Physical and Chemical Changes Pure Substances Mixtures States

Physical and Chemical Changes Pure Substances Mixtures States of Matter

Chemistry • The study of the composition, structure, and properties of matter and the changes it undergoes. • We, the Chemist, ask… – What is that made of? – How does that behave when you change environmental conditions?

Everything that has mass and volume is called matter.
• Weight – depends on gravity – You weigh less on the moon than you do on earth • Mass – stays the same no matter where you are
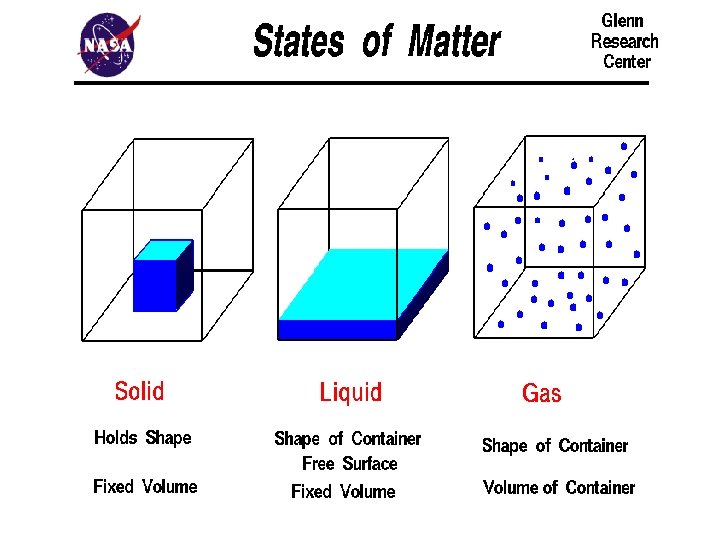

Solids §Particles of solids are tightly packed, vibrating about a fixed position. §Solids have a definite shape and a definite volume. §Solids have an infinite number of free surfaces.

Liquids §Particles of liquids are tightly packed, but are far enough apart to slide over one another. §Liquids have an indefinite shape and a definite volume. §Liquids have one free surface.

Gases §Particles of gases are very far apart and move freely. §Gases have an indefinite shape and an indefinite volume. §Gases have no free surfaces.

Fluids • Gases and liquids • Contain particles that slide past each other

All matter, regardless of state, undergoes physical and chemical changes. These changes can be microscopic or macroscopic.

Physical Change • A change that produces a different appearance of the substance but does not change the composition of the substance

Chemical Change • A process that causes a change in the composition of the substance.

Physical Properties • Physical properties are observable • Examples are: – Mass – Volume – Density – Melting point – Boiling point – Hardness – Physical State – solid, liquid, or gas

Physical Properties Help ID Substances! • A physical property can be used to identify a substance – shape, color, odor, texture • Example: What is… – Round – Orange – Smells rubbery – Bounces – A Basketball!!

Chemical Properties • Chemical properties are related to the elements that make up a substance. • They are not as easy to observe a physical properties • Examples: – Flammability – Reactivity

Physical vs. Chemical Properties • Physical properties are readily observable • Chemical properties can only be observed as the object is changing • Example – you do not know if an object is flammable until you set it on fire!
Is iron transforming into rust a physical or chemical change A. Physical B. Chemical Does the substance change composition or

The color of sulfur is yellow. . A. Physical B. Chemical Does the substance change composition or

Dynamite explodes to form a mixture of gases A. Physical B. Chemical Does the substance change composition or

Aluminum melts at 933 K A. Physical B. Chemical Does the substance change composition or
Plants use CO 2 to make sugar A. Physical B. Chemical Does the substance change composition or

• Physical and chemical properties may be intensive or extensive.

• Intensive properties such as density, color, and boiling point do not depend on the size of the sample of matter.

• Extensive properties such as mass and volume do depend on the quantity of the sample.

• Physical properties are those that we can determine without changing the identity of the substance we are studying.

• The physical properties of sodium metal can be observed or measured. It is a soft, lustrous, silver-colored metal with a relatively low melting point and low density. • Hardness, color, melting point and density are all physical properties.

• Chemical properties describe the way a substance can change or react to form other substances. These properties, then, must be determined using a process that changes the identity of the substance of interest.

