Physical and Chemical Changes Physical and Chemical Properties



















- Slides: 22

Physical and Chemical Changes

Physical and Chemical Properties Everything around us has physical and chemical properties.

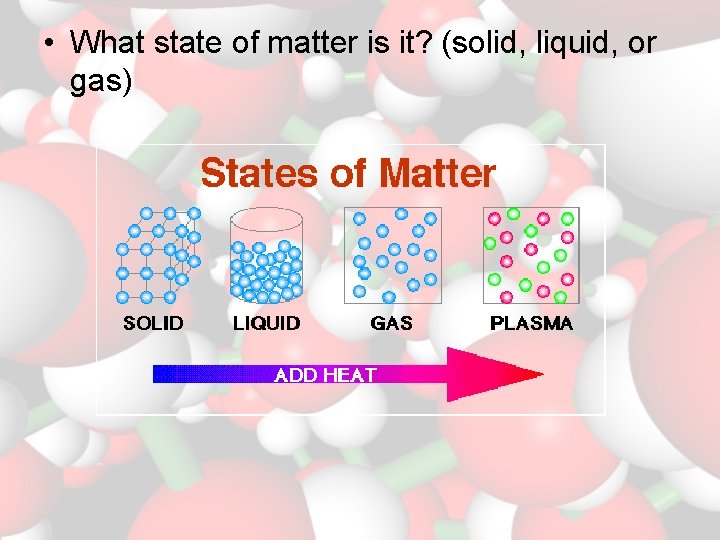
Physical Properties • Characteristics you can observe without changing the substance into something new. – Use your senses to describe – state of matter – measurement

• Use your senses… How does it look, smell, sound, feel, taste (don’t taste it unless the teacher says it’s ok)

• What state of matter is it? (solid, liquid, or gas)

• Measure it…. mass, weight, volume, length, width, height, density, temperature, magnetic?

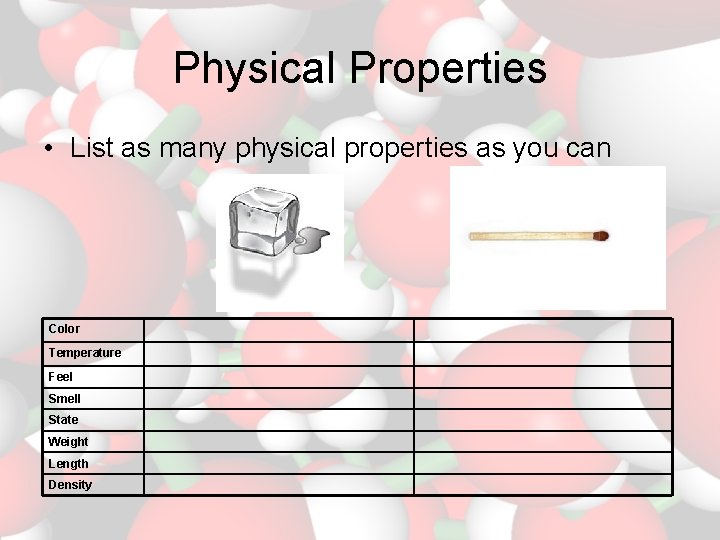
Physical Properties • List as many physical properties as you can Color Temperature Feel Smell State Weight Length Density

Chemical Properties • Characteristics that you can only observe by changing the substance into something new. – You can’t always tell chemical properties just by looking • Will it burn, explode, react with other elements?

Chemical Properties • List as many chemical properties as you can Will it burn Will it explode Can it react with other things

Physical and Chemical Changes Matter around us can undergo changes. Physical Changes Chemical Changes

Physical Change • A change in which the state or energy level of something changes but not its chemical composition – Change of state – Change of shape – Dissolving

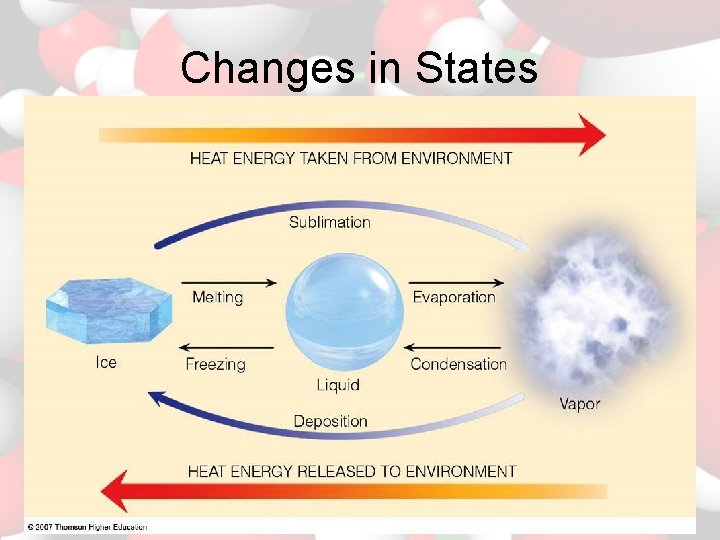
Physical Changes changing state melting – solid to liquid condensation – gas to liquid freezing – liquid to solid sublimation – solid to gas vaporization – liquid to gas deposition – gas to solid

Changes in States

Physical Changes change of shape Matter can change shape cutting chewing grinding crumpling bending

Physical Changes dissolving When you mix or dissolve one thing into another, it is a physical change

Chemical Change • A change in which the composition of matter changes and new substances are formed. Chemical bonds are formed and broken. – It cannot be turned back into the original substance

Chemical Change • Signs that a chemical change may have taken place – – – Color change Absorption or release of energy Odor Formation of gases or solids Can’t be reversed

Chemical Changes Iron + oxygen and water = rust silver + oxygen and water = tarnish

Chemical Changes Burned wood Cooked food

Chemical Changes Digestion Spoiled food

Chemical Changes Lightning bug Antacid tablet

Physical or Chemical orange copper Orange juice Orange cake Physical Chemical Copper jewelry Physical Statue of Liberty Chemical