Physical and Biological Factors that affect the water

Physical and Biological Factors that affect the water quality Unit 1: Hydrosphere

Warm Up: Read the multichoice questions and select the correct answer. Copy your answers in the warm up section. 1. Which best explains how estuaries support plant and animal life? a. The dry land removes pollutants. b. The water provides energy for growth. c. The habitat area provide nutrients and shelter. d. The cold temperatures decreases dissolved oxygen. 2. Which contributes most in supporting life on the deep ocean floor? a. Cold water temperatures b. Extreme water pressure c. Hydrothermal vents d. Lack of predators 3. Why are upwelling in the ocean essential for some organisms to survive? a. Because many animals need low-tide conditions each day b. Because currents carry predators away from the surface c. Because the warm water kills bacteria before it can harm plankton d. Because needed nutrients and dissolved gases are brought into new areas 4. Which contains the most water? a. lakes c. oceans b. rivers d. streams

Water Quality ● Water quality is a measure of the physical, chemical and biological factors that affect a body of water. ● Water quality often determines the number and types of organisms that can live in a water system. ○ Scientist use field and laboratory equipment to analyze the water.

Physical and Biological Factors ● The health of a water system is determined by the balance between: Physical variables - Temperature - Turbidity - Water movement Chemical variables - Dissolved oxygen and other gases - p. H - Nitrates - Salinity - Phosphorus Biological variables - Bioindicator or macroinvertebrates

1. Temperature ● The temperature of water in rivers and lakes determines the kinds of organisms that can survive there. ● Why it is important? : ○ Aquatic organisms depend on certain temperature range for optimal health. ○ Also affects many other variables: types of plants and animals, dissolved O 2, and the susceptibility of organisms to parasites, pollution, and diseases. ● Cold H 2 O = hold more dissolved O 2 ● Warm H 2 O = hold less dissolved O 2 ● Factors influencing it: ○ whether conditions - shade vs. sunlight ○ waste in runoff from urban sources or groundwater inflow

Temperature ● Ice at the air-water interface can lead to lower dissolved oxygen levels in the winter.

2. Turbidity ● Turbidity is a measure of how clear water is. ○ The more suspended solids there are in a water sample, the less transparent it is. ● High turbidity ○ is not a favorable sign ○ can be associated with organic pollution ○ can lead to increased water temperatures, low dissolved oxygen, and even physical impairment of aquatic organisms

Turbidity

Turbidity ● How it affects temperature? ○ High turbidity absorbs more heat, making H 2 O temperature to rise. ○ Low turbidity, H 2 O cooler. ● Relation to dissolved oxygen: ○ Too cloudy H 2 O = difficult for plants to perform photosynthesis ● Impact on organisms living in high turbidity. ○ A lot of sediments can clog into the gills. ○ Increase dissolved O 2

3. Dissolved Oxygen ● Dissolved Oxygen (DO) is a measure of the amount of oxygen in water that is available for chemical reactions and for use by aquatic organisms. ● DO balance in water is important for the survival of certain microorganisms and zooplankton. ● How the DO gets in the water? ○ Through diffusion with the air ○ Waste product of photosynthesis by aquatic plants and phytoplankton. ● DO levels vary with seasons and over 24 hr cycles.

Dissolved Oxygen ● High levels of DO = better and water taste better. ● Low levels of DO = sensitive animals may move away, weaken, or die.

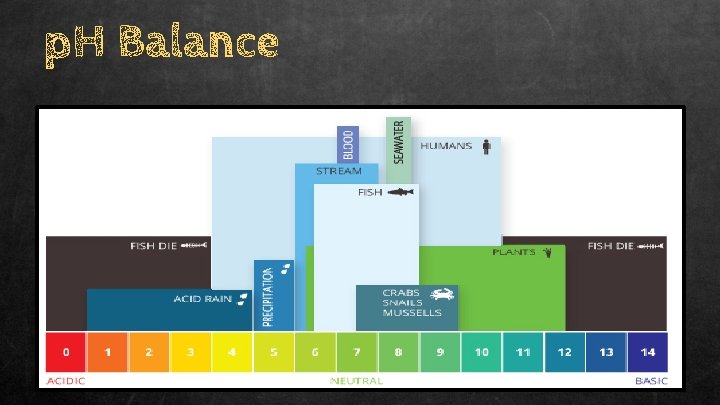
4. p. H Balance ● p. H level is a measure of how acidic or basic water is. ○ Is measured on scale that ranges from 0 to 14, with 7 considered neutral. ○ Acidic = less than 7 ○ Basic = higher than 7

p. H Balance ● Neutral water = 7 ● Swampy water = 3 - 5 ● Marine water = 7 - 9 ● Factors that influence the p. H of water: Too acidic Too basic Things like acidic rain Overgrowth of algae Too much nitrogen
p. H Balance

5. Nitrogen and Phosphorus ● Nitrates and phosphorus are essential plant nutrients. ○ Are chemicals that pose possible health risks to humans if their presence in drinking water is not controlled. ● The major sources of nitrates in surface water include runoff contaminated with fertilizers, septic tank leakage, sewage, and erosion of natural deposits. ● Phosphates usually enter waterways from human and animal waste, laundry, cleaning and industrial effluents.

Nitrogen and Phosphorus ● Harmful to: ○ Body of water ■ Much growth of aquatic plants ■ Fish dies ■ Eutrophication is excessive richness of nutrients in a lake or other body of water, frequently due to runoff from the land, which causes a dense growth of plant life and death of animal life from lack of oxygen. ○ Humans ■ Restrict the supply of O 2

Nitrogen and Phosphorus A sudden population growth in algae is called an algae bloom. Alga blooms cloud water and deplete the water of oxygen (hypoxia) that animals need to survive.

6. Bio-indicators are organisms the scientists use to monitor the health of an ecosystem. ○ Macroinvertebrates are organisms that don’t have a backbones and are often used as bio-indicators because they don’t move around much, easy to see, and have long life cycles. ○ Physical and chemical changes that occur in the organisms are a sign that a problem may exist.

Bio-indicators ● Examples of information offered by these organisms: 1. Early warning of environmental damage 2. Environmental stressor on health 3. Organism affected by pollution 4. Potential harm human 5. Effectiveness of help clean H 2 O ● Why they are important? ○ They measure the presence and abundance of different kinds of algae and other indicators of H 2 O quality.

Activity: Piecing it Together Instructions: 1. Each piece of the puzzle represent a factor that affect the quality of the water. 2. With the information from the slide show presentation, answer the questions in each piece of the puzzle. 3. Draw and color each piece. 4. Cut and paste each piece between pages 22 - 23 in your interactive notebook.
- Slides: 20