts of other substances.
- Slides: 6