• One of the chemical properties of alkali metals such as sodium and potassium is that they react with water. To determine this, we would have to combine an alkali metal with water and observe what happens. • In other words, we have to define chemical properties of a substance by the chemical changes it undergoes.

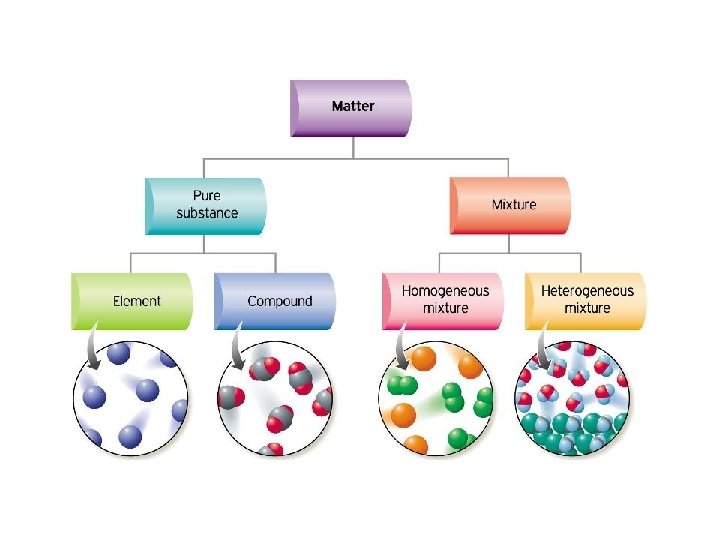
Matter can be identified as either an element, compound, or a mixture.
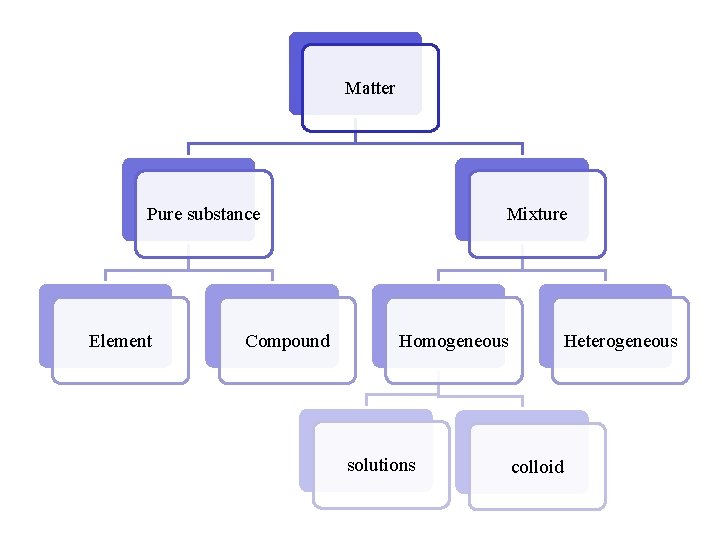
Matter Pure substance Element Compound Mixture Homogeneous solutions Heterogeneous colloid

A pure substance cannot be further broken down or purified by physical means. A pure substance is matter of a particular kind. Each pure substance has its own characteristic properties that are different from the set of properties of any other pure substance.

• Fixed composition • Cannot be separated into simpler substances by physical methods (physical changes) • Can only be changed in identity and properties by chemical methods • Properties do not vary

Compounds • Can be decomposed into simpler substances by chemical changes, always in a definite ratio Ex. H 2 O, HCl, C 6 H 12 O 6 Elements • Cannot be decomposed into simpler substances by chemical changes Ex. Na, Pb, Ca

Mixtures are two or more substances that are NOT chemically combined. Mixtures do not: · Have constant boiling points · Have constant melting points

• Variable composition • Components retain their characteristic properties • May be separated into pure substances by physical methods • Mixtures of different compositions may have widely different properties

Homogenous mixtures look the same throughout but can be separated by physical means (boil, decanter, distill, centrifuge)

Solutions are homogenous mixtures that do not scatter light. These mixtures are created when something is completely dissolved in pure water. Therefore, they are easily separated by distillation or evaporation. Examples: sugar water, salt water

Colloids are homogeneous mixtures. They can be described as a substance trapped inside another substance. They can be identified by their characteristic scattering of light (Tyndall Effect) For example: air trapped inside the fat molecules in whipped cream, milk, fog

Tyndall Effect

• Do not have same composition throughout • Components are distinguishable • Easily separated by physical means Examples: fruit salad, vegetable soup, sand, gravel

Sugar water is considered a _________ A. Heterogeneous mixture B. Homogeneous mixture

Italian dressing is a/an ________ A. element B. compound C. Heterogeneous mixture D. Homogeneous mixture

Carbon dioxide is a/an ________ A. element B. compound C. Heterogeneous mixture D. Homogeneous mixture

Salt water is a/an _________ A. element B. compound C. Heterogeneous mixture D. Homogeneous mixture

Titanium is a/an _______ A. element B. compound C. Heterogeneous mixture D. Homogeneous mixture

Lemonade is a/an _________ A. element B. compound C. solution D. colloid

Mayonnaise is a/an _________ A. element B. compound C. solutions D. colloid

Basic Research • Carried out to for the sake of increasing knowledge • When you research your favorite music groups or study the stats and plays of your favorite football team

Applied Research • Carried out to solve a problem • Research toward discovering a new cancer drug

Quantitative vs. Qualitative Observations Quantitative observations – numerical observations; the mass of the object is 23 g Qualitative observations – non-numerical observations; the liquid is warm

Precision vs. Accuracy Precision – the closeness of a set of measurements of the same quantity made in the same way Accuracy – the closeness of measurements to the correct or accepted value of the quantity measured

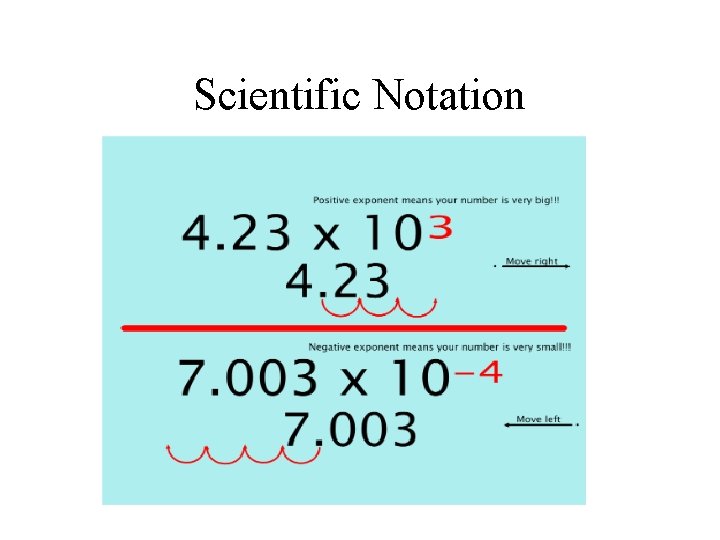
Scientific Notation • A way to express either very large or very small numbers • The coefficient must be greater than 1 or less than 10 • The exponent may be either positive or negative

Scientific Notation Circle the numbers that are in scientific notation. 14. 5 x 108 3. 9 x 10 -4 1. 6984275 x 1014 0. 682 x 10 -1 1. 90 x 100

Scientific Notation

Scientific Notation Converting from standard form to scientific notation. Standard Form Scientific Notation 0. 0047 (original number is less than 1) _______ 22, 598. 7 (original number is greater than 10) _______ 595 ________
Scientific Notation

Scientific Notation Converting from scientific notation to standard form Scientific Notation Standard Form 3. 772 x 104 (exponent is positive) _______ 9. 8 x 10 -3 (exponent is negative) _______ 5. 36042 x 106 _______

Scientific Notations • Performing calculations in scientific notation • ***use the “ee” button on your calculator Example: (3. 4 x 106)(8. 792 x 109) = 2. 99 x 1016 In your calculator, enter “ 3. 4 2 nd ee 6 X 8. 792 2 nd ee 9 enter”

Scientific Notation Practice calculations (5. 44 x 10 -3)(6. 669 x 108) = (1. 72 x 1016)(3. 99 x 10 -5) = (8. 116 x 103)
- Slides: 